Introduction
Infectious mononucleosis (IM) is a common disease in children and adolescents that most often results from acute Epstein-Barr virus (EBV) infection. In early childhood, it is usually asymptomatic or mildly symptomatic, while 30-50% of teenagers and young adults develop a broad variety of clinical symptoms of IM [1]. The classic triad of symptoms, including fever, sore throat, and generalized lymphadenopathy, is often accompanied by fatigue, mild headache, and hepatosplenomegaly.
The available data from the literature show that the percentage of children with hepatitis (usually asymptomatically elevated transaminases) in the course of EBV infection ranges from 50% (Polish study) to 80-90% (studies conducted in England and Greece) [2–4]. These data concern patients hospitalized due to IM. The activity of liver transaminases in serum most often does not exceed two or three times the upper limit of normal [5]. Although cholestatic hepatitis is rare and usually reported in adults, it can also develop in children; in a Polish study, it occurred in 10.8% of pediatric patients with hepatitis due to primary EBV infection [2, 6].
In approximately 10% of patients with infectious mononucleosis-like syndrome, the etiological factor is not EBV and may include cytomegalovirus (CMV), primary hepatotropic viruses, human immunodeficiency virus (HIV) – acute retroviral disease, or adenovirus [7]. Primary EBV and CMV infections in immunocompetent individuals cause similar clinical and laboratory symptoms. Studies report a more severe course of the disease in patients with dual-positivity for EBV and CMV than in those with positive only anti-VCA IgM for EBV [8]. Moreover, according to some authors, CMV infection may be associated with more severe hepatitis [9, 10].
Acute EBV and CMV coinfection confirmed by positive serological test results (immunoglobulin M [IgM]) is considered by many researchers and clinicians to be a false-positive finding due to the cross-reaction of serum antibodies [11–14].
From April 2023 to May 2024, we observed an increase in the number of patients hospitalized in our department with hepatitis in the course of IM and positive serological test results for EBV and CMV compared to the previous years.
The present study aimed to compare the course (in terms of clinical symptoms and laboratory and imaging test results) of hepatitis in pediatric patients with positive anti-VCA IgM for EBV and in those with dual serological positivity for EBV and CMV, and to identify the differences between the cohorts.
Material and methods
Patients
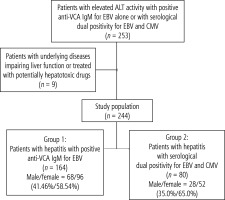
Our retrospective, single-center study was based on the analysis of the clinical data of patients aged 0-18 years who were hospitalized for acute hepatitis due to EBV infection (with or without positive IgM for CMV) at the Department of Children’s Infectious Diseases, Medical University of Warsaw, Regional Hospital of Infectious Diseases in Warsaw, between November 2017 and May 2024.
We established the following exclusion criteria:
coexisting viral hepatitis (hepatitis A virus [HAV], hepatitis B virus [HBV], hepatitis C virus [HCV]),
preexisting human immunodeficiency virus (HIV) infection,
other liver diseases (e.g., metabolic or autoimmune) associated with impaired liver function,
hepatotoxic drug therapy due to underlying chronic diseases and other causes of toxic liver damage,
chronic diseases affecting the host’s immune response (e.g., immunodeficiencies, severe hemoglobinopathies, cancer).
Methods
We considered the onset of the disease to be the first day of symptoms typical for infectious mononucleosis syndrome (fever, lymphadenopathy, sore throat). Some patients experienced isolated gastrointestinal symptoms, jaundice, hepatosplenomegaly, or increased liver aminotransferase activity. We confirmed EBV infection using a serological test detecting IgM antibodies to the EBV viral capsid antigen (anti-EBV VCA IgM, chemiluminescence immunoassay – CLIA test system, LIAISON EBV IgM, DiaSorin S.p.A., Italy) as a marker which does not occur in chronic disease. Similarly, the diagnosis of acute CMV infection was based on the detection of immunoglobulin M antibodies to CMV in serum or plasma samples (chemiluminescence immunoassay – CLIA test system, LIAISON CMV IgM, DiaSorin S.p.A., Italy). Both tests are routinely performed in patients suspected of having infectious mononucleosis. Depending on clinical indications, tests for other hepatotropic viruses were ordered.
