Introduction
Oropharyngeal carcinoma (OPC) is among the fastest-growing head and neck cancers, with approximately 100 000 new cases annually in high- income countries, largely driven by human papillomavirus (HPV) infection [1]. Despite recent advances in the management of OPC, chemoradiotherapy (CRT) remains the cornerstone of non-surgical management in patients with locally advanced disease [2]. Although numerous prognostic and predictive factors have been investigated, only HPV testing, most commonly performed using p16 immunohistochemistry (IHC), has been effectively integrated into clinical practice, as most other biomarkers remain limited by cost, complexity, timing, or insufficient reliability [3].
The role of amino acids in cancer metabolism remains an active area of research. Several studies have explored amino acid profiling as a potential prognostic or predictive tool in different cancer types [4–7]. Citrulline, an amino acid involved in arginine metabolism, has been investigated as a biomarker of intestinal injury and mucositis [8]. Intestinal mucositis, though less routinely assessed than oral mucositis, can contribute to chemotherapy interruptions and worse overall survival (OS) [9]. Importantly, a strong association between oral and intestinal mucositis has been repor- ted [10]. Moreover, citrulline was also described as an important factor influencing tumour proliferation and progression, as well as interaction with tumour immune microenvironment [11, 12].
Recent studies highlight plasma citrulline as a marker of enterocyte damage during chemotherapy. Low citrulline levels have also been linked to impaired enterocyte function in conditions such as Crohn’s disease and short bowel syndrome, where they may contribute to hypoalbuminemia, malnutrition, and subsequently reduced OS [13, 14].
In this study, we reanalysed a previously published dataset originally designed to investigate the role of microRNAs in oropharyngeal cancer patients. Our focus here was to explore citrulline predictive value for progression-free survival (PFS) and OS in this patient population.
Material and methods
Study group
Our study group consisted of patients included in the study evaluating predictive markers for adverse events in the course of OPC treatment. The primary cohort consisted of patients treated in three oncology centres in Poland: Copernicus Memorial Multidisciplinary Centre for Oncology and Traumatology in Lodz, Radom Oncology Centre, and Holy Cross Cancer Centre in Kielce. Given that concurrent CRT remains the standard of care for most patients with locally advanced, non-surgically managed OPC, we restricted our analysis to this treatment group to ensure clinical and therapeutic homogeneity. As citrulline levels typically rise in response to enterocyte damage and oral mucositis, both commonly induced by chemotherapy, patients receiving radiotherapy alone were not included in our study. The selection process is summarized in Figure 1.
Figure 1
Comparison of the citrulline level between clinical variables
HN – head and neck, RT – radiotherapy The primary cohort was established to evaluate predictive markers of adverse events during the treatment of oropharyngeal cancer. Patients were recruited from three major oncological centres in Poland. Initially, 152 patients were screened for inclusion in the study protocol. Of these, 87 met the preliminary eligibility criteria, but only 64 were ultimately included in the final analysis. Sixty-five patients were excluded during the preliminary screening phase. Some patients met more than one exclusion criterion.
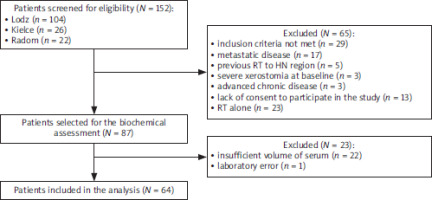
The staging was performed according to the American Joint Committee on Cancer 7th edition staging system [15]. All patients enrolled in the study underwent qualification to radiotherapy according to the centre protocol, which is based on the standard treatment according to the National Comprehensive Cancer Network. All patients were treated with intensity-modulated radiation therapy (bilateral irradiation in all patients) using conventional fractionation – with a planned total dose of 69.96–70.0 Gy in 33–35 fractions, five fractions/week (daily Monday–Friday) [16] with concomitant systemic treatment, including either weekly platinum-based hemotherapy (cisplatin 40 mg/m2) or every three weeks platinum (cisplatin 100 mg/m2)). Induction chemotherapy was allowed if followed by concurrent CRT, using either the PF regimen (cisplatin 100 mg/m2 on day one with 5-FU 1000 mg/m2 administered by continuous infusion on days 1–4, every 21 days) or the TPF regimen (docetaxel 75 mg/m2, cisplatin 75 mg/m2 on day 1 with 5-FU 1000 mg/m2 administered by continuous infusion on days 1–4, every 21 days).
