Introduction
Stanford acute type A aortic dissection (ATAAD) has a mortality rate that can reach up to 20% [1]. ATAAD repair necessitates intricate circulatory and cerebral protection. Many neuroprotective techniques have been offered; however, the ideal strategy for ATAAD repair remains disputable [2]. The axillary, common carotid, innominate, femoral arteries, and the ascending aorta or left ventricular apex are currently the most preferred sites for arterial cannulation [3]. Nevertheless, it is currently unclear which cannulation method is best for repairing ATAAD.
Currently, the favored approach for reducing neurological adverse effects is moderate hypothermia with antegrade cerebral perfusion (ACP) via the axillary artery (AXA) [4]. However, AXA cannulation has potential complications such as relatively prolonged operative time and brachial plexus injury, limb ischemia, and seroma [3]. Innominate artery (IA) cannulation using a single sternal approach has been more frequently performed in recent years to avoid these complications associated with additional infraclavicular incision and AXA cannulation [5–7].
In a published study, Di Eusanio was the first to propose using the IA as an arterial cannulation site for aortic surgery. However, he performed the procedure in only two of the 55 patients with ATAAD [5]. Despite recent reports of the use of IA cannulation in ATAAD repair, there are still concerns about the uninterrupted continuation of arterial inflow in the site [6, 7].
Aim
The aim of this study was to compare IA and AXA cannulation in ATAAD repair with early- and mid-term results.
Material and methods
Patient population
This study was conducted with the approval of Hitit University Erol Olçok Education and Research Hospital Non-Invasive Research Ethics Committee (Date: 10/07/2024, Decision No: 2024-38). All procedures were carried out in accordance with the ethical rules and the principles of the Declaration of Helsinki. All patients provided informed consent for data collection. The preoperative, intraoperative, and postoperative data were obtained from the electronic hospital and patients’ records. This study excluded patients who underwent elective surgery or redo operations for subacute or chronic type A aortic dissection.
The patients’ baseline demographic characteristics are summarized in Table I. Forty-six patients who underwent emergency surgical repair for ATAAD between January 2021 and April 2024 were retrospectively evaluated. ATAAD was defined as an aortic dissection (AD) detected within 14 days after the onset of symptoms. The anatomy of AD was classified according to the DeBakey classification [8]. Prior to the procedures, all patients underwent transthoracic echocardiography (TTE) performed by expert cardiologists. The radiologist and cardiovascular surgery team then performed computed tomography angiography (CTA), thoroughly examined the images, and verified the findings intraoperatively (Figures 1, 2). At presentation, dissection extent was DeBakey type I in 37 (80.4%) and type II in 9 (19.6%). The patients were grouped as those with right AXA cannulation (group 1, n = 18) and IA cannulation (group 2, n = 28). The groups were compared based on preoperative, intraoperative, and postoperative data. The GERAADA score was calculated individually for each patient using a web-based tool by a cardiovascular surgeon who was blind to the study [9]. All procedures were carried out by different surgeons using similar techniques.
Table I
Demographic and preoperative data
| Parameter | Axillary cannulation (group 1) N = 18 | Innominate cannulation (group 2) N = 28 | P-value |
|---|---|---|---|
| Gender | 0.772+ | ||
| Male | 10 (55.6%) | 15 (51.7%) | |
| Female | 8 (44.4%) | 13 (44.8%) | |
| Age | 64.4 ±12.5 | 61.6 ±11.7 | 0.442* |
| Height [cm] | 166 ±7.7 | 167.2 ±8.9 | 0.619* |
| Weight [kg] | 78.5 ±14.6 | 84.7 ±13.6 | 0.154* |
| DM | 7 (38.8%) | 18 (64.2%) | 0.114+ |
| HT | 13 (72.2%) | 24 (85.7%) | 0.473+ |
| HL | 5 (27.7%) | 15 (53.5%) | 0.137+ |
| CAD | 9 (50%) | 10 (35.7%) | 0.365+ |
| Stroke | 0 | 2 (7.1%) | 0.517+ |
| COPD | 1 (5.5%) | 3 (10.7%) | 1.000+ |
| CKD | 1 (5.5%) | 7 (24.9%) | 0.130+ |
| Smoking | 5 (27.7%) | 8 (28.5%) | 1.000+ |
| Cardiac tamponade | 6 (33.3%) | 6 (21.4%) | 0.493+ |
| EF | 52 ±7.3 | 50.5 ±6.9 | 0.481* |
| Admission HCT | 39.4 ±5.4 | 38.1 ±4.9 | 0.388* |
| GERAADA Score | 17.3 ±13.3 | 11.4 ±4.6 | 0.035* |
Figure 1
A – Preoperative computed tomography angiography(CTA) image showing innominate artery dissection (blue arrow) and left carotid artery occlusion with thrombi (yellow arrow). B – Postoperative CTA image of the same patient showing the innominate artery cannulation site (arrow)
The choice between AXA and IA cannulation was ultimately left to the judgment of the operating surgeon. IA was not used for cannulation in the presence of atherosclerosis, aneurysmal dilatation of the IA, small IA diameter, or extension of ATAAD to the IA.
