Introduction
Aortic stenosis (AS) represents the most prevalent valvular heart disease in developed countries, driven by an active pathophysiology involving lipid infiltration, inflammation, and progressive calcification rather than passive degeneration. The natural history of severe, symptomatic AS is poor; historically, survival declines precipitously after symptom onset, averaging 5 years for angina, 3 years for syncope, and less than 2 years for heart failure. Given that AS prevalence approaches 10% in octogenarians, effective management constitutes a critical public health priority [1]. For decades, surgical aortic valve replacement (SAVR) was the sole standard of care. However, the advent of transcatheter aortic valve implantation (TAVI) has revolutionized this landscape. Initially reserved for inoperable patients with prohibitive surgical risk (EuroSCORE II > 20% or STS > 10%), TAVI has since demonstrated non-inferiority or superiority to SAVR across the entire risk spectrum. The landmark 10-year data from the NOTION trial and 5-year data from PARTNER 3 have allayed early concerns regarding long-term durability, validating TAVI as a durable solution even for lower-risk cohorts [2]. This accumulation of evidence has culminated in a paradigm shift reflected in the 2025 ESC/EACTS Guidelines for the Management of Valvular Heart Disease. The recommendation for TAVI has been expanded to patients aged ≥ 70 years, shifting the decision matrix from surgical risk assessment to lifetime management. In this new era, the heart team must look beyond the index procedure to plan for future coronary access and potential reinterventions (TAVI-in-TAVI) over a patient’s extended life expectancy. The latest evidence from the PARTNER 3 and Evolut Low Risk trials supports this approach [3–5]. Consequently, the modern TAVI operator must navigate complex scenarios previously considered contraindications, including bicuspid aortic valves, pure aortic regurgitation, and small aortic annuli (Table I). This review synthesizes the latest evidence on procedural optimization (the “minimalist” TAVI approach), device selection in complex anatomies, and the emerging role of biomarkers in predicting valve degeneration.
Table I
Integrated decision matrix for TAVI versus SAVR in the contemporary era
[i] CABG – coronary artery bypass grafting, CAD – coronary artery disease, ICD – implantable cardioverter-defibrillator, LV – left ventricular, LVOT – left ventricular outflow tract, MR – mitral regurgitation, PCI – percutaneous coronary intervention, RV – right ventricular, SAVR – surgical aortic valve replacement, TAVI – transcatheter aortic valve implantation.
Patient selection
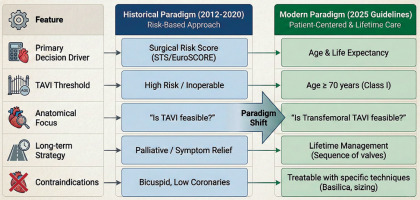
Patient selection for TAVI has evolved from a risk-based “rescue” strategy to a comprehensive lifetime management approach (Figure 1). While early guidelines relied heavily on surgical risk scores (EuroSCORE II, STS-PROM), the 2025 ESC/EACTS Guidelines have established age and estimated life expectancy as the primary determinants of treatment modality [5]. The ideal TAVI candidate should be ≥ 70 years old, with suitable anatomy, including a small aortic annulus [6], femoral access feasibility [7], and symmetric valvular calcifications, which are associated with lower risk of paravalvular leak. Clinical features that correlate with better long-term outcomes include normal systolic pulmonary arterial pressure, lower left ventricular filling pressures, and preserved right ventricular systolic function [8]. Furthermore, comorbidities play an important role in post-TAVI outcomes. The absence of chronic kidney disease, insulin-dependent diabetes, advanced coronary artery disease, and atrial fibrillation correlates with improved survival [9]. Registries such as NOTION, PARTNER 2, and FRANCE-TAVI, with median follow-up periods of 5–10 years, have demonstrated that the principal causes of adverse outcomes and mortality following TAVI are attributable to comorbidities rather than valve-related complications. Notably, most deaths are non-cardiovascular in origin, underscoring the disease-modifying role of TAVI in the treatment of AS in elderly patients.
Figure 1
Evolution of TAVI decision-making: from risk-based selection to lifetime management
BASILICA – bioprosthetic or native aortic scallop intentional laceration to prevent iatrogenic coronary artery obstruction, EuroSCORE – European System for Cardiac Operative Risk Evaluation, STS – Society of Thoracic Surgeons, TAVI – transcatheter aortic valve implantation.