Serum collection
Venous blood samples were collected from OPC patients prior to treatment start. Peripheral blood was collected in serum separating tubes and processed within two hours of collection by centrifugation at 2400 × g for 10 min. Serum samples were stored at –80°C for further use.
HPV16 status assessment
For p16 IHC, a CINtec p16INK4a histology kit (DakoCytomation BV, Heverlee, Belgium) was used with a 70% nuclear and cytoplasmic staining cutoff [17]. Both positive and negative control specimens were included in every immunostaining run.
Serum citrulline assessment
Serum citrulline levels were measured using a double- sandwich enzyme-linked immunosorbent assay (ELISA) system (Qayee-Bio, China). 96-well assay plates were commercially coated with citrulline antibody. Standard citrulline (6.25–100 nmol/ml) was added to individual wells. The serum samples were diluted 1 : 5 in special diluent and added to the wells (50 µl/well) and 50 µl of horseradish peroxidase into each well, except blank well. Then the plate incubated for one hour at 37°C, followed by five washes. Thereafter, 50 µl of chromogen solution A and then 50 µl of chromogen solution B was added to each well and incubated for 10 min at 37°C in the dark. The reaction was stopped with a stop solution (50 µl/well), and the blue colour changed into yellow immediately. The optical density (OD) was measured at 450 nm with wavelength correction at 620 nm using an ELISA reader (Devyser, USA). Measurement was carried out within 15 minutes after adding the stop solution. All serum samples were tested in duplicate, and the results were averaged. Unknown concentrations were extrapolated from the standard curve using GraphPad Prism 9 (GraphPad Software, San Diego, CA, USA) based on the OD values of the samples and the corresponding standard concentrations.
Statistical analysis
Nominal variables were expressed as percentages and analysed using the χ2 test with appropriate corrections if needed: the Yates correction for continuity or Fisher’s exact test. The normality of the distribution of continuous variables was verified with the Shapiro-Wilk test. Continuous variables were characterized using mean and standard deviation. The comparisons between two independent groups were performed using t-test. The association between the citrulline level and OS as well as PFS was assessed using Cox proportional hazard regression. All statistical analyses were conducted using R programming language (v. 4.5.1).
Results
Group characteristics
In total, 64 OPC patients treated with CRT were included in the study, comprising 15 (23.4%) women and 49 (76.6%) men with mean age of 69.9 ±8.5 years. The predominant localization of OPC was palatine tonsil (n = 42, 65.2%), followed by the base of the tongue (n = 9, 15.6%) and posterior pharyngeal wall (n = 7, 10.9%). Most patients presen- ted with T2 disease (n = 38, 58.4%) and nodal involvement (n > 0, n = 55, 85.9%), with stage IV predominating (n = 47, 73.4%). Human papillomavirus positivity was confirmed in 29 patients (45.3%). Detailed group characteristics are presented in Table 1.
Table 1
The characteristics of oropharyngeal carcinoma patients
The mean baseline level of serum citrulline was 42.7 ±10.1 nmol/ml, with no significant associations observed between citrulline levels and clinical variables such as sex, tumour advancement, or treatment regimen (Figure 2).
Survival analysis
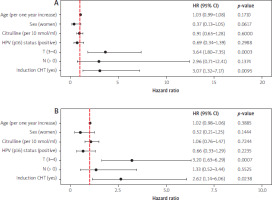
In univariable Cox regression, both the larger tumour size (HR 3.64, 95% CI: 1.80–7.35, p = 0.0003), and induction chemotherapy (HR 3.07, 95% CI: 1.32–7.17, p = 0.0095) were significantly associated with poorer OS. For PFS, T stage (HR 3.20, 95% CI: 1.63–6.29, p = 0.0007), and induction chemotherapy (HR 2.62, 95% CI: 1.14–6.06, p = 0.0238) also predicted worse outcomes. The baseline citrulline level was not associated with OS or PFS in the overall cohort (Figure 3).
Figure 3
Summary of univariate models for overall survival and progression-free survival
CHT – chemotherapy, HPV – human papillomavirus, HR – hazard ratio, OS – overall-survival, PFS – progression-free survival Panel A presents the univariate Cox models for overall survival, and Panel B presents the univariate Cox models for progression-free survival.