Operative technique
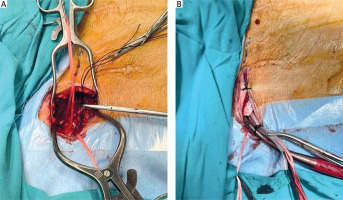
Prior to general anesthesia, the patients were monitored using near-infrared spectroscopy. In all patient groups, left radial artery was cannulated to measure systemic arterial blood pressure. After the standard median sternotomy incision, the incision was extended 2–3 cm cranially in the neck for IA cannulation. The left innominate vein was encircled with silastic tape before incising the tissues within the avascular plane. IA and other arch vessels were subsequently encircled with tape. After systemic heparinization (300–400 IU/kg to achieve activated coagulation time > 480 s), if the origin of the IA was not severely dissected, a partial side clamp was applied to the distal IA. A 10 mm Dacron graft was anastomosed to the true lumen of the IA with polypropylene 6-0 and an outer Teflon felt strip (Figure 3). The graft was de-aired then connected to the arterial line (Figure 4 A).
Figure 3
A – Illustration of innominate artery cannulation, B – 10 mm Dacron graft end-to-side anastomosis to innominate artery using side biting clamp
Figure 4
A – Intraoperative images of innominate cannulation, B – ligated graft (arrow) after weaning from cardiopulmonary bypass
An incision was performed in the right deltopectoral groove in the infraclavicular fossa, parallel to the clavicle, in order to access the right AXA in patients who were selected for AXA cannulation. The axillary vein was exposed by incising the pectoralis major fascia and splitting its fibers. The pectoralis minor muscle was retracted laterally to access the brachial plexus and AXA (Figure 5 A). Following systemic heparinization, a straight-tipped arterial cannula was inserted into the AXA using the Seldinger technique with the assistance of a guidewire and dilators (Figure 5 B).
Figure 5
A – The right axillary artery is exposed and encircled with tape. B – The arterial cannulation process is demonstrated, showing a straight arterial cannula secured to the skin after being placed in the right axillary artery
Cardiopulmonary bypass (CPB) was accomplished using arterial cannulation and venous drainage into the right atrium via a two-stage cannula. A catheter was inserted into the right superior pulmonary vein to facilitate suction. Del Nido cardioplegic solution was used to achieve cardioplegic arrest after aortic cross-clamping (XCL). After examination of the ascending aorta, proximal aortic repair was performed by selecting the appropriate surgical procedure according to the condition of the aortic valve, aortic root, sinus of Valsalva and coronary buttons. Then, the distal anastomosis was performed in an open fashion with ACP. In 4 cases, requiring arch repair, ACP and moderate hypothermic circulatory arrest (20.1–28°C) were performed. Depending on the aortic diameter and the site of its entry tear, the distal repair involved either hemiarch or total arch replacement. Both groups’ CPB flow was controlled during the ACP to maintain the radial artery pressure between 50 and 60 mm Hg, with a flow rate of 10–12 ml/kg/min. In addition to the Trendelenburg position, the head of the patient was packed in ice for brain protection, and hydrocortisone and mannitol medications were applied. At the end of the repair, the graft anastomosed to the IA was cut and oversewn with a double 5-0 polypropylene running suture. In cases where AXA cannulation was performed, AXA was primarily repaired with prolene sutures after decannulation.