Despite a decade of investigation, evidence regarding the long-term advantages of TAVI over SAVR remains inconclusive. Biancari et al. studied patients with EuroSCORE II < 4% who underwent TAVI or SAVR for AS in the OBSERVANT study [10]. This prospective, observational, non-randomized study demonstrated that 10-year survival among low-risk patients who received early-generation TAVI prostheses was inferior to that observed with SAVR. Similarly, Awad et al. performed a systematic review and meta-analysis comparing the performance of TAVI and SAVR [11]. TAVI was associated with significantly increased risks of 2-year permanent pacemaker implantation and major vascular complications compared with SAVR. However, TAVI significantly reduced rates of patient–prosthesis mismatch and postoperative atrial fibrillation, whereas SAVR was associated with lower rates of paravalvular leak. Thus, TAVI and SAVR remain valid alternatives, and treatment decisions should be individualized based on annular and aortic root anatomy, operative risk, and comorbidity burden.
Yuan et al. conducted a systematic literature search to assess the effects of postoperative neurological events on mortality following TAVI [12]. Irrespective of other postoperative complications, TAVI appears superior to SAVR with regard to 1-year all-cause mortality. TAVI is therefore recommended for elderly patients with symptomatic severe aortic stenosis who are at prohibitive surgical risk or have contraindications to SAVR.
Pre-TAVI diagnostic work-up
Patients undergoing TAVI require computed tomography (CT) imaging of both the peripheral arterial vasculature and the heart. Evaluation of the peripheral vasculature is essential to assess the bifurcation of the superficial and deep femoral arteries and to identify any aortic pathology. This information is necessary for safe vascular puncture and device delivery. Additionally, assessment of femoral artery caliber enables prediction of the appropriate balloon diameter for achieving hemostasis following the procedure. Cardiac CT provides critical information regarding optimal fluoroscopic projections, annular dimensions, and the presence and distribution of calcification or fibrosis [13–15].
Self- versus balloon-expandable device
The choice between self-expandable valves (SEVs) and balloon-expandable valves (BEVs) for TAVI depends on aortic valve morphology, annular dimensions, and the presence of aortic regurgitation. Current evidence suggests that SEVs tend to demonstrate superior hemodynamic performance, with lower pressure gradients and reduced incidence of patient–prosthesis mismatch, as demonstrated by the SMALL-TAVI registry published in EuroIntervention [16].
A meta-analysis by Ahmed et al. [6] showed that BEVs were associated with significantly smaller indexed effective orifice areas and higher transvalvular mean pressure gradients compared with SEVs. The risk of prosthesis–patient mismatch was significantly higher among patients receiving BEVs. Although no significant differences were observed between BEVs and SEVs regarding 30-day and 1-year all-cause mortality, 30-day stroke rates, vascular complications, paravalvular leak, or permanent pacemaker implantation, patients receiving BEVs demonstrated a significantly increased risk of 1-year cardiovascular mortality compared with those receiving SEVs.
Pre-existing conduction abnormalities, such as atrioventricular block, left or right bundle branch block, or trifascicular block, favor the selection of BEVs, which are associated with lower rates of permanent pacemaker implantation following TAVI. Conversely, SEVs offer the advantage of repositionability before final deployment and superior conformability to the aortic annulus, making them preferable in cases of concomitant aortic regurgitation or larger annuli.
Indications for TAVI continue to expand and now include patients with large annuli. Implantation of larger prostheses is often associated with greater procedural complexity, which may affect outcomes. Wilczek et al. reported that TAVI procedures requiring an Evolut R 34 prosthesis are more technically challenging than those with smaller valves [17]. Paravalvular leaks are observed more frequently with the Evolut R 34, resulting in lower device success rates.
Prevention of TAVI complications
The growing adoption of TAVI has created a need for standardized pathways to optimize procedural efficiency, minimize complications, and facilitate early hospital discharge. The most common complications remain vascular events related to femoral access and the requirement for permanent pacemaker implantation [18].
Romagnoli et al. developed a less invasive, totally endovascular (LITE) technique for TAVI [19], consisting of precise access-site puncture using combined angiographic-guidewire and ultrasound guidance, radial or ulnar artery as the default secondary access, and non-invasive pacing via guidewire stimulation or external programming of a pre-existing permanent pacemaker. In a study of 1,022 consecutive TAVI patients, major access-site vascular complications occurred in approximately 1.2% of cases, with an equivalent incidence of VARC-3 type 2 or greater bleeding events. Observed mortality was substantially lower than predicted by established risk models: EuroSCORE II estimated approximately 6%, STS-PROM approximately 4%, and the STS/ACC TVT TAVR mortality score approximately 3%.
