Introduction
Metabolic dysfunction-associated steatotic liver disease (MASLD), formerly called non-alcoholic fatty liver disease (NAFLD), is a progressive condition that can lead to cirrhosis and hepatocellular carcinoma [1, 2]. MASLD is a common liver disease worldwide and one of the leading causes of liver transplantation in the U.S.A. [3]. Additionally, MASLD is associated with life-threatening events, including coronary artery diseases and cerebral infarction [4]. While treatment options for MASLD are limited, maintaining a healthy lifestyle remains top priority for managing patients with MASLD.
It is well known that MASLD patients experience significant health and economic burdens [5]. Consequently, these patients report poor health-related quality of life (HRQoL) in both physical and psychological aspects [6]. This poor HRQoL is associated with MASLD-related symptoms, including fatigue and abdominal pain. Furthermore, it is related to higher healthcare resource use and lower work productivity, including higher rates of absenteeism, presenteeism, and overall work impairment. These negative effects likely begin at an early stage of MASLD and increase with disease progression. Therefore, it is crucial to address these burdens from an early stage of the disease.
Patients with MASLD have a high prevalence of periodontal diseases [7]. Periodontal diseases are chronic inflammatory conditions caused by periodontopathic bacteria. They begin with gingivitis, an inflammation of the gum, and can progress to periodontitis, which involves resorption of the bone surrounding the tooth. As the disease advances, the tooth-supporting apparatus is destroyed, potentially leading to tooth loss [8]. Ultimately, oral functions deteriorate as a result of sustained periodontal diseases. Given these factors, patients with MASLD likely experience a significant oral health burden, which may reduce their oral health-related quality of life (OHRQoL). However, there is limited information on OHRQoL and oral functions in patients with MASLD.
In the present study, we examined OHRQoL using the General Oral Health Assessment Index (GOHAI), a widely used questionnaire. Additionally, we investigated oral functions, including the numbers of functional and dysfunctional teeth, and explored the association between oral functions and MASLD.
Material and methods
Subjects
Patients with MASLD were recruited at Jichi Medical University Hospital from 2020 to 2022. MASLD was defined according to published criteria [1] and ultrasonographic examination was used for diagnosis of steatosis. Exclusion criteria encompassed participants who consumed over 30 grams of alcohol per day for men or 20 grams per day for women, as well as those with other liver diseases. Advanced liver fibrosis, including liver cirrhosis, was also excluded due to its high complication rate with periodontitis [9]. Advanced liver fibrosis was determined by morphological changes of the liver and clinical parameters, including fibrosis-4 (FIB-4) index > 2.67 or Wisteria floribunda agglutinin-positive Mac-2 binding protein glycosylation isomer (M2BPGi) 2.4. Control subjects (CSs) were recruited at Jichi Medical Health Check-up center and at a single manufacturing company. CSs were defined as participants who did not receive any medications. Additionally, individuals with a fatty liver index (FLI) over 60 were excluded from the control group. Laboratory data for patients with MASLD and CSs were obtained from medical records in Jichi Medical University Hospital, the Health Check-up Center at Jichi Medical University, and the health promotion office at the company. Written informed consent was obtained from all participants. The present study was approved by the Institutional Review Board of Jichi Medical University Hospital (permission number A19-070) and conducted in accordance with the Declaration of Helsinki.
Oral examinations
All participants underwent oral examinations at the Oral and Maxillofacial surgery division of Jichi Medical University Hospital. The examinations included OHRQoL assessment and objective evaluations of oral conditions, including oral hygiene, periodontal disease, caries, number of teeth, and oral functions. The GOHAI was used to assess OHRQoL [10]. The GOHAI measures the degree of restriction in physical and psychosocial aspects of life due to oral-related difficulties across three domains: oral function, psychosocial impact, and pain and discomfort.
The evaluation is based on a total score of 12 items, each scored between 1 and 5 points, with a maximum score of 60 points and a minimum score with 12. A Japanese-translated version of the questionnaire was used in the present study.
