Introduction
Interleukin-23 (IL-23) blockade has emerged as one of the most promising novel therapeutic approaches in Crohn’s disease (CD). IL-23 is a pivotal inflammatory cytokine composed of a specific p19 subunit and a p40 subunit shared with IL-12, and it contributes to intestinal inflammation through downstream mediators such as interleukin-22 (IL-22) [1]. Increased mucosal expression of IL-23 has been consistently observed in patients with CD, while genome-wide association studies have linked polymorphisms within the IL-23 and IL-23 receptor (IL-23R) genes to disease susceptibility [2–4]. Variants in the IL-23R gene modulate serum IL-22 levels, which correlate with disease activity, suggesting that the IL-23–IL-22 axis plays a critical role in mucosal immune dysregulation [5–7]. Early clinical data have shown that blockade of IL-23 results in a reduction of IL-22 concentrations, with higher baseline IL-22 associated with better clinical response, supporting its potential as both a disease activity marker and a pharmacodynamic biomarker of IL-23 inhibition [8].
Risankizumab (RZB), a humanized monoclonal antibody selectively binding to the IL-23 p19 subunit, prevents its interaction with IL-23R, effectively disrupting the inflammatory cascade [9]. Initially approved for plaque psoriasis and psoriatic arthritis, RZB demonstrated significant efficacy and good tolerability in phase 2 trials in CD, particularly at the 600 mg intravenous induction dose [10]. However, the absence of a response plateau prompted further investigation in the phase 3 ADVANCE and MOTIVATE trials, which compared 600 mg and 1200 mg intravenous induction regimens in patients with moderate-to-severe CD refractory or intolerant to conventional and biologic therapies. Both studies confirmed significant clinical and endoscopic improvement with RZB, accompanied by a favorable safety profile, even among patients with prior biologic failures [9].
Against this background, a new clinical question emerges: could IL-23 blockade with RZB remain effective even as a fourth- or fifth-line therapy in patients who have failed multiple biologics, including antitumor necrosis factor therapy (anti-TNF), anti-integrin, and anti-IL-12/23 agents? As therapeutic sequencing in CD becomes increasingly complex, understanding the efficacy of RZB in this highly refractory population may redefine realistic treatment expectations. Exploring such cases may not only illuminate the potential of IL-23 inhibition in the most challenging scenarios but also rekindle hope for patients once considered beyond reach of remission. Importantly, in our group RZB was administered to patients with no remaining approved therapeutic options, who therefore received treatment through an emergency drug access.
Material and methods
Study design
This was a retrospective, observational multicenter case series conducted in four tertiary inflammatory bowel disease (IBD) referral centers in Poland.
Population
Adult patients (> 18 years old) starting RZB treatment for CD before 1st December 2025 were included. Patients with ulcerative colitis (UC) or IBD unclassified were excluded from the study. Participants were initiated on standard induction dosing of RZB (600 mg administered as an intravenous infusion at weeks 0, 4, and 8), followed by maintenance dosing of 360 mg administered subcutaneously at week 12 and thereafter every 8 weeks. RZB was used in patients for whom no other approved therapeutic options were available, and treatment was provided through an emergency drug access.
Data collection
Data on patients’ demographics, including age at diagnosis, age at RZB initiation, sex, body mass index (BMI), smoking status, extraintestinal manifestations (EIM), disease location and behavior according to the Montreal classification, previous and concomitant therapies, prior exposure to biologics or small molecules, and corticosteroid (CS) use at each time point, were recorded. Clinical disease activity was assessed using the Crohn’s Disease Activity Index (CDAI), which was routinely captured during visits at all participating centers. Relevant clinical biomarkers, including C-reactive protein (CRP), fecal calprotectin (FC), and hemoglobin (HGB), were collected. Additional clinical assessments included patient-reported improvement or remission of EIM and evaluation of perianal fistula activity. Details regarding RZB therapy, including start and stop dates, were documented. Patients’ data were retrieved up until the last standard RZB induction administration or the last follow-up visit, whichever occurred later. The reporting of this study adhFtableeres to the Harmonized Protocol for Real-World Evidence Reporting guideline [11].