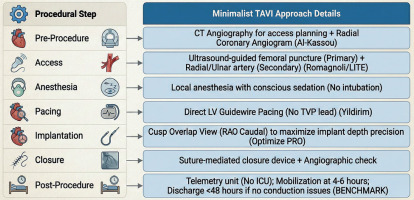
The BENCHMARK registry represents another example of procedural pathway optimization, emphasizing standardized best practices throughout the entire care process, from preprocedural evaluation to discharge (Figure 2) [20]. This investigator-initiated, observational, multicenter registry enrolled 1,491 TAVI patients. Key elements included comprehensive assessment of comorbidities and frailty, CT- and ultrasound-guided vascular access, early evaluation for permanent pacemaker requirement, and expedited mobilization and discharge. Hospital length of stay decreased from approximately 7–8 days to 5–6 days, with reductions in intensive care unit time and procedural duration. Clinical outcomes remained excellent, with 30-day mortality of approximately 0.6% and stable rates of stroke, major bleeding, acute kidney injury, and valve-related readmission. Some analyses also suggested a modest reduction in permanent pacemaker implantation.
Figure 2
The minimalist TAVI pathway: procedural optimization for reduced complications and early discharge
CT – computed tomography, ICU – intensive care unit, LITE – less invasive-totally endovascular, LV – left ventricular, RAO – right anterior oblique, TAVI – transcatheter aortic valve implantation, TVP – temporary venous pacing.
Regarding pacing strategies, complications may arise from temporary transvenous pacing. Yildirim et al. evaluated the efficacy and safety of left ventricular guidewire pacing versus conventional right ventricular pacing in 635 TAVI patients [21]. Left ventricular guidewire pacing was associated with fewer pericardial complications (0% vs. 1.5%), lower rates of major vascular events (1.2% vs. 3.5%), and reduced bleeding, supporting a safer overall profile.
Implantation depth represents another important consideration, particularly regarding permanent pacemaker requirement and postprocedural aortic regurgitation. For SEVs, the cusp-overlap technique facilitates visualization of the non-coronary cusp, which serves as the anatomical landmark for achieving a higher implantation position that is safer for the conduction system. The Optimize PRO study demonstrated that the cusp-overlap technique improves clinical outcomes, with a 30-day permanent pacemaker rate of 6.4% compared with 11.1% in the overall cohort [22]. The incidence of more than mild aortic regurgitation was low (< 1%). Mean implantation depth at the non-coronary cusp was approximately 3–4 mm, and hospital length of stay was reduced to 1–2 days.
Vascular injury and bleeding complications remain common following TAVI. Al-Kassou et al. investigated the impact of preprocedural coronary angiography access site and found that transradial access was associated with significantly lower rates of vascular complications following subsequent TAVI compared with transfemoral access [23]. However, although the rate of major bleeding was numerically lower with transradial access, no statistically significant association was detected between coronary angiography access site and TAVI-related bleeding complications. A further study found that age was not significantly associated with pacemaker implantation after TAVI, although additional investigation is needed to evaluate differences in other TAVI-related complications across age groups [24].
To investigate temporal changes in biomarker levels as markers of native aortic annulus calcification and early-stage bioprosthetic valve degeneration, Sorysz et al. measured serum levels of matrix metalloproteinase-3, matrix metalloproteinase-9, and osteopontin using positron emission tomography/computed tomography [24]. The authors identified significant negative correlations between these biomarkers and follow-up effective orifice area index and effective orifice area.
Özderya et al. evaluated the relationship between aortic knob calcification assessed on preoperative chest radiography and the requirement for post-TAVI pacemaker implantation [25]. Aortic knob calcification may serve as a cost-effective and readily accessible predictor of permanent pacemaker requirement following TAVI [26].
Aortic stenosis and coronary artery disease
Patients with AS frequently have concomitant obstructive coronary artery disease (CAD), as both conditions share similar pathophysiological mechanisms and risk factors [27]. Consequently, screening for CAD is essential when evaluating patients for TAVI. The optimal management strategy has been a subject of considerable debate, particularly regarding the timing of percutaneous coronary intervention (PCI) [28–32]. The principal considerations include coronary engagement (which may be challenging when PCI is performed after TAVI), functional assessment (which is often confounded by the hemodynamic effects of AS), and contrast-induced nephropathy (when PCI is performed concomitantly with TAVI). Tarantini et al. emphasized the importance of valve selection when future coronary access is anticipated, particularly in younger patients with multiple cardiovascular risk factors [33]. In such cases, a balloon-expandable intra-annular valve may represent the optimal choice.