The following examinations were performed by one dentist and one dental hygienist engaged in clinical practice there. Oral hygiene was assessed using the simplified debris index (DI-S), the simplified calculus index (CI-S), and the simplified oral hygiene index (OHI-S) [11, 12]. These indices were measured on six representative teeth, with scores from 0 to 3 (Supplementary Fig. 1A, B). The DI-S and CI-S were calculated by the total scores of plaque (debris) and tartar (calculus) divided by 6 (DI-S, CI-S: maximum 3 points, minimum 0 points). The OHI-S was the sum of DI-S and CI-S (maximum 6 points, minimum 0 points). The tongue coating index (TCI) was measured as an additional oral hygiene status indicator, since the tongue coating consists of bacteria, damaged epithelial cells, food remnants, and leucocytes [13]. The dentist divided the tongue surface into 9 blocks according to the protocol and assigned a 3-point score (0-2) for the extent of tongue coating in each block (Supplementary Fig. 1C). The total score of each block was divided by 18 to calculate the TCI [14]. Periodontal health was evaluated by measuring probing pocket depth (PPD) and bleeding on probing (BOP) according to a guideline available at (https://www.perio.jp/publication/upload_file/guideline_perio_2022_en.pdf) (accessed May 10, 2024). Briefly, PPD was defined as the distance from the gingival margin to the base of the clinical periodontal pocket (Supplementary Fig. 2A, B). PPD was measured at six sites per tooth for all teeth. The presence of bleeding during probing was also recorded (Supplementary Fig. 2B). Mean PPD, number of sites with PPD ≥ 4 mm, and frequency of BOP positive sites were calculated for each subject. Periodontal inflamed surface area (PISA) and periodontal epithelial surface area (PESA) were also determined [15]. The DMFT (decayed, missing, and filled teeth) index was calculated according to the World Health Organization criteria. The numbers of natural teeth (excluding the third molars) and functional teeth (that can be used for biting, not including mobile teeth, residual roots, and prosthetically restored missing teeth, such as dentures, implants, or bridges) were recorded. Several tests were conducted to assess oral function: Salivary collection time, an indicator for dry mouth, was defined as the time to collect 1 mL of unstimulated saliva. Chewing ability was measured with a gummy jelly test [16]. The test food was a cylindrical-shaped gummy jelly with a diameter of 14 mm, height of 10 mm, and weight of 2.3 g. Participants were asked to chew the test food for 20 seconds without swallowing it and then hold 10 ml of water in their mouth before spitting it out together with the jelly. Glucose eluted from the chewed jelly into the water was measured using a Gluco Sensor GS-II (GC Corporation, Tokyo, Japan). Tongue pressure was measured using a JMS tongue pressure measuring device (TPM-02, JMS Co., Ltd., Hiroshima, Japan) [16]. This device consists of a probe, connecting tube, and main body. The probe has a 25-mm long, 15-mm wide balloon. Participants were asked to sit in a relaxed position, place the balloon in their mouth, and hold the plastic cylinder at the midpoint of the central incisors with their lips closed. The examiners also held the probe to keep it in the correct position. Participants were then asked to raise their tongue and compress the balloon against the hard palate for 7 seconds with maximum voluntary force. Oral-lip motor function was assessed by oral diadochokinesis (ODK) for the /pa/, /ta/ and /ka/ sounds [16]. Participants were asked to articulate each syllable repetitively as quickly as possible for 5 seconds. The syllables allowed for assessment of the three major articulatory organs: the lips, the tongue tip, and the tongue dorsum. Articulation counts were measured using a digital counter (Kenkou-kun Handy, Takei Scientific Instruments Co., Ltd., Tokyo, Japan), and oral diadochokinesis was calculated separately for each syllable as the articulation count per second.
Statistics
Categorical variables were expressed as numbers (percentage), and continuous variables as mean ± standard deviation. Differences between groups were examined using Pearson’s chi-square test for categorical variables or the Mann-Whitney U test for continuous variables. The Kolmogorov-Smirnov test was used to confirm normality of the data distribution. Because data were not normally distributed, differences between groups were assessed using the Mann-Whitney U test. Single regression analyses were performed to examine the association between MASLD and oral conditions. Multivariate binary logistic regression analysis was used to identify independent predictors of MASLD. Statistical analyses were conducted using SPSS Statistics version 28 (IBM Corp., Armonk, NY, USA). A value of p < 0.05 (*) was considered statistically significant, while p < 0.10 (†) was considered potentially informative.
Results
Background of subjects
Twenty-one patients with MASLD and 31 CSs participated in the present study. Two patients with MASLD and 5 CSs were excluded based on the entry criteria (Fig. 1). Ultimately, 19 patients with MASLD and 26 CSs were analyzed. The characteristics of participants are shown in Table 1. There was no significant difference in age between the two groups. Patients with MASLD were characterized by a high body mass index, high frequency of dyslipidemia, and elevated transaminase levels. No significant difference in fibrosis-4 (FIB-4) index was observed between CS and MASLD groups. Additionally, the mean level of M2BPGi, a marker for liver fibrosis, was 0.72 (±0.21) in patients with MASLD. The maxima of the FIB-4 index and M2BPGi in the MASLD group were 1.9 and 1.24, respectively. Thus, the MASLD group did not include patients with advanced fibrosis.