Definitions of response and remission
Clinical remission and response were determined by the CDAI at 12 weeks of induction. Clinical response was defined as a decrease in CDAI of at least 70 points, and clinical remission was defined as a CDAI of less than 150. CS-free remission was defined as remission without CS use at that time point (irrespective of baseline CS status). CRP remission and FC remission were defined as a CRP level of less than 5.0 mg/l and a FC level of less than 150 µg/ml, respectively. HGB normalization was defined as an HGB level within the sex-specific normal range. Patient-reported improvement or remission of EIM and perianal fistula activity was recorded at each time point.
Outcomes
The primary endpoints were clinical remission, defined as a CDAI score of less than 150 points at the assessed visit and clinical response, defined as a reduction in CDAI of at least 70 points and at least 25% from baseline. Secondary endpoints included treatment safety, assessed as the incidence of all adverse events (AE) and serious adverse events (SAE); CS-free remission, defined as a CDAI score of less than 150 without CS use at that time point; reduction of CS dose, defined as a decrease of at least 50% from baseline; patient-reported improvement in EIM; and patient-reported improvement in perianal fistula symptoms.
Statistical analysis
Statistical software R (version 4.4.2) was used to prepare the analysis. For description of numeric variables, mean and standard deviation or median and interquartile range were used, depending on normality. Categorical variables were presented with counts and %/percentages of the respective group. Comparisons between follow-up measurement of CDAI and its baseline value were run with paired t test, due to normal distribution of differences. Distribution normality was assessed with the Shapiro-Wilk test, skewness and kurtosis values. 95% confidence intervals for proportions of primary and secondary endpoints were calculated using the Wilson method. All statistical calculations assumed a = 0.05.
Results
The group consisted of 57.1% of males and 42.9% of females with a mean age of 35.2 years and a median BMI of 21.5. Most patients were Caucasian (92.9%), and comorbidities occurred in 21.4%. The mean disease duration was 13.9 years. According to the Montreal classification, the group was predominantly A2 and L3, with 14.3% showing L4 involvement. Disease behavior was distributed across B1–B3, and perianal disease was present in 71.4%. A total of 78.6% of the group had undergone previous surgery for CD (Table I).
Table I
Demographic and clinical baseline characteristics of the study group (n = 14)
The group had a universal history of conventional therapy, with nearly all patients previously exposed to mesalazine (5-ASA), CS, and thiopurines, and most to methotrexate and budesonide. In contrast, current use of these agents was minimal, ranging from 0% to 7.1% (Table II).
Table II
Conventional treatment characteristics of the study group
| Treatment type | Past | Current |
|---|---|---|
| 5-ASA, n (%) | 14 (100.0) | 1 (7.1) |
| Budesonide, n (%) | 11 (78.6) | 0 (0.0) |
| CS, n (%) | 14 (100.0) | 1 (7.1) |
| AZA/MP, n (%) | 14 (100.0) | 0 (0.0) |
| MTX, n (%) | 13 (92.9) | 1 (7.1) |
The group showed universal exposure to all major advanced therapies, with nearly all patients receiving infliximab (IFX), adalimumab (ADA), vedolizumab (VDZ), and ustekinumab (UST), and 92.9% receiving upadacitinib (UPA). Dose escalation was frequently required, particularly for IFX (64.3%) and UST (57.1%). Primary non-response was uncommon across therapies, whereas secondary loss of response was the predominant reason for discontinuation, especially for ADA (85.7%) and UST (78.6%). Adverse events accounted for a smaller proportion of treatment terminations. Dual-targeted therapy (DTT) was used in 28.6% of the group, with secondary non-response being the main cause of cessation (Table III).
Table III
Advanced treatment of the study group
The group presented with high clinical activity, with a mean CDAI of 369 and markedly elevated FC levels. Inflammatory markers showed moderately increased CRP and mildly reduced HGB. EIM were present in 42.9% of patients, and colonoscopy was available in 42.9%, revealing mainly moderate-to-severe endoscopic activity. Half of the patients assessed had Rutgeerts i3–i4 lesions. Perianal fistulas were common (57.1%), with active disease in most affected individuals. CS were used by 42.9% of the group, typically at moderate doses (Table IV).