In the subgroup of HPV-positive patients, however, both the larger tumour size (HR: 12.21 95% CI: 3.17–47.05, p < 0.0001) and higher baseline citrulline level (HR: 1.07, 95% CI: 1.00–1.15, p = 0.0341) were significantly associated with poorer OS. Similarly, both the larger tumour size (HR: 19.05 95% CI: 3.80–95.42, p < 0.0001) and higher baseline citrulline level (HR: 1.11 95% CI: 1.03–1.19, p = 0.0040) were associated with shorter PFS (Table 2). In contrast, no significant relationship was observed in the HPV-negative group, although a trend toward significance was noted for citrulline and OS (HR: 0.95 95% CI: 0.90–1.00, p = 0.0631) (Table 2).
Table 2
Summary of multivariable Cox for overall survival and progression-free survival in subgroup analysis based on human papillomavirus (p16) status
Discussion
In our study, we evaluated the value of the pretreatment citrulline level in prediction of treatment outcomes in OPC patients. Even though the citrulline level was not associated with OS and PFS in the whole group, the subgroup analysis revealed that the citrulline level was significantly associated with OS and PFS in HPV-positive patients. Nonetheless, the exact relationship between the citrulline level, HPV infection and CRT treatment is not fully elucidated.
Oral mucositis is a well-established adverse effect of both chemotherapy and radiotherapy, particularly when used concurrently [10]. In contrast, injury to the small- intestinal epithelium is less frequently assessed clinically, but remains a recognized complication of cytotoxic therapy [9]. Citrulline, synthesized primarily in the small intestine as part of the arginine metabolism pathway, decreases in response to intestinal injury [18]. Indeed, Barzał et al. [8] described citrulline as a potential biomarker of chemotherapy-induced enterocyte damage. The nasopharyngeal and oropharyngeal epithelium does not produce citrulline in significant amounts, aside from minor local production as a by-product of nitric oxide synthase activity or as an effect of oral bacteria production [19, 20].
It is worth considering why the prognostic association of citrulline appeared to be restricted to the HPV-positive subgroup. HPV-positive OPCs are characterized by a highly immunogenic tumour microenvironment, enriched in CD8+ and CD4+ T-cells, macrophages, and other immune effector populations, which contribute to their generally better prognosis and sensitivity to immunomodulatory therapies [21, 22]. Serologic responses to the viral HPV16 oncoproteins E6 and E7 are also associated with enhanced antitumor immune activation and improved clinical outcomes that might be suppressed by elevated levels of Il-23 secreted by M2 macrophages [23, 24]. Last, but not least, HPV- related OPC have usually enriched glycolysis and OXPHOS pathway with NRF2 driving tumour growth, ROS and mitochondrial respiration compared to HPV-negative ones [12, 25]. In vitro and in vivo (mice) it was found that citrulline stimulates the proliferation, migration, invasion, epithelial-mesenchymal transition of lung cancer cells [12]. Therefore, the increased citrulline level might just simply fuel tumour growth and metabolism directly as a nutrient source. Moreover, knowing that citrulline might activate the Il-6/STAT pathway, it can block immune response and reduce prognostically beneficial infiltration [12, 26–28]. The same type of interaction might exist between citrulline and Il-23 and subsequently block serologic response (however, it is not confirmed yet and needs to be investigated) to HPV16 oncoproteins. Finally, the citrulline cycle is employed by CD8+ T-cells to remove toxic ammonia. Cytosolic arginine is converted to nitric oxide and citrulline. Therefore, elevated citrulline levels may serve as a biomarker of arginine depletion and ammonia stress, both of which are known to impair the T-cell response to tumors [11].
Limitations
Several limitations should be acknowledged. First, although the prospective design and standardized sample collection strengthen the reliability of our findings, the cohort size was relatively modest. Second, the original study was designed to investigate serum microRNAs as biomarkers of xerostomia in patients with oropharyngeal cancer undergoing radiotherapy. The current analysis of citrulline in relation to oncologic outcomes was therefore post hoc, and the study was not powered or specifically sized to detect prognostic effects on cancer control or survival. Finally, the lack of an independent external validation cohort limits the generalizability of our findings.