Table 1
Demographic characteristics of subjects
| Variable | All (N = 45) | CSs (n = 26) | MASLD (n = 19) | p-valuea |
|---|---|---|---|---|
| Age (years) | 50.8 ±12.1 | 48.4 ±11.2 | 54.1 ±12.8 | 0.202 |
| Sex (female) | 17 (37.8%) | 9 (34.6%) | 8 (42.1%) | 0.757 |
| BMI (kg/m2) | 24.5 ±4.9 | 22.1 ±3.2 | 27.9 ±5.1 | < 0.001 |
| TG (mg/dl) | 104.7 ±51.2 (44)b | 84.0 ±41.9 | 134.6 ±49.6 (18)b | 0.001 |
| HDL-C (mg/dl) | 61.2 ±16.5 (44)b | 65.7 ±16.3 | 54.8 ±15.0 (18)b | 0.024 |
| LDL-C (mg/dl) | 125.4 ±27.1 (44)b | 125.3 ±26.5 | 125.6 ±28.7 (18)b | 0.981 |
| AST (IU/l) | 28.2 ±24.1 | 20.7 ±8.5 | 38.5 ±33.6 | < 0.001 |
| ALT (IU/l) | 36.0 ±40.4 | 17.7 ±9.7 | 61.1 ±52.1 | < 0.001 |
| γ-GTP (IU/l) | 50.9 ±59.0 | 29.5 ±20.3 | 80.2 ±79.8 | 0.001 |
| FIB-4 index | 1.1 ±0.4 (44)b | 1.1 ±0.4 (25)b | 1.1 ±0.4 | 0.943 |
a p < 0.05, using Mann-Whitney U-test (for continuous variable) or chi-square test (for categorical variables)
b For each variable with missing data, the number of subjects with complete data is shown in parentheses
CSs – control subjects, MASLD – metabolic dysfunction associated steatotic liver disease, BMI – body mass index, TG – triglycerides, HDL-C – high-density lipoprotein cholesterol, LDL-C – low-density lipoprotein cholesterol, AST – aspartate aminotransferase, ALT – alanine aminotransferase, γ-GTP – gamma-glutamyl transpeptidase, FIB-4 index – Fibrosis-4 Index
Patients with MASLD had low OHRQoL
We first investigated OHRQoL using the GOHAI. The total GOHAI score was significantly lower in patients with MASLD (Fig. 2A), indicating poor OHRQoL. Among GOHAI components, the physical score was significantly low. Pain and discomfort scores also tended to be lower in the MASLD group. In contrast, there was no significant difference in total score of psychological components. Within the physical functioning questionnaire, eating and talking components were significantly lower in the MASLD group (Fig. 2B). In the psychological questionnaire, patients with MASLD reported worrying about being noticed by others due to oral problems, although they did not hesitate to eat in public (Fig. 2B).
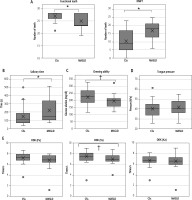
Low OHRQoL in physical functioning is supported by decreased numbers of functional teeth in patients with MASLD
As patients with MASLD reported low oral function in the OHRQoL assessment, we investigated the status of teeth, salivary secretion time, chewing ability, tongue pressure, and tongue-lip motor function. Patients with MASLD reported difficulty in biting and eating, and changes in food type and volume (Fig. 2B). Supporting these findings, the number of functional teeth was significantly lower in patients with MASLD (Fig. 3A). Additionally, patients with MASLD had a higher number of DMFT. Furthermore, patients with MASLD exhibited longer salivary secretion time (Fig. 3B). In contrast, actual chewing ability values were within normal limits, despite a tendency of lower chewing ability in the MASLD group (Fig. 3C). There was no significant difference in tongue pressure between CS and MASLD groups (Fig. 3D). Patients with MASLD reported difficulty in fluent speech (Fig. 2B). In the ODK assessment, where subjects repeated vocalization of “pa”, “ta”, and “ka”, the MASLD group showed a tendency for less frequent pronunciation of “ta”, but no significant differences were observed in other pronunciations (Fig. 3E).