The laboratory indicator of ongoing hepatitis was increased over the upper limit of normal (concerning age and sex references) activity of serum alanine aminotransferase (ALT) [15]. Additionally, we assessed the activity of the serum aspartate aminotransferase (AST), γ-glutamyl transferase (γ-GT), and bilirubin concentrations in the fractions. AST and γ-GT values were related to age- and sex-specific norms [15]. Other laboratory tests included a full blood count with differential and C-reactive protein (CRP) levels. We defined leukocytosis based on age and sex standards [16]. Thrombocytopenia was defined as a platelet count of less than 100,000 per mm3.
During the abdominal ultrasound examination (which was performed in the vast majority of patients), we paid special attention to the size of the liver, spleen, and lymph nodes in the liver hilum, as well as to the pathology of the gallbladder and bile ducts. Splenomegaly and hepatomegaly on ultrasound examination were diagnosed when the longitudinal dimension of these organs exceeded the 97.5th percentile for age [17].
In both study groups, we identified patients with a more severe course of the disease who met at least one of the following criteria:
at least three of the following signs or symptoms: nausea/vomiting, darkening of urine (the color of urine darker than in a healthy state), discoloration of stool, jaundice, pruritus, abdominal pain or
at least one laboratory abnormality of the following: ALT activity equal to or greater than five times the upper limit of normal for sex and age, increased γ-GT activity in serum, direct bilirubin concentration equal to or greater than 20% of total serum bilirubin concentration or
signs of inflammation in the gallbladder/biliary tract on abdominal ultrasound.
Statistical analysis
We performed a statistical analysis of the collected medical records using STATISTICA 13.3 (StatSoft, Kraków, Poland). The data are presented as the means (standard deviations; SDs) unless otherwise indicated. The comparison of clinical features between study cohorts was performed using Fisher’s exact test or Pearson’s chi-square test (for categorical variables) and the Mann-Whitney U test or Kruskal-Wallis test for continuous variables, such as laboratory findings, the duration of signs and symptoms, and the length of hospitalization. None of the studied groups showed a normal distribution concerning the analyzed variables. The level of statistical significance was set at p < 0.05.
Results
Demographic and clinical characteristics
We performed a retrospective analysis of the medical records of 253 pediatric patients who were hospitalized due to hepatitis with increased ALT activity in the course of EBV infection with or without positive IgM for CMV. Nine patients (3.55%) were excluded from the study because of underlying diseases associated with impaired liver function or potentially hepatotoxic drug therapy. For the study, we recruited 244 children and adolescents aged 0-18 years and divided them into two groups. The first cohort consisted of 164 patients (67.21%) with hepatitis during EBV infection (the male-to-female ratio was 1 : 1.41). In the second group of 80 (32.79%) children with increased ALT activity due to EBV infection with positive IgM for CMV, the male-to-female ratio was 1 : 1.86. The enrollment of patients in the study is presented in the flowchart (Fig. 1).
The age distributions in both cohorts were similar: 12.02 ±5.00 years in the first group and 11.92 ±5.33 years in the second group (p = 0.851), with a predominance of children aged 3-8 and 13-18 years as typical epidemiological features of IM.
Children in the second group more often suffered from chronic diseases (29; 35.80%) than did patients in the first group (42; 25.61%), although this difference was not statistically significant (p = 0.085). The most common were allergic diseases (bronchial asthma, inhalant or food allergy, atopic dermatitis), neuropsychiatric disorders (autism, depressive disorders, recurrent headaches), and adenoid hypertrophy. The incidence of particular disease entities did not differ significantly between the groups. The drugs used to treat the above conditions did not affect liver function.
Clinical course and complications
The analysis of the course of the disease did not reveal any significant differences between the study groups in terms of the duration of signs and symptoms or hospitalization. These data are presented in Table 1.