Study endpoints and follow-up
Primary endpoints compared between the AXA cannulation and IA cannulation groups were operative mortality (defined as all-cause mortality at 30 days); occurrence of postoperative neurological injury at 30 days; major adverse cardiac and cerebrovascular events (MACCEs), all-cause mortality, and need for emergency cardiac surgery. Secondary endpoints were re-exploration, stroke, upper limb ischemia, wound infections, and other complications.
Following the procedure, patients were monitored at weekly, monthly, quarterly, semi-annual, and annual intervals. Outcome data were collected from the electronic hospital database, as well as from patients’ general practitioners, the patients, and their families. These included postoperative, 1-month, and late follow-up data.
Statistical analysis
The Kolmogorov–Smirnov test was used to analyze the normal distribution of the data. Normally distributed continuous variables were expressed as mean ± standard deviation (SD), or median values if abnormally distributed. Categorical variables were expressed as numbers and percentages. Demographic characteristics and perioperative variables were compared using the independent samples t-test or Mann–Whitney-U test for continuous variables and the χ2 test or Fisher exact test for categorical variables between two groups. Patients were compared between groups in terms of demographic data, intraoperative data, and postoperative data. Univariate logistic regression analysis was performed to determine the risk factors for predicting early mortality. Moreover, Hosmer–Lemeshow goodness-of-fit statistics were used to assess the model fit. The odds ratio, 95% confidence intervals (95% CI), and significance levels for each variable were determined. The Kaplan–Meier test was used to demonstrate the mid-term effect of cannulation site on mortality. In all statistical tests, a p-value < 0.05 was accepted as significant. All statistical analyses were performed using IBM SPSS Statistics for Windows, version 25.0 (Armonk, NY: IBM Corp.).
Results
In the study cohort, the site used for initial cannulation was AXA in 18 (39.1%) and IA in 28 (60.9%) patients. Patient characteristics and demographics are summarized in Table I. When the preoperative data were evaluated, no statistically significant difference was found between the groups in terms of comorbid factors (p > 0.05). A total of 83.3% of patients had DeBakey type I in the AXA group, and 78.5% of patients had DeBakey type I in the IA group.
CPB, XCL, and hypothermic circulatory arrest times were similar in both groups. In the AXA group, hemiarch replacement was performed in 12 (66.6%) patients, while isolated ascending aortic replacement was performed in 5 (27.7%) patients. In the IA group, hemiarch replacement was performed in 20 (71.4%) patients, and ascending aortic replacement was performed in 4 (14.2%) patients. ACP was performed 12 (66.6%) of patients in the AXA group, while it was performed in 27 (96.4%) patients in the IA group (p = 0.041). AXA cases had a shorter ACP time (23 ±5.74 versus 28.7 ±8.3 min, p = 0.03) (Table II). Ascending aortic diameters were significantly larger in the AXA group than in the IA group (55 ±2.4 versus 47.5 ±3.43 mm, p < 0.001).