Fig. 3
Assessment of oral function. A) Number of functional teeth and decayed, missing, and filled teeth (DMFT). B) Salivary time. C) Chewing ability. Glucose elution < 100 mg/dl is considered as low chewing ability. D) Tongue pressure. Pressure < 30 kPa is considered as low tongue pressure; E) ODK (Pa), ODK (Ta), and ODK (Ka). Less than 6 times/s is considered as low ODK
Oral hygiene and periodontal conditions in MASLD patients
Given that poor periodontal conditions contribute to tooth loss, we investigated oral hygiene and periodontal conditions in patient with MASLD. OHI-S was significantly higher in the MASLD group (Fig. 4A), indicating poorer oral hygiene status compared to CSs. Among the OHI-S components, the DI-S was notably elevated in MASLD patients (Fig. 4A). Additionally, the median TCI was significantly higher in the MASLD group (Fig. 4B). These data consistently demonstrated that MASLD patients had poorer oral hygiene across multiple assessments.
Fig. 4
Assessment of oral hygiene and periodontal conditions. A) OHI-S, DI-S, and CI-S; B) TCI; C) Number of sites with PPD ≥ 4 mm and mean PPD; D) BOP (%); E) PISA; F) PESA
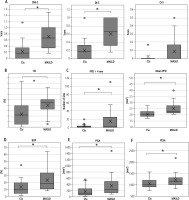
We then examined periodontal condition using simplified diagnostic criteria from the World Health Organization. The prevalence of periodontitis, defined as at least one pocket with PPD ≥ 4 mm, was not significantly different between CSs (17 subjects, 65.4%) and MASLD (15 patients, 78.9%) groups. MASLD group had a higher proportion of severe periodontitis (at least one pocket in which PPD ≥ 6 mm). The numbers of participants with severe periodontitis were similar: 3 (11.5%) in CS and 4 (21.1%) in MASLD. Thus, we additionally performed detailed analyses. MASLD patients had a higher number of sites with PPD ≥ 4 mm (Fig. 4C). Mean PPD was deeper in the MASLD patients (Fig. 4C). Additionally, BOP frequency was higher in MASLD patients (Fig. 4D). As a result, the PISA and PESA were larger in the MASLD group (Fig. 4E, F). These results indicate that MASLD patients had poorer periodontal conditions overall.
Association between functional teeth and MASLD
While periodontal diseases have been associated with the development of MASLD, little information exists on the relationship between tooth number and MASLD. Logistic regression analysis, with MASLD as the dependent variable, is shown in Table 2. In the unadjusted model, higher body mass index (BMI) was significantly associated with the presence of MASLD (higher; OR = 1.475, 95% CI: 1.160-1.874) and a greater number of functional teeth was negatively associated with MASLD (OR = 0.715, 95% CI: 0.539-0.948). In the adjusted model, a greater number of functional teeth remained negatively associated with MASLD (OR = 0.407, 95% CI: 0.204-0.813). These findings suggest that a decreased number of functional teeth is independently associated with MASLD, even after accounting for periodontal disease.
Table 2
Unadjusted and adjusted odds ratios and 95% CI for MASLD
Discussion
To the best of our knowledge, the present study is the first to report that patients with MASLD have low OHRQoL. Additionally, MASLD patients showed a decreased number of functional teeth, which was associated with the presence of MASLD. Furthermore, we identified several unique features of MASLD patients: 1) They experience oral problems even at an early stage of MASLD; 2) Subjective assessments of low oral function show low oral function even when objective assessments remain normal; 3) They worry about being noticed in public due to oral problems but do not hesitate to eat in public.
The finding that MASLD patients have poor OHRQoL is not surprising, as patients with diabetes also show low OHRQoL [17]. Diabetes patients have been reported to have poor oral hygiene, a high frequency of periodontitis, and fewer natural teeth [18]. One of the main reasons in diabetes patients is dry mouth [17], which was also observed in MASLD patients through longer salivary collection time compared to controls. In addition, 26% of MASLD patients in our study had type 2 diabetes. As expected, MASLD patients exhibited poor oral hygiene, worse periodontal disease, dental caries, and a decreased number of functional teeth. A high frequency of oral problems can reduce OHRQoL [19]. Thus, poor OHRQoL appears to be a common feature in patients with metabolic dysfunction.