Table 1
Differences in demographic and clinical features between the study groups
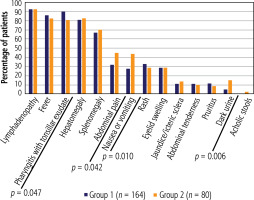
A detailed analysis of the prevalence of particular symptoms and signs of IM revealed significant differences concerning the following:
pharyngitis with tonsillar exudate was more common among patients with isolated EBV infection, 148 (90.24%) vs. 65 (80.25%) in patients with EBV and CMV dual positivity; p = 0.047;
nausea or vomiting occurred more often in the second study group: 35 (43.21%) vs. 45 (27.44%) in the first group; p = 0.010 (OR = 2.01 [95% CI: 1.15-3.51]);
abdominal pain was reported more frequently in children in Group 2 (37 (45.67%) than in those in Group 1 (52 (31.71%)); p = 0.042 (OR = 1.81 [95% CI: 1.05-3.13]);
dark urine was observed in 12 (14.81%) individuals with EBV and CMV serological dual positivity and in 8 (4.88%) with EBV monoinfection; p = 0.006 (OR = 3.39 [95% CI: 1.33-8.67]).
The frequency of other symptoms of mononucleosis syndrome did not differ significantly between the study groups. Figure 2 shows the frequency of IM symptoms in both cohorts.
Complications (except those related to the liver and biliary tract) were diagnosed in 24 (14.63%) patients in the first group and 13 (16.25%) in the second group (without any significant difference). The most frequently observed sequelae were as follows (number and percentage of patients in Groups 1 and 2, respectively): upper respiratory tract obturation: 68 (41.46%) vs. 25 (31.25%); rash after amoxicillin administration: 27 (16.46%) vs. 8 (10%); thrombocytopenia (with or without purpura): 4 (2.44%) vs. 3 (3.75%); and urticaria: 2 (1.22%) only in the first group.
The frequency of infectious complications (or cooccurring infections) was significantly higher in children in the first group: 80 (48.78%) than in those in the second group: 28 (34.57%, p = 0.036). The most frequently diagnosed infections were as follows (in Groups 1 and 2): bacterial pharyngitis (usually caused by Streptococcus pyogenes): 54 (32.93%) vs. 16 (20.00%); acute otitis media: 4 (2.44%) vs. 3 (3.75%); oral candidiasis (“thrush stomatitis”): 3 (1.83%) in the first group; and bacterial pneumonia: 2 (1.22%) vs. 1 (1.25%). However, only scarlet fever was diagnosed significantly more often in the first Group: 2 (1.22%) vs. 2 (2.50%), p = 0.027.
Laboratory and imaging test results
When we analyzed the values of the inflammation parameters, we detected significant differences in the CRP serum concentration – 21.93 ±16.54 mg/dl in Group 1 vs. 15.15 ±12.85 mg/dl in Group 2 (p = 0.001) – and the percentage of neutrophils in the manual white blood cell smear: 32.07 ±13.25% in the first group vs. 25.33 ±12.35% in the second group (p = 0.000). Leukocytosis occurred at a comparable frequency in both study groups: 119 (72.56%) patients in the first group and 59 (74.68%) children in the second group. Lactate dehydrogenase (LDH) level was higher, though not significantly, in children with EBV and CMV dual positivity (p = 0.089). Table 2 presents the comparison of laboratory test results between the study groups.
Table 2
Comparison of laboratory test results between study groups
| Variable | Group 1 (n = 164) | Group 2 (n = 80) | p-value |
|---|---|---|---|
| Mean ± SD (range) | |||
| White blood cell count (cells/µl)a | 14.41 ±6.40 (4.47-49.00) | 15.03 ±5.58 (5.10-33.90) | 0.136 |
| Neutrophils (%)a | 32.07 ±13.25 (5.00-77.00) | 25.33 ±12.35 (2.00-57.00) | 0.000 |
| Atypical lymphocytes (%)a | 25.71 ±14.56 (0.00-70.00) | 28.27 ±15.18 (6.00-80.00) | 0.276 |
| CRP (mg/dl)b | 21.93 ±16.54 (0.41-75.00) | 15.15 ±12.85 (5.00-58.00) | 0.001 |
| PCT (ng/ml)c | 1.98 ±7.04 (0.05-37.00) | 0.60 ±0.72 (0.08-3.44) | 0.787 |
| Platelets (cells/µl)d | 209.97 ±67.17 (9.80-4.55) | 205.87 ±74.49 (9.7-3.66) | 0.658 |
| LDH (U/l)e | 686.37 ±238.60 (413.00-1427.00) | 842.67 ±283.14 (400-1350) | 0.089 |
The serum activity of ALT in the successive weeks of the disease in both study groups is presented in Figure 3. The analysis conducted over time revealed no significant differences between the cohorts. In patients positive for EBV and CMV, the values were higher throughout the disease than in children with isolated EBV infection, but without reaching statistical significance (in the first week, p = 0.083). The values discussed are presented in Table 3.
