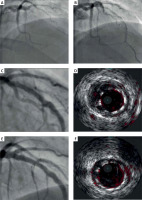
A 33-year old man was admitted to the department of cardiology with a diagnosis of ST-segment elevation myocardial infarction (MI) of the anterior wall after the pre-hospital cardiac arrest. On admission, the patient was conscious, hemodynamically stable with persistent chest pain. The medical history of the family for premature cardiac disease was negative. Prior to admission, the patient had been diagnosed with hypertension and dyslipidaemia. The echocardiography showed left ventricular ejection fraction mildly reduced to 40%, driven by akinesia of the apex and adjacent segments. Urgent coronary angiography revealed an occlusion in the middle segment of the left anterior descending artery (LAD) (Figure 1 A). Due to the presence of a large thrombus, aspiration thrombectomy was performed following predilatation with a semi-compliant balloon of 2.5 × 20 mm to 12 atm (Figure 1 B). Consequently, three cardiac arrests occurred in the mechanism of ventricular fibrillation (VF) successfully treated with defibrillation. In spite of several thrombectomy passages, the thrombotic mass remained large and blood flow was impeded. The operator decided to administer a GP IIb/IIIa inhibitor (abciximab) and to stabilize the patient with implantation of a suboptimally sized drug-eluting stent, Xience Pro (Abbott Laboratories, IL, USA), of 3.5 × 28 mm to 16 atm (Figure 1 C). There were no further episodes of VF and the patient remained haemodynamically stable. Further pharmacological treatment included dual antiplatelet therapy (ASA plus ticagrelor), and continuous abciximab infusion for 12 h followed by low molecular weight heparin in a therapeutic dose. After 3 days, follow-up coronarography with intravascular ultrasound (IVUS) revealed a significantly malapposed stent with a good flow in the LAD (Figure 1 D). The IVUS-guided postdilatation with non-compliant balloons of 4.0 × 15 mm and 4.5 × 15 mm to 20 atm provided good stent apposition (Figures 1 E, F). The further hospitalization was uneventful. In the 3-year follow-up, the patient has been asymptomatic and his left ejection fraction has returned to normal.
Figure 1
A – initial left coronary artery angiography with acute occlusion in the mid-LAD. B – partially restored flow in distal segment of the LAD exhibiting a large thrombus burden. C – final angiographic outcome of the index procedure after implantation of a drug-eluting stent of 3.5 × 28 mm to 16 atm. D – ultrasound examination showing underdeployment of the stent in the mid-LAD. E – angiogram of the LAD with the stent post-dilated with a non-compliant balloon of 4.0 × 15 mm to 20 atm. F – optimal stent apposition confirmed in IVUS examination
Percutaneous coronary interventions (PCI) in acute settings differ significantly from planned procedures. During primary PCI, any instrumentation in the vessel with a high thrombus burden increases the risk of distal embolization (DE), worsening the patient’s prognosis [1]. Thrombectomy was introduced to mitigate myocardial damage caused by DE but did not appear to be better in comparison to PCI alone in the TOTAL study [2]. Post-dilation with high pressure constituting a basis for an optimal effect of PCI is associated with impaired microvascular perfusion in acute settings [3]. Moreover, during the MI circulating catecholamines constrict the arteries, rendering vessel diameter difficult to assess and to properly treat. Therefore, new approaches for index PCI in patients with a high thrombus burden need to be established in order to improve outcomes.
In the present case, the strategy based on restoring flow at the first stage, optimal medical therapy and optimization of the implanted stent was uncomplicated and provided a good outcome in the 3-year observation. We assume that two-stage procedures might be a safe and effective way in selected primary PCIs, especially with a high thrombus burden. Nonetheless, the presented method should not be applied routinely. Stent underexpansion increases the risk of passing the guidewire under the stent struts, which might lead to catastrophic procedure outcomes. Moreover, stent malapposition is a well-known factor of acute stent thrombosis.