The findings clearly demonstrated that poor OHRQoL occurs even at an early stage of MASLD. The mean FIB-4 index values were almost equivalent between control and MASLD groups. Additionally, the maximum values of FIB-4 index and M2BPGi were 1.9 and 1.24, respectively, indicating that our study included only early-stage MASLD patients. This finding contrasts with systemic HRQoL, where poor HRQoL is typically associated with more advanced liver disease. Cortesi et al. reported that patients with decompensated cirrhosis had poor systemic HRQoL, but not those with chronic hepatitis, including MASLD, compared to the general population [6]. Ascites and hepatic encephalopathy, representative complications in decompensated cirrhosis, can reduce systemic HRQoL. Although some studies demonstrated poor systemic HRQoL in patients with MASLD, this poor HRQoL disappeared after adjusting for confounding factors, including age, sex, and education [6]. Thus, it is necessary to take oral health into account when assessing HRQoL.
We also found that MASLD patients recognized oral problems before actual loss of oral functions. Among five questions related to oral functions, four showed significantly low scores compared to the control group. For instance, MASLD patients reported difficulty in chewing and speaking fluently. However, objective assessment did not always show impaired functions in the MASLD group. Indeed, the chewing ability of most of the MASLD patients was within the normal range. This dissociation between subjective and objective assessments may reflect the oral conditions and mental status. First, the number of functional teeth was lower in the MASLD group. Indeed, the number of functional teeth can cause low oral functions in GOHAI [20]. Second, patients with MASLD report anxiety in systemic HRQoL assessment [6]. Thus, MASLD patients may feel some oral problems before actual loss of oral functions. Although our study showed deteriorated periodontal disease in MASLD patients, periodontitis itself does not always affect oral functions [21].
In psychological assessments, MASLD patients worry about being noticed in public due to oral problems. Oral issues such as periodontitis can cause halitosis, potentially leading to reduced social interaction and exercise [22]. Patients with periodontal diseases have sedentary behavior [23]. These data suggest that oral problems may reduce opportunities to exercise in public. As a result, oral problems may interfere with motivation to maintain a healthy lifestyle. Additionally, MASLD patients did not report hesitation to eat in public, which may contribute to the development of metabolic syndrome-related diseases [17], including MASLD.
There is a bidirectional relationship between oral health and MASLD [24]. Interventions for MASLD, including exercise and dietary changes, can alter oral microbiota, potentially improving oral health [25, 26]. Conversely, oral care can ameliorate serum transaminase levels in MASLD patients [27], and treatment for periodontal disease has beneficial effects on OHRQoL [28]. Loss of oral function may lead to changes in eating habit, shifting from hard to soft foods. MASLD patients reported changes in both the volume and content of their food intake. Although we did not examine food intake in the present study, there is an association between oral condition and food intake [29]. For instance, people with a low number of teeth consume a low amount of fiber-rich food, such as vegetables [29]. As a result, serum levels of beta carotene, folate, and vitamin C were reported to be low in these patients [30, 31]. These nutritional components act as antioxidants and have protective roles against MASLD and periodontal diseases. Oxidative stress can promote cell death caused by ferroptosis, leading to advanced MASLD [32]. Thus, oral care is important in prevention of MASLD.
Our study has some limitations. First, the number of participants was relatively small due to several labor-intensive examinations, which were not performed in former studies [33, 34], including salivary collection time, chewing ability, and diadochokinesis assessment. As a result, our study did not have sufficient power to perform subgroup analysis of the associations between oral health and several conditions, including gastroesophageal reflux disease, non-erosive reflux disease, diabetes, medications, and the impact of oral problems among patients with MASLD. Of note, gastroesophageal reflux disease and diabetes may affect the oral health by changing gastrointestinal motility [35, 36]. Second, the diagnosis of hepatic steatosis in the control group was made by FLI, which does not completely rule out patients with steatosis. However, FLI shows high performance in diagnosis of steatotic liver disease, suggesting that the frequency of steatotic liver disease in the control group is not high. Third, there is a paucity of data on the association between oral health and MASLD, because the disease name and diagnostic criteria changed in 2023 [1]. Although cardiometabolic factors were required to diagnose MASLD, approximately 96-99% of NAFLD patients met the criteria of MASLD [37]. Hence, it is possible to use the data of NAFLD as a reference. Fourth, there is a lack of examinations for the relationship between oral health and oral microbiota. Oral microbiota may influence the composition of gut microbiota [38], which can also have an impact on MASLD. Further studies should address these limitations and further explore the complex relationships between oral health, microbiota, and MASLD.
In conclusion, MASLD patients exhibited low OHRQoL even at early stages of disease. They also showed poor oral hygiene, deteriorated periodontal conditions, and fewer functional teeth. A decreased number of functional teeth was independently associated with MASLD after adjusting for the effect of periodontitis. Furthermore, MASLD patients had unique characteristics in their eating behaviors. These findings provide new insights into the management of MASLD, particular concerning oral health, across all stages of the liver disease.