Table IV
Summary of the qualification visit
[i] CDAI – Crohn’s Disease Activity Index, CRP – C-reactive protein, EIM – extraintestinal manifestations, CS – corticosteroids, Hgb – hemoglobin, IQR – interquartile range, M – mean, Me – median, SD – standard deviation, SES – Simple Endoscopic Score for Crohn’s Disease. *Available for n = 4 patients. **Proportion calculated to all patients with perianal fistulas. ***Proportion calculated to all patients with CS.
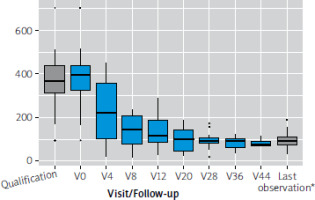
The group demonstrated a rapid and sustained reduction in CDAI from baseline across all follow-up visits, with mean decreases consistently exceeding 140–300 points. Improvements were statistically significant at every time point, and the last observation confirmed a robust overall clinical response (Table V). The plot in Figure 1 shows a clear and progressive decline in CDAI values across the treatment period. Already by Visit (V) 4, the median CDAI drops substantially, and subsequent visits demonstrate further stabilization at low disease activity levels, with narrowing interquartile ranges (IQR) indicating reduced variability within the group. Outliers become less prominent over time, suggesting that most patients achieved consistent improvement. The last observation confirms durable clinical benefit, with CDAI values remaining far below baseline levels.
Table V
Evolution of CDAI during treatment process in the study group
Figure 1
Boxplot presenting distribution of CDAI in the total study group through the qualification and treatment process (*last available measurement for each patient)
At week 12, more than half of the group achieved clinical remission and the vast majority demonstrated clinical response, with further improvement observed at the last follow-up. CS-free remission increased from 50% to over 70% during continued therapy, and all patients on CS reduced their dose. Improvements were also seen in extraintestinal manifestations and particularly in perianal fistulas, with rates rising from 62.5% to 87.5%. AE were infrequent and no severe events occurred (Table VI).
Table VI
Treatment efficacy in the study group (primary and secondary endpoints)
[i] CI – confidence interval, CS – corticosteroids. *Last available measurement for each patient. **CS dose reduction – proportion calculated to all patients with CS at V0. Improvement in extra-intestinal manifestations/EIM (extra-intestinal manifestations) – proportion calculated for all patients with EIM at V0. Improvement in perianal fistulas – proportion calculated for all patients with perianal fistulas at V0.
Discussion
In this multicenter real-world group of highly refractory CD, RZB demonstrated robust clinical effectiveness despite patients’ extensive prior treatment exposure, including multiple biologics, small molecules, and in some cases even DTT. These findings are particularly noteworthy given the advanced disease characteristics of the group: long-standing CD, high baseline CDAI, severe biochemical inflammation, a high prevalence of penetrating or stricturing behavior, frequent perianal disease, and a substantial history of prior surgical interventions. Such a population is typically underrepresented in clinical trials and is widely regarded as difficult to treat, often with limited expectations for meaningful disease control. Against this backdrop, the magnitude and consistency of RZB-induced improvement observed in our study underscore the therapeutic potential of selective IL-23 blockade as a viable option even at later lines of therapy.
The biological rationale supporting the use of RZB in refractory CD is firmly grounded in the central role of the IL-23 pathway in shaping chronic intestinal inflammation. IL-23, composed of the p19 and p40 subunits, drives downstream cytokines such as IL-22 and promotes the persistence of pathogenic T helper (Th) 17 cells lymphocytes responses [1]. Upregulation of IL-23 in mucosal tissue of CD patients has been repeatedly demonstrated, while genome-wide analyses have identified polymorphisms in the IL-23 and IL-23R genes as important contributors to disease susceptibility [2–4]. Notably, IL-23R variants influence circulating IL-22 levels, which correlate with clinical activity and may serve as mechanistic biomarkers linking genetic predisposition to the disease phenotype [5–7]. Importantly, previous studies have shown that IL-23 blockade reduces IL-22 concentrations and that higher baseline IL-22 predicts a stronger therapeutic response [8]. These observations reinforce the biological plausibility that patients with severe disease – often characterized by heightened IL-22 expression – may still benefit substantially from p19 inhibition even after failure of multiple treatment classes.
