Patients with hepatocellular carcinoma (HCC) may manifest a variety of paraneoplastic syndromes, such as hypercalcaemia, hypercholesterolaemia, erythrocytosis, and hypoglycaemia [1, 2]. The incidence of these syndromes varies greatly, from 2% up to 40%, and they may be present as a single syndrome or as combinations of two, three, or even four syndromes [2, 3]. Usually, these manifestations develop late in the course of the disease, and they are generally associated with a large tumour burden and unfavourable prognosis [4]. Hypoglycaemia (serum glucose level < 60 mg/dl) is observed in 2.8–27% of HCC patients, usually in the late stages of the disease, whereas early occurrence as an initial presentation of HCC is very uncommon [5, 6]. In most cases, hypoglycaemia is severe and clinically evident with sweating, palpitations, confusion, and episodes of unconsciousness requiring intravenous administration of glucose to avoid the hypoglycaemic attacks [5, 6].
Herein, we report a case of a young man with a huge HCC associated with asymptomatic hypoglycaemia in whom selective hepatic artery chemoembolisation (TACE) of the tumour initially and subsequent tumour resection resulted in the restoration of blood glucose levels. A 47-year-old man presented with a main complaint of epigastric distention, general fatigue, loss of appetite, and body weight loss of 14 kg over the last 3 months. He denied smoking or alcohol and drug consumption, and his medical history, including liver disease history, was unremarkable. Despite weight loss, he was not malnourished; his body weight was 82 kg, and his body mass index was 25.59 kg/m2. On physical examination, the left lobe of the liver was found grossly enlarged, occupying the entire upper abdomen and extending to the umbilicus, with an irregular surface, hard consistency, and mild tenderness on palpation. The right liver was also palpable 5 cm below the right costal margin at the midclavicular line and slightly tender. The spleen was non-palpable. Spider nevi, palmar erythema, oedema of the legs, jaundice, or ascites were all absent. There were no lung rales, cardiac murmur, arrhythmia, or an abnormal bruit over the liver surface. He was admitted to our hospital for diagnostic evaluation of a presumptive tumour on the left lobe of the liver. Haematological tests disclosed microcytic, hypochromic anaemia, red cell count 5.47 × 106/mm3 (reference range: 4.5–6.5 × 106/mm3), haematocrit 34.1% (reference range: 40–54%), haemoglobin 11.2 g/dl (reference range: 13.5–17.5 g/dl), mean corpuscular volume 62.3 fl (reference range: 76–96 fl), mean corpuscular haemoglobin 20.1 pg (reference range: 27–34 pg), mean corpuscular haemoglobin concentration 31.9 g/dl (reference range: 25–32 g/dl), serum iron concentration of 13 µg/dl (reference range: 70–180 µg/dl), and serum ferritin level of 203 µg/l (reference range: 30–400 µg/l). The white blood cell count was 9060/mm3 (reference range: 3500–10,800/mm3) with 87.4% of neutrophils (reference range: 40–75%) and 5.4% of lymphocytes (reference range: 20–45%), the platelet count was 288,000/mm3 (reference range: 150,000–400,000/mm3) and the prothrombin time was 13.2 s (control 12.2 s) with an international normalised ratio (INR) of 1.13 (reference range: 0.68–1.17). Liver function tests revealed the following values: aspartate aminotransferase 124 U/l (normal range: < 35 U/l), alanine aminotransferase 66 U/l (normal range: < 45 U/l), g-glutamyl transpeptidase 85 U/l (normal range: 9–55 U/l), alkaline phosphatase 152 U/l (normal range: 30–120 U/l), total bilirubin 1.3 mg/dl (normal range: 0.3–1.2 mg/dl), direct bilirubin 0.4 mg/dl (normal range: < 0.2 mg/dl). Total serum protein levels were 6.5 g/dl (reference range: 6–8.3 g/dl) with 2.9 g/dl of albumin (reference range: 3.5–5.2 g/dl) and 3.6 g/dl of globulin (reference range: 2.5–3.1 g/dl) whereas serum concentration of C-reactive protein (CRP) was elevated at 18.35 mg/dl (normal range: < 0.5 mg/dl). Serum levels of sodium, potassium, calcium, phosphorus, and magnesium as well as blood urea nitrogen and creatinine concentrations were within their normal range, while serum total cholesterol and high-density lipoprotein cholesterol (HDL-cholesterol) levels were