The latest ESC/EACTS guidelines on valvular heart disease include updated recommendations for CAD evaluation in patients undergoing TAVI [34]. PCI before TAVI should be considered for major epicardial vessels (> 2.5 mm diameter) with > 90% stenosis (Class IIa) and may be considered for stenoses > 70% in the proximal segments of major vessels (Class IIb). Invasive coronary angiography may be omitted if preprocedural CT excludes obstructive CAD. These recommendations underscore the greater prognostic benefit of treating severe AS with TAVI compared with performing PCI for moderate coronary stenoses in patients with significant comorbidities.
Wilimski et al. evaluated the impact of prior coronary revascularization, whether by PCI or coronary artery bypass grafting, on clinical outcomes following TAVI [35]. TAVI appears to be a safe and effective procedure for the treatment of severe AS in patients with previous coronary revascularization.
Bicuspid aortic valve
A systematic review by Goulden et al. [36] demonstrated high procedural success rates (~93%) and marked hemodynamic improvement in patients with bicuspid aortic valve and severe AS undergoing TAVI, with significant reductions in mean pressure gradient (from 47 to 10 mm Hg) and significant increases in effective orifice area. Over the past 3 years, accumulating registry data have supported the extension of TAVI to this anatomical subset. In particular, the BIVOLUTX, Neo2 BAV, and STABILITY registries [37–39] demonstrated high procedural success rates, significant improvements in pressure gradients and valve area, low rates of paravalvular leak, acceptable long-term mortality, and very few cases requiring reintervention (Table II). Data on permanent pacemaker implantation in patients undergoing TAVI for bicuspid aortic valve stenosis remain limited. Süygün et al. demonstrated that preprocedural right bundle branch block on electrocardiography and shorter membranous septum length are independent risk factors for pacemaker implantation; these parameters should be considered during preprocedural planning to guide clinical decision-making [40].
Table II
Comparative profile of TAVI in complex anatomical subsets
Aortic regurgitation
TAVI for pure aortic regurgitation (AR) represents an emerging option for patients at high surgical risk. Evidence remains limited, but registry data are promising (Table II). The principal challenge with TAVI in AR is the absence or paucity of annular calcification, which compromises secure valve anchoring; notably, the PANTHEON registry reported a 12% incidence of device migration [41]. Additionally, complications such as hemorrhage and permanent pacemaker implantation occurred more frequently compared with TAVI for AS.
A key limitation of TAVI for AR is the lack of a standardized procedural protocol comparable to that established for AS. Development of a dedicated approach specific to this indication is warranted.
Valve-in-valve
Valve-in-valve (ViV) TAVI represents a valid alternative for degenerated aortic bioprostheses in elderly patients. The choice between redo surgery and ViV should be individualized based on patient frailty, vascular access suitability, prosthesis dimensions, and comorbidity burden. The selection between SEVs and BEVs does not significantly impact 1-year survival [42]. A meta-analysis by Giordana et al. [43], including 26 studies with 1,448 patients and a median follow-up of 12–13 months, reported median mortality rates of 6.5% at 30 days and 14.5% at 1 year. Long-term follow-up data are needed to better define the durability and benefit of ViV treatment.
Follow-up after TAVI
TAVI has altered the natural history of severe AS and significantly improved patient outcomes and quality of life (Table III). Patients undergoing TAVI demonstrate improved medium-term survival (5-year follow-up) compared with untreated AS. An international registry [44] involving 2,414 patients over 13 years reported overall survival of 67.6% at 5 years and 26.9% at 10 years, whereas mortality in untreated AS is estimated at 45% at 4 years.
Table III
Evolution of TAVI outcomes and complication management
However, the principal benefits of TAVI extend beyond mortality reduction. The most meaningful outcomes are reflected in quality of life and reduction in cardiovascular death. The FRANCE-TAVI registry [45], which enrolled 3,159 TAVI patients, demonstrated initial improvement in quality of life, with subsequent deterioration in 15% of patients within the following year; 19% of these patients died within 1 year. Predictors of adverse outcomes included frailty, diabetes, chronic obstructive pulmonary disease, critical preoperative status, reduced left ventricular ejection fraction, elevated mean aortic gradient at discharge, permanent pacemaker requirement, anticoagulation therapy, and prolonged hospital length of stay. These findings reflect the typical TAVI population: elderly patients with multiple comorbidities. Consequently, while TAVI substantially reduces major cardiovascular events, mortality related to non-cardiovascular comorbidities (such as respiratory and neurological disorders) remains unchanged. This underscores the need for standardized, optimized protocols to minimize procedural complications and hospitalization duration.