Table II
Perioperative, procedural, and postoperative data
| Parameter | Axillary cannulation (group 1) N = 18 | Innominate cannulation (group 2) N = 28 | P-value |
|---|---|---|---|
| DeBakey type | 1.000+ | ||
| Type 1 | 15 (83.3%) | 22 (78.5%) | |
| Type 2 | 3 (16.7%) | 6 (21.4%) | |
| Aortic diameter [mm] | 55 ±2.4 | 47.5 ±3.43 | 0.001* |
| CPB time [min] | 138.9 ±60.8 | 136 ±39.3 | 0.843* |
| Cross-clamp time [min] | 99.1 ±31.4 | 92.6 ±24.5 | 0.442* |
| Minimum temperature | 20 ±3.4 | 25 ±4.2 | 0.001* |
| ACP | 12 (66.6%) | 27 (96.4%) | 0.041+ |
| ACP time [min] | 23.0 ±5.74 | 28.7 ±8.3 | 0.03* |
| TCA | 6 (33.3%) | 2 (7.1%) | 0.041+ |
| TCA time [min] | 23 ±2.55 | 28.5 ±4.95 | 0.09* |
| Root intervention | 0 | 1 (3.5%) | 1.000+ |
| Hemiarch intervention | 12 (66.6%) | 20 (71.4%) | 0.872+ |
| Total arch intervention | 1 (5.5%) | 3 (10.7%) | 0.246+ |
| Only ascending aortic intervention | 5 (27.7%) | 4 (14.2%) | 0.398+ |
| Re-exploration | 5 (27.7%) | 3 (10.7%) | 0.230+ |
| Postoperative stroke | 2 (11.1%) | 1 (3.5%) | 0.549+ |
| Postoperative TIA | 0 | 1 (3.5%) | 1.000+ |
| End-organ damage | 3 (16.6%) | 5 (17.8%) | 1.000+ |
| ICU stay [day] | 7.9 ±10.9 | 15.2 ±54.9 | 0.580* |
| Hospital stay [day] | 12.5 ±13.05 | 10.7 ±8.08 | 0.574* |
| 30-day mortality | 6 (33.3%) | 6 (21.4%) | 0.493+ |
Undoubtedly, the most important parameters that could be potentially affected by cannulation strategies are postoperative mortality and rates of occurrence of new cerebral events. Regarding postoperative variables, there was no statistically significant difference in new cerebral events and early mortality. Early (30-day) mortality was 33.3% (n = 6) in the AXA group, while it was 21.4% (n = 6) in the IA group (p = 0.493). In the AXA group, 4 out of 6 patients who died were lost intraoperatively due to failure to wean from CPB. One patient who underwent total arch replacement was monitored in the intensive care unit with extracorporeal membrane oxygenation (ECMO) support but died on the 8th postoperative day due to progressive renal failure unresponsive to medical treatment. Another patient could not be weaned off the ventilator during postoperative follow-up due to Acinetobacter pneumonia and succumbed to sepsis after a prolonged postoperative course.
In the IA group, 1 patient who underwent root replacement died due to prolonged intubation followed by sepsis. Four patients, including one who underwent total arch replacement, died intraoperatively due to coagulopathy and failure to wean from CPB. In 1 patient, hemodynamic instability developed on the third postoperative day due to myocardial infarction with ST-segment elevation. The patient was treated with intra-aortic balloon pump (IABP) insertion and transferred to the coronary angiography unit, where RCA percutaneous coronary intervention was performed. Despite subsequent hemodynamic instability and the initiation of ECMO, this patient died on the 10th postoperative day.
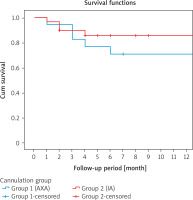
A new cerebral event (stroke or TIA) was seen in 2 (11.1%) patients in the AXA group, while it was seen in 2 (7.1%) patients in the IA group (p = 0.549). Remaining outcomes, including procedure type, intensive care unit length of stay, hospital stay, end-organ complications and need for re-exploration, are summarized in Table II. There was no significant difference in outcomes between the two groups. Kaplan-Meier survival analysis showed no different difference between the two groups at 1 year (66.7% vs. 82.8%; p = 0.558 log-rank test; Figure 6).
Figure 6
Kaplan–Meier analysis survival estimates according to cannulation site (group 1 = axillary artery, group 2 = innominate artery)
Logistic regression performed with mortality as the dependent variable on significant (p < 0.05) variables in the univariate analysis found coronary artery disease (OR = 4.364, 95% CI = 1.080–17.631, p = 0.039), aortic diameter (OR = 1.219, 95% CI = 1.029–1.444, p = 0.022), lowest body temperature (OR = 0.788, 95% CI = 0.658–0.945, p = 0.01), and using the AXA for arterial inflow (OR = 0.115, 95% CI = 0.025–0.523, p = 0.005) to be independent predictors of mortality (Table III).