decreased to 95 mg/dl (normal range: 150–200 mg/dl) and 24 mg/dl (normal range: > 35 mg/dl), respectively. Fasting serum glucose concentration was decreased at 53 mg/dl (normal range: 70–105 mg/dl), whereas serum levels of insulin (7.9 mU/l) and C-peptide (691 pmol/l) were both normal as estimated by an enzyme-linked immunosorbent assay (Mercodia, Uppsala, Sweden). Serum levels of a-fetoprotein (AFP) and CA-125 were elevated at 2900 ng/ml (reference range: 0–7 ng/ml) and 175.9 U/ml (reference range: 0–35 U/ml), respectively. Carbohydrate antigen 19-9 (CA 19-9) and carcinoembryonic antigen (CEA) were within normal limits. Serum test for hepatitis B surface antigen (HBsAg), antibodies to hepatitis C virus (anti-HCV), and human immunodeficiency virus antibodies were all negative. However, antibodies to the hepatitis B virus surface (anti-HBs) and core (anti-HBc) antigens were both positive. Despite low blood glucose levels, the patient denied episodes of hypoglycaemic attacks and denied hunger sensation, cold sweating, palpitations, tremulousness, confusion, or episodes of unconsciousness when specifically asked for these symptoms. Abdominal ultrasonography (US) revealed a large mass in the left lobe of the liver with heterogeneous and hypoechogenous areas, whereas computed tomography (CT) showed a huge tumour measuring 23 cm in diameter, with central necrosis, occupying segments II, III, and IV (Figure 1). These findings were confirmed by magnetic resonance imaging (MRI) tomography (Figure 2). Satellite nodules in the liver, tumour emboli in the portal or hepatic veins, portal vein thrombosis, enlarged regional lymph nodes, or ascites were not found. The CT scan of the chest was normal. Based on these features and the elevated AFP level, the diagnosis of HCC was made. Hepatic arteriography (selective angiography of the celiac trunk) revealed a huge hypervascular tumour in the left lobe with the main tumour-feeding artery being the replaced left hepatic artery arising from the left gastric artery. After superselective catheterisation using a micro-guide wire, successful transcatheter arterial chemoembolisation was performed with an emulsion of lipiodol and doxorubicin hydrochloride along with polyvinyl alcohol particles 250–355 µm in size. Twelve hours after TACE, serum glucose level was increased to 68 mg/dl. This increase was sustained over the following days with fasting glucose levels varying between 65 and 73 mg/dl (normal range: 70–105 mg/dl) without intravenous glucose infusion. Two weeks after TACE, the patient underwent extended left hepatectomy together with resection of segment I and lymphadenectomy of lymph nodes around the porta hepatis, hepatic and common bile ducts, and hepatoduodenal ligament and along with the common hepatic artery. Resection was difficult because of the size of the tumour; in fact, the entire left lobe of the liver was replaced by the tumour. The resected specimen weighed 3.3 kg. The tumour measured 25 × 24 × 13.5 cm and contained extensive areas of necrosis, presumably not only because of its large size but also due to the preoperative TACE (Figure 3). The pathological diagnosis was moderately differentiated hepatocellular carcinoma comprising large neoplastic cells with severe nuclear pleomorphism, prominent nucleoli, frequent mitotic figures, and evident atypical mitoses. The cells were arranged predominantly in solid clusters. Adjacent non-tumorous liver parenchyma showed dilation of portal spaces, sinusoidal dilation, moderate perisinusoidal fibrosis, or peliosis. There was no evidence of vascular invasion by the tumour, and the resection margins were microscopically clear. Metastatic deposits were not found in any of the 17 resected lymph nodes. Tumour resection resulted in a further increase of serum glucose, with its level being consistently within the normal range. Fasting glucose levels recorded on serial measurements until patient discharge were over 87 mg/dl at all times in the absence of intravenous glucose administration. Tumour-associated hypoglycaemia is a common feature of insulin-producing pancreatic islet cell tumours but has also been described as a paraneoplastic syndrome in patients with malignant or benign tumours