Data on sex-specific outcomes following TAVI remain limited. In a recent study of 519 men who underwent TAVI, multivariate analysis demonstrated that serum albumin levels, atrial fibrillation, and STS score were independently associated with all-cause mortality [46].
Given the role of inflammatory cytokines in aortic stenosis progression, Özbek et al. investigated the impact of epicardial adipose tissue on outcomes in patients undergoing TAVI [47]. Multivariate analysis revealed that epicardial adipose tissue volume and albumin level were independently associated with adverse outcomes. Recently, investigators examined whether iron deficiency might contribute to impaired physical performance improvement following TAVI. Błaszkiewicz et al. [48] found that iron deficiency had no significant effect on exercise capacity before or after TAVI, suggesting that iron deficiency does not impact physical capacity in patients with severe AS.
TAVI and mitral regurgitation
The clinical impact of baseline mitral regurgitation (MR) on outcomes following TAVI remains incompletely characterized. Zheng et al. assessed the impact of baseline MR on outcomes in 120 consecutive patients with severe AS undergoing TAVI [49]. Overall survival did not differ significantly between patients with non-significant and significant baseline MR. However, patients demonstrating MR improvement after TAVI had significantly higher survival than those with unchanged or worsened MR during 2-year follow-up. Functional class generally improved at 1 year, with only 8% of patients with non-significant MR and 17% of those with significant MR remaining in NYHA class III or IV. Significant baseline MR was not associated with increased risk of all-cause mortality at 2 years and improved in most patients by 1 year, whereas patients with unchanged or worsened MR demonstrated increased all-cause mortality.
Rangel et al. investigated changes in MR severity after TAVI and their effect on 5-year mortality [50]. Among 820 patients, 167 showed improvement in MR grade. Systolic pulmonary artery pressure and NT-proBNP decreased in patients with MR improvement. However, there was no significant difference in 5-year mortality based on baseline MR severity, nor was MR improvement associated with reduced mortality compared with patients whose MR remained stable or worsened.
Conclusions
TAVI has fundamentally altered the natural history of severe AS, improving quality of life and reducing the incidence of major adverse cardiovascular events in elderly patients. However, mortality remains substantial, largely attributable to the inherent frailty of this population. Implementation of standardized procedural pathways is essential to minimize complications in frail patients and reduce hospitalization-related morbidity and mortality. The extension of TAVI to younger patients, as endorsed by the 2025 ESC/EACTS guidelines for valvular heart disease, represents a paradigm shift toward comprehensive lifetime management of this condition.
Several key areas warrant further investigation. First, long-term durability data beyond 10 years are needed, particularly for newer-generation devices implanted in younger, lower-risk patients for whom bioprosthetic valve degeneration and the feasibility of reintervention (TAVI-in-TAVI) become critical considerations. Second, the optimal management of concomitant coronary artery disease – including the timing of revascularization and strategies to preserve future coronary access – requires further clarification through dedicated randomized trials. Third, the extension of TAVI to complex anatomical subsets, including bicuspid aortic valves and pure aortic regurgitation, necessitates continued refinement of patient selection criteria, procedural techniques, and device design.
The emerging role of biomarkers such as MMP-3, osteopontin, and epicardial adipose tissue volume in predicting structural valve deterioration offers a promising avenue for biological risk stratification. Integration of these markers into clinical practice may enable earlier identification of patients at risk for accelerated valve degeneration and inform preventive strategies. Furthermore, the landmark findings of the EARLY TAVR trial challenge conventional management of asymptomatic severe AS, potentially expanding indications to include prophylactic intervention before symptom onset or irreversible myocardial remodeling.
In conclusion, TAVI has evolved from a rescue therapy for prohibitive-risk patients to the cornerstone of aortic stenosis management across the risk spectrum. As indications continue to expand and patient longevity increases, the field must prioritize lifetime management strategies, procedural standardization, and the integration of novel prognostic tools to optimize outcomes for this growing patient population.