Table III
Regression analysis (for all-cause mortality)
| Parameter | Odds ratio (95% CI) | P-value |
|---|---|---|
| Age | 1.008 (0.953–1.065) | 0.791 |
| Gender | 1.244 (0.329–4.708) | 0.747 |
| BSA | 0.134 (0.002–8.103) | 0.337 |
| DM | 0.842 (0.227–3.129) | 0.797 |
| HT | 1.481 (0.268–8.199) | 0.653 |
| HL | 2.369 (0.622–9.020) | 0.206 |
| CAD | 4.364 (1.080–17.631) | 0.039** |
| COPD | 0.970 (0.091–10.317) | 0.980 |
| Cardiac tamponade | 0.350 (0.085–1.441) | 0.146 |
| CPR | 6.800 (0.557–82.995) | 0.133 |
| EF | 0.983 (0.894–1.082) | 0.731 |
| GERAADA score | 1.075 (0.99–1.156) | 0.052 |
| DeBakey type | 3.385 (0.377–30.398) | 0.276 |
| Cannulation site (AXA) | 0.115 (0.025–0.523) | 0.005** |
| CPB time | 1.006 (0.993–1.020) | 0.338 |
| Cross-clamp time | 1.021 (0.995–1.047) | 0.108 |
| Lowest body temperature | 0.788 (0.658–0.945) | 0.01** |
| TCA | 0.387 (0.073–2.058) | 0.266 |
| ACP | 0.258 (0.053–1.264) | 0.095 |
| Aortic diameter | 1.219 (1.029–1.444) | 0.022** |
BSA – body surface area, DM – diabetes mellitus, HT – hypertension, HL – hyperlipidemia, CAD – coronary artery disease, COPD – chronic obstructive pulmonary disease, CPR – cardiopulmonary resuscitation, EF – ejection fraction, CPB – cardiopulmonary bypass, TCA – total circulatory arrest, ACP – antegrade cerebral perfusion.
There were no perfusion problems, injuries to the axillary vessels, or brachial plexus. One (5.6%) patient of the AXA group had temporary upper limb paresthesia that resolved completely in 6 months. One (5.6%) patient in the same group had a superficial wound infection. We had no complications related to IA cannulation site in terms of bleeding, dissection, or right arm ischemia. Only 1 (3.6%) patient in the IA group developed sternal dehiscence and underwent rewiring 3 weeks later.
Discussion
ATAAD is still one of the most fatal cardiovascular pathologies, despite advancements in treatment that have allowed surgeons to continuously reduce surgical mortality. To prevent aortic rupture or the progression of the dissection flap, reduce organ malperfusion, and ensure optimal brain protection, an appropriate cannulation technique for ATAAD must be determined [10]. However, the location and quality of the dissected arteries, the preoperative critical condition of the patient, and the surgeon’s preference are the determining factors in selection of the cannulation site. Femoral artery cannulation, which had previously been widely used, began to be abandoned after a few studies highlighted potential risks such as retrograde flow and debris embolization, malperfusion, and stroke risk [11]. Other methods have now been developed, including AXA cannulation, IA cannulation, and direct aortic cannulation employing the Seldinger technique under ultrasonographic epiaortic guidance or even a transapical route [12, 13].
Currently, ACP has been reported to be useful in reducing perioperative malperfusion in ATAAD repair [10]. In both elective and emergency cases, many surgeons favor right AXA cannulation for ACP. The positive aspects of using the AXA for inflow include preserving antegrade true lumen blood flow both before and after the cooling period and offering a source for unilateral ACP in the event of circulatory arrest. One important benefit of AXA cannulation is that it eliminates the need to manipulate the acutely dissected ascending aorta, reducing the potential for rupture and proximal or distal dissection extension [14]. In ATAAD patients, we commonly perform hemiarch replacement, which includes excision of the entire lesser curvature and almost all of the dissected aorta. We performed hemiarch replacement in 66.6% of patients in the AXA group and in 71.4% of patients in the IA group. ACP was performed in 66.6% of patients in the AXA group, while it was performed in 96.4% in the IA group (p = 0.041). AXA cases had shorter ACP time in our study cohort. In addition, in parallel with current literature, new cerebral events (stroke or TIA) were only seen in 11.1% in the AXA group, while they were seen in 7.1% in the IA group.