of epithelial or mesenchymal origin and, more rarely, in haematopoietic tumours, a condition also known as non-islet cell tumour hypoglycaemia (NICTH). Hypoglycaemia as a distinct paraneoplastic syndrome occurs in 2.8–27% of HCC patients and is most commonly found among Chinese patients rather than patients from Western countries. Two distinct forms of HCC-associated hypoglycaemia have been described [7]. The most common type A hypoglycaemia (87% of cases) is of mild to moderate severity and is easily controlled by increased carbohydrate intake or glucose infusion. This type of hypoglycaemia occurs usually in the terminal stage of the disease and is associated with impaired appetite, malnutrition or cachexia, hepatic failure, and poorly differentiated, rapidly growing tumours. In contrast, type B hypoglycaemia is severe, sometimes difficult to control, and does not relate to appetite loss, muscle wasting, or cachexia. It develops early and is associated with well-differentiated, slow-growing tumours. Although the pathogenesis of HCC-related hypoglycaemia has not been fully clarified, the mechanisms underlying type A and type B hypoglycaemia are probably different. Type A hypoglycaemia has been suggested to relate to increased glucose requirements by the tumour, progressively reduced hepatic glucose production due to liver failure, and to patient cachexia. Type B hypoglycaemia has been attributed to overproduction and secretion of substances with insulin-like activity such as insulin-like growth factor (IGF) I and II by the tumour. IGF-I is produced in the liver under the control of growth hormone, whereas IGF-II is produced mainly in peripheral tissues in a growth hormone-independent way. Both IGF-I and IGF-II are structurally homologous to insulin and circulate in the blood bound to IGF binding proteins such as IGF binding protein-3 and the acid labile subunit, which are also produced by the liver and exert their insulin-like activity by binding to insulin and IGF receptors. Increased tumour expression of IGF-II at both mRNA and protein level and elevated serum IGF-II levels have been documented in patients with NICTH, including HCC patients [8]. Overproduction and incomplete post-translational processing of IGF-II by tumour cells result in increased levels of a high-molecular-weight abnormal form of the IGF-II peptide called ‘big’ IGF-II. Big IGF-II is biologically active but unlikely to mature. IGF-II does not bind to circulating IGF binding proteins, thus resulting in enhanced bioavailability and increased binding to insulin and IGF receptors in the liver, muscle, and other peripheral tissues, thereby inhibiting hepatic gluconeogenesis and accelerating peripheral glucose uptake with subsequent hypoglycaemia. Big IGF-II binding to IGF-I receptors inhibits pituitary growth hormone secretion through negative feedback with subsequent reduced production of the growth hormone-dependent proteins IGF-I, IGF binding protein-3, and the acid labile subunit, thus further increasing the amounts of free IGF-II and aggravating the hypoglycaemia. Therefore, the complete biochemical profile of patients with type B non-islet cell tumour hypoglycaemia includes low serum levels of glucose, insulin, C-peptide, growth hormone, IGF-I, and IGF binding protein-3, whereas ‘big’ IGF-II levels are elevated, and by negative feedback mechanism, the production of mature IGF-II is suppressed. Sometimes, total serum IGF-II level may be within normal range or increased, but the IGF-II/IGF-I ratio is characteristically increased. However, determination of serum levels of IGF-I, IGF-II, IGF binding proteins, and especially of the ‘big’ IGF-II requires specialised assays that are not widely available, and therefore the complete profile of metabolic alterations is rarely reported in full. Our patient had a massive but not metastatic, moderately differentiated HCC and was clinically fit. He was not malnourished or cachectic despite some weight loss, and there was no evidence of liver failure, although he was positively tested for hepatitis B virus infection. The hypoglycaemia was mild and asymptomatic and was found by routine biochemistry analysis. Interestingly, decreased serum levels of IGF-I and