Fig. 3
Mean activity (±SD) of alanine-aminotransferase (ALT) in successive weeks of hepatitis in Group 1, p = 0.309, and Group 2, p = 0.864. The Kruskal-Wallis test was used for changes over time
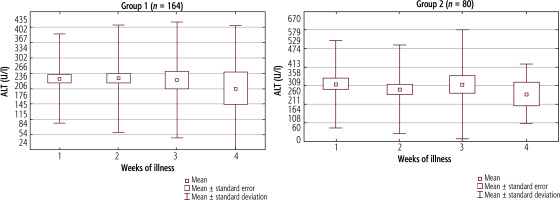
Table 3
Values of alanine-aminotransferase (ALT) activity in serum of patients in Groups 1 and 2 during successive weeks of the disease
Aspartate aminotransferase activity in the serum did not significantly differ between successive weeks of disease in either study group (Fig. 4). In the first group, AST values were highest in the second week of illness, with a decreasing trend in the third and fourth weeks. In the second group, the highest AST levels were observed during the first and fourth weeks. However, AST activity was significantly higher in the second group only in the first week (p = 0.023). The values of AST in the serum in successive weeks of illness in both study groups are presented in Table 4.
Fig. 4
Mean activity (±SD) of aspartate-aminotransferase (AST) in successive weeks of hepatitis in Group 1, p = 0.107, and Group 2, p = 0.116. The Kruskal-Wallis test was used for changes over time
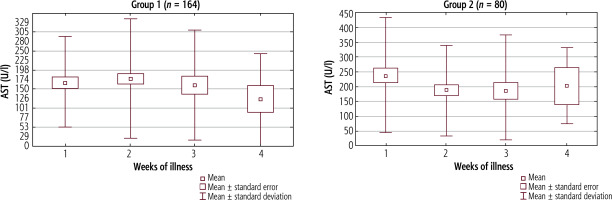
Table 4
Aspartate-aminotransferase (AST) activity in serum of patients in Groups 1 and 2 during successive weeks of disease
Analysis of γ-GT revealed the highest activity in the first group in the second week of the disease and in the second group in the first week (Fig. 5). In both cohorts, the differences between the successive weeks were not statistically significant (although in Group 1 the p-value reached 0.082). In the second week the activity of this enzyme showed a non-significant difference between groups (p = 0.091). The values of γ-GT activity in the serum in successive weeks of illness in both study groups are presented in Table 5.
Fig. 5
Mean activity (±SD) of γ-glutamyl transferase (γ-GT) in successive weeks of hepatitis in Group 1, p = 0.082, and Group 2, p = 0.308. The Kruskal-Wallis test was used for changes over time
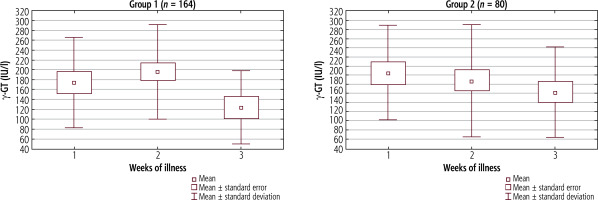
Table 5
Values of γ-glutamyl transferase (γ-GT) activity in serum of patients in Groups 1 and 2 during successive weeks of disease
The concentration of total bilirubin in the serum in both groups was the highest in the first week of the disease and was similar, as shown in Figure 6. The greatest difference was observed in the second week. The above data are presented in Table 6. The percentage of children whose direct bilirubin concentration exceeded 20% of total serum bilirubin in the first group was 80.95%, and in the second group it was 78.26% (this difference was not significant).