Despite being generally well tolerated, AXA cannulation may cause a number of technical obstacles and complications [15]. First, it necessitates a different incision. Following the cannulation, seroma development, arm ischemia, brachial plexus injuries, low cardiopulmonary flow, blood loss, and malperfusion may occur [15, 16]. In this study, there were no perfusion problems and no injuries to the axillary vessels or brachial plexus. One (5.6%) patient in the AXA group had temporary upper limb paresthesia that resolved completely in 6 months. One (5.6%) patient in the same group had a superficial wound infection. In preoperative CTA evaluation, we generally prefer AXA cannulation in cases with tears in the IA. Evaluation of the study results showed that AXA cannulation is feasible, safe, and effective for most ATAADs, supporting its continued use as a reliable cannulation option.
Another option for ACP in aortic surgery is IA cannulation. Although Di Eusanio proposed the IA as an arterial cannulation location in aortic surgery, its implementation in ATAAD has been quite uncommon [5]. Due to the proximity of the IA to the dissected aorta, surgeons have concerns regarding the safety of cannulating this region. Notably, the absence of an additional incision, ease of access in obese patients, the relative reduction in operative time, and a lower incidence of cannula-related complications during and after the procedure represent significant advantages. [17]. In the present study, no complications related to IA cannulation site – in terms of bleeding, dissection, or right arm ischemia – were observed. Previous research suggested that IA cannulation may provide better brain protection and enable the treatment of cerebral malperfusion in patients with ATAAD [18]. Additionally, in this study, there was no significant difference in CPB and cross-clamp times between groups. There was no statistically significant difference in new cerebral events, early mid-term mortality, intensive care unit length of stay, hospital stay, end-organ complications, or need for re-exploration between the two groups. Therefore, IA cannulation may be considered a safe and effective option in patients selected for ATAAD repair.
Without a doubt, the most intriguing topic is how cannulation techniques affect the results of ATAAD repair. Studies in the literature predominantly consist of reports presenting experiences from both elective and emergency operations. Meta-analyses have clearly shown that AXA cannulation is superior to femoral cannulation in terms of early mortality, neurological complications, and mid-term and long-term results [19, 20]. However, in studies comparing AXA with IA cannulation, similar neurologic and mortality outcomes were observed in the elective aortic procedures [21, 22]. Payabyab et al. reported the results of 75 ATAAD patients who underwent IA cannulation; the incidence of perioperative stroke was 9.3%, and the 30-day mortality rate was 14.7% [23]. Wong et al. described the outcomes of right AXA cannulation in 83 patients with ATAAD and reported a stroke rate of 11% and hospital mortality of 17% [24]. In the present study, there was no statistically significant difference in early mortality between groups. Neurologic outcomes were also similar between the two groups. The incidence of early mortality was 21.4% in the IA group and 33.3% in the AXA group, which is consistent with previously published reports for hemiarch repair [5, 25]. However, the reason for the higher mortality in the AXA group is that the number of cases with cardiac tamponade was higher than in the IA group. This situation may be explained by the significantly larger ascending aortic diameters in the AXA group compared to the IA group. Furthermore, we did not find any significant difference in survival between groups in the mid-term follow-up results.
Even though the findings of the current study did not show a significant difference between the two cannulation procedures, we believe that each patient ought to undergo an appropriate cannulation strategy. Secondary to AXA cannulation, IA cannulation is an acceptable alternative in cases with suitable anatomy. However, the surgeon must be careful to avoid malperfusion when the dissection flap extends to the AXA. Although distal anastomosis with ACP is definitely necessary for ATAAD repair, we propose that the cannulation sites should be used separately or in combination, contingent on the patient’s morphology and the assessment of flow adequacy.
The small sample size, lack of randomization, and retrospective nature are important limitations of this study. Since redo operations were not included, sufficiently detailed results could not be obtained. Other cannulation strategies in ATAAD repair need to be confirmed in comparative, large-scale, future randomized controlled trials.
Conclusions
The use of AXA cannulation is a reliable and safe method for arterial inflow during surgical repair of ATAAD. However, we believe that also IA cannulation with a side graft is a simple, safe, and effective technique to establish both CPB and ACP for ATAAD repair in appropriate patients. Cannulation of the IA with a side graft provides satisfactory neurologic outcomes; therefore, it is a viable option for CPB and ACP in ATAAD surgery. IA cannulation reduced complications related to the cannulation site as compared to AXA cannulation. It provided a safe alternative for ACP as regards the incidence of early mortality and other complications.