The clinical development program of RZB provides further context for interpreting our findings. In the phase 2 study, RZB showed encouraging efficacy and safety, particularly at the higher induction dose of 600 mg [10]. Subsequent large phase 3 induction trials, ADVANCE and MOTIVATE, confirmed significant improvements in clinical and endoscopic outcomes across both the 600 mg and 1200 mg intravenous dosing arms, including in patients who were refractory to prior biologic therapies [9]. Notably, in MOTIVATE – the trial exclusively enrolling patients with prior biologic failures – response rates remained substantial, supporting the concept that IL-23-driven inflammation persists even in heavily pre-treated individuals. Our results extend these observations to an even more challenging real-world population: patients not only refractory to biologics but who in many cases had failed several therapeutic classes, including anti-TNF, anti-integrin, and anti-IL-12/23 agents, alongside small molecules such as UPA. A significant portion of the group had also previously received DTT, indicating profound disease refractoriness. Despite this, RZB induced rapid improvements in clinical activity by week 4, with sustained and deepening responses through later follow-up.
By week 12, more than half of patients achieved clinical remission and almost 86% achieved clinical response – figures comparable to, and in some cases exceeding, those reported in earlier clinical trials, despite our population’s more complex baseline profile. At the last observation, remission rates approached 80%, highlighting the durability of RZB effectiveness. Additionally, CS-free remission increased markedly over time, and all patients receiving CS were able to taper their dose, confirming the capacity of RZB to reduce steroid dependency – a key therapeutic goal in modern CD management. Improvement in EIM was observed in half of affected patients, and nearly 90% of those with perianal fistulas reported symptomatic improvement by the final follow-up. While endoscopic evaluations were available only for a subset, the predominance of moderate-to-severe baseline lesions and the parallel clinical improvement suggest meaningful endoscopic healing trends, consistent with the mucosal benefits documented in controlled trials [9, 10].
The consistency of CDAI reduction across follow-up visits – routinely exceeding 200–300 points – further underscores the depth of response. The progressive narrowing of interquartile ranges and reduction of outliers reflect an increasingly homogeneous treatment effect, even in patients with previously unstable disease trajectories. Taken together, these observations highlight that selective IL-23 blockade remains effective despite prior immunologic exposures that likely altered or exhausted other inflammatory pathways. This reinforces the concept that IL-23-driven inflammation may represent a persistent and therapeutically exploitable mechanism even after failure of upstream cytokine blockade such as UST (anti-p40), which targets both IL-12 and IL-23 but does not selectively inhibit the p19 subunit. The contrast between p40 and p19 inhibition may be clinically meaningful, consistent with emerging immunological data suggesting that more focused suppression of IL-23 may better disrupt sustained Th17-mediated inflammation [1, 3].
Importantly, the safety profile in our group aligns with previous RZB studies. AE were infrequent, mild, and did not necessitate treatment discontinuation, while no severe adverse events occurred. This is particularly relevant for patients with long treatment histories, where cumulative immunosuppression raises concerns regarding tolerability. The favorable safety outcomes observed here support the use of RZB even in extensively pre-treated individuals.
The findings of this study, while compelling, must be interpreted within the limitations inherent to a retrospective design. Heterogeneity in follow-up duration, incomplete endoscopic assessments, and the relatively small sample size may constrain generalizability. Nonetheless, the real-world nature of the study offers valuable insights into treatment outcomes in populations often excluded from randomized trials.
Conclusions
This study demonstrates that RZB provides substantial and durable clinical benefits in a real-world group of highly refractory CD patients, including patients who had failed multiple biologic classes and dual-targeted regimens. These results extend the evidence base supporting IL-23 p19 inhibition and suggest that RZB represents a promising therapeutic option even at advanced stages of treatment sequencing. As therapeutic pathways continue to expand, IL-23 blockade may emerge as a cornerstone strategy capable of restoring disease control in patients previously considered beyond reach of remission.