Fig. 6
Mean total bilirubin serum concentration (±SD) in successive weeks of hepatitis in Group 1, p = 0.698, and Group 2, p = 0.769. The Kruskal-Wallis test was used for changes over time
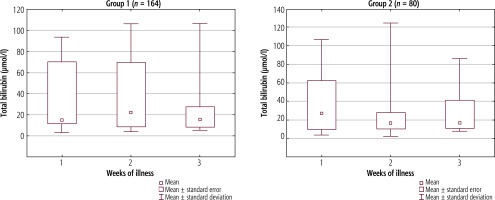
Table 6
Serum total bilirubin concentrations in Groups 1 and 2 during successive weeks of the disease
For all patients in both groups, the Quick index values were within normal limits.
All the children underwent abdominal ultrasound examination, with significantly more abnormal results (100% of patients) in the second group than in the first group (66.46%), p = 0.045. However, the frequency of particular abnormalities on abdominal ultrasound did not differ significantly between the study groups, although they occurred more often in children with EBV and CMV dual positivity: hepatomegaly in 71 (43.29%) patients in the first group vs. 41 (51.25%) in the second group (p = 0.532); enlargement of the lymph nodes in the liver hilum in 63 (38.41%) in Group 1 and 40 (50.00%) in Group 2 (p = 0.921); and abnormalities of the gallbladder and bile ducts in 18 (10.97%) children in the first group vs. 16 (20.00%) in the second group (p = 0.297). Abnormalities of the gallbladder and biliary tract included thickening of the gallbladder wall, dilatation of the bile ducts, and accumulation of fluid in the hilum of the liver and around the gallbladder.
Holistic assessment of the course of hepatitis
Our criteria for a more severe course of the disease were met by 96 (58.54%) children from the first group and by 60 (74.07%) patients from the second group, p = 0.018 (OR = 2.02 [95% CI: 1.13-3.64]).
Treatment
All study patients received supportive therapy for fever, dehydration, respiratory tract obturation, or severe sore throat.
Empirical antibiotic therapy (most often due to pharyngitis) had been implemented before hospitalization (and final diagnosis) in 90 (54.87%) patients from the first group and 34 (41.98%) from the second group (p = 0.116). Amoxicillin was most frequently prescribed: 41 (25.00%) patients with EBV monoinfection vs. 15 (18.52%) children with EBV and CMV dual-positivity, p = 0.275. Twenty-seven patients (16.46%) from the first group and eight (9.87%) from the second group developed an allergic-toxic rash after amoxicillin (p = 0.176). Cefuroxime axetil was the second most commonly used antibiotic.
After admission to the hospital (and IM diagnosis), 83 (50.61%) children from the first group required antibiotic therapy compared to only 27 (33.33%) patients from the second group (p = 0.011). The most common indication was pharyngitis. Cefuroxime was the most frequently used agent: in 61 (73.49%) children from the first group and 18 (66.66%) from the second group. Five (6.02%) children with EBV monoinfection and three (1.11%) with EBV and CMV dual positivity required therapy with two antibiotics. The second antimicrobial agent most frequently used was clindamycin (due to inflammation of the cervical lymph nodes).
All patients fully recovered without any chronic complications.
Discussion
Our retrospective study aimed to compare the clinical course of hepatitis between pediatric patients with EBV monoinfection (Group 1) and those with EBV and CMV dual positivity (Group 2). The percentage of children belonging to the second group was 32.79% of all patients, which was comparable to that reported in other studies (20-42%) [1, 18, 19].
This study covers the period from November 2017 to May 2024. In our previous study, we described the course of hepatitis caused by primary EBV infection in children hospitalized from August 2017 to March 2023 [2]. The number of patients with hepatitis due to EBV mono-infection is comparable in both studies. From April 2023 to May 2024, the majority of children with hepatitis associated with IM syndrome had positive serological test results for EBV and CMV.
The studies conducted thus far among patients with IM and hepatic involvement usually showed that positive IgM for CMV was the effect of the antigenic cross-reactivity related to the EBV infection [1, 12, 13, 20]. Most of these studies were conducted in adult patients. The phenomenon of positive antibodies against both viruses can be explained by the polyclonal production of antibodies against other viruses from the Herpesviridae family (e.g., CMV, varicella-zoster virus, human herpes virus 6) caused by acute EBV infection [21]. According to the second theory, EBV infection causes transient immunosuppression, which favors the reactivation of latent CMV infection [20].
Some authors have suggested that liver involvement in EBV infection may be the cause of double positivity for EBV and CMV by stimulating the production of antibodies [1].
In our study, the age distribution in both groups was similar, which is inconsistent with the observations of other authors, indicating more frequent coexistence of positive results for EBV and CMV among younger children [1, 18].
Complaints and symptoms that may indicate the development of cholestasis were significantly more frequently reported among children with positive serological test results for both viruses (Group 2). Nausea, vomiting, and abdominal pain occurred approximately twice as often as they did in the first group, and darkening of urine occurred more than three times as often.
Pharyngitis and tonsillitis (usually due to Streptococcus pyogenes) were observed significantly more often among patients with positive serological testing only for EBV. Other concomitant infections were also diagnosed more frequently in the first group, although this difference was not statistically significant.
The above observations were reflected in a significantly higher CRP concentration in the serum and the
percentage of neutrophils in the manual smear of white blood cells.
The difference in the incidence of scarlet fever between study groups was statistically significant, but this result should be questioned due to the small number of patients with this condition in both cohorts.
Alanine aminotransferase and AST activities were greater among children with positive EBV and CMV antibodies throughout the disease course (at the 4-week follow-up), but a significant difference was observed for both transaminases only in the first week. Similar results were reported by Sohn et al. [1].
The γ-GT activity and total serum bilirubin concentration did not differ significantly between the study groups. The percentage of children with direct bilirubin concentrations exceeding 20% of total serum bilirubin was also comparable. No disorders of the synthetic function of the liver were found in any of the patients; the Quick index was normal.
In the second group, abnormal abdominal ultrasound results were obtained significantly more often. Particular abnormalities (hepatomegaly, enlarged lymph nodes in the liver hilum, thickening of the gallbladder wall, or accumulation of fluid around the gallbladder) also occurred more frequently in this group, although the differences were no longer statistically significant.
Children with EBV and CMV dual positivity met the criteria (created for our study), indicating a more severe course of hepatitis (than usually observed in IM) more than twice as often as did patients from the first group.
In children with EBV monoinfection, antibiotic therapy after hospital admission was used significantly more often (usually due to acute bacterial pharyngitis). The rate of prescription of antibiotics in the first group reached 50.61%, which is consistent with the results of studies by other authors [10, 18]. In children diagnosed with bacterial pharyngitis, the McIsaac clinical criteria were met, and the S. pyogenes rapid test was performed. Patients with streptococcal pharyngitis received cefuroxime after hospital admission (although it is not the first-line therapy), because EBV infection may predispose to bacterial superinfections of etiology other than S. pyogenes (bacterial superinfection of mixed etiology).
The strength of this work is the large number of patients included in our study.
An inherent limitation of this study is its retrospective nature, which is based on medical records, and the absence of some data in a few cases. Only patients who required hospitalization were evaluated. Another limitation is the lack of confirmation of acute CMV infection with additional tests, such as polymerase chain reaction (PCR), IgG seroconversion, or detection of CMV antigen; therefore, a false-positive result due to antibody cross-reaction cannot be ruled out.
In conclusion, the results of our study highlighted several characteristics of children with dual positivity for EBV and CMV compared to those who were positive for EBV only. In children with dual positivity for EBV and CMV, we observed more pronounced clinical symptoms of hepatitis and more frequently abnormal ultrasound images of the liver and bile ducts. These differences may reflect true EBV and CMV coinfection or may result from individual variability in immune system function.
Nevertheless, the above uncertainties may be clarified in subsequent prospective studies using broader molecular diagnostic methods.