IGF binding protein-3 have been described in patients with chronic liver disease [9], with hepatitis B virus infected patients showing increased circulating levels of incompletely processed “big” IGF-II in the absence of hypoglycaemia [10]. Unfortunately, IGF-II and “big” IGF-II levels could not be measured, and therefore, the type of hypoglycaemia could not be determined with certainty. Nevertheless, a close association between hypoglycaemia and tumour burden was apparent since selective hepatic artery chemoembolisation initially and subsequent tumour resection resulted in the restoration of blood glucose levels. In addition, serum levels of insulin and C-peptide were normal. In this context, we believe that the low serum glucose level in our patient was due to increased glucose utilisation by the huge-sized tumour. Apart from “big”, IGF-II production increased glucose utilisation by the tumour and decreased hepatic glucose production are possible factors contributing to hypoglycaemia in HCC patients. Functional tumour imaging by fluorodeoxyglucose-positron emission tomography revealed a predominant uptake of glucose by the tumour and a mild uptake by skeletal and heart muscles [6], whereas Eastman et al. in their case of a young patient who developed hypoglycaemia coincident with extrahepatic recurrence of HCC after successful liver transplantation, showed that hypoglycaemia in HCC with IGF II production is predominantly due to glucose uptake by skeletal muscle and suppression of glucose production. Optimal treatment of HCC-associated hypoglycaemia after initial management of the hypoglycaemia by intravenous glucose infusion involves radical tumour resection if possible, or by appropriate systemic treatment. Hypoglycaemia and NICTH-associated metabolic alterations are fully reversible after successful surgical removal of the tumour [8], as in our patient. Interestingly, even if complete resection is not possible, reduction of the tumour mass by percutaneous ethanol injection [6] or by intrahepatic Adriamycin infusion [11] is useful for the control of hypoglycaemia. In patients with unresectable tumours, palliative systemic chemotherapy with doxorubicin alleviated HCC-associated hypoglycaemia [12], although systemic chemotherapy with Adriamycin and cisplatin was ineffective [13]. Counter-regulatory hormones like glucocorticosteroids, and somatostatin analogues such as octreotide, glucagon, and growth hormone either alone or in combination have been used to treat hypoglycaemia in patients with inoperable tumours or recurrent hypoglycaemia with some success, although in many cases they were not effective or had only a transient effect [14]. Artificial intelligence (AI) is revolutionising the diagnosis and management of liver cancer. Recent advancements showcase AI’s ability to analyse medical imaging data, like computed tomography scans and magnetic resonance imaging, accurately detecting and classifying liver cancer lesions for early intervention. Predictive models aid prognosis estimation and recurrence pattern identification, facilitating personalised treatment planning [15, 16].
Figure 1
Contrast-enhanced CT scan of the abdomen showing a huge heterogeneous tumour occupying the left lobe of the liver. Transverse (A) and coronal (B) view
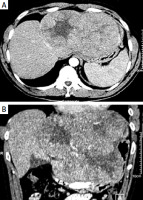
Figure 2
Magnetic resonance imaging appearance of the tumour (axial post Gd-chelate T1-weighted image, arterial phase)
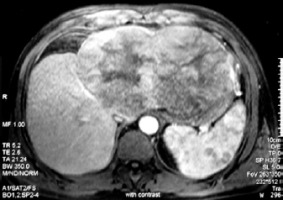
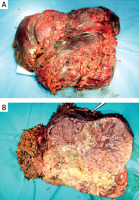
Figure 3
Gross appearance (A) and coronal plane cross section (B) of the resected specimen showing extended areas of necrosis attributable to tumour size and previous hepatic artery chemoembolization
In these cases, continuous intravenous glucose infusion is the last therapeutic measure to relieve the hypoglycaemic symptoms. In conclusion, we report a case of a young man with a huge HCC associated with asymptomatic hypoglycaemia in whom selective arterial chemoembolisation of the tumour initially and subsequent tumour resection resulted in sustained restoration of blood glucose levels, suggesting that hypoglycaemia was a consequence of increased glucose utilisation by the tumour.













