Introduction
Although contemporary clinical guidelines have contributed to lowering the incidence of deep sternal wound infection (DSWI) [1, 2], this complication continues to represent one of the most serious adverse events after coronary artery bypass grafting (CABG). Reported rates range from 0.5% to 8%, varying with patient characteristics, operative technique, and perioperative management strategies [3–8]. When DSWI occurs, it carries substantial clinical consequences, including prolonged hospitalization, impaired postoperative recovery, reduced quality of life, and a marked increase in mortality risk [3–10]. These considerations underscore the ongoing need to refine preventive strategies.
Several patient-specific characteristics, such as diabetes, elevated body mass index, older age, chronic pulmonary disease, and insulin dependence, are well-established predictors of DSWI [11]. At the same time, despite robust evidence supporting improved long-term outcomes with multiple arterial grafting, its adoption in clinical practice remains limited, typically ranging from 4% to 32% [12–20]. Concerns related to technical complexity and the perceived increased risk of sternal complications, particularly when using bilateral internal thoracic arteries (BITA), are frequently cited barriers.
In recent years, the use of topical antibiotic agents applied directly to the sternal edges has emerged as a cost-effective and straightforward method to reduce postoperative sternal wound complications. Several studies have reported encouraging results with vancomycin paste as an adjunctive prophylactic measure in cardiac surgery [21].
Aim
Given ongoing concerns about DSWI in patients undergoing BITA grafting, we sought to evaluate the effect of topical vancomycin paste on the incidence of DSWI in individuals undergoing primary isolated CABG with BITA.
Material and methods
Study design
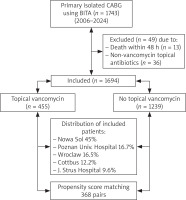
This observational, retrospective study was designed in accordance with STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines [22]. Between January 2006 and December 2024, 1,743 patients who underwent primary isolated CABG using BITA, with or without additional grafts, were analyzed retrospectively at five centers: Zbigniew Religa Heart Center “Medinet”, Nowa Sol, Poland; Zbigniew Religa Heart Center “Medinet”, Wroclaw, Poland; Sana Heart Center, Cottbus, Germany; J. Strus Hospital, Poznan, Poland; and Poznan University of Medical Sciences, Poland. Data were collected retrospectively from the surgical database at each center. This database captures detailed information on preoperative, intraoperative, and postoperative variables for all patients undergoing cardiac surgery. Patients were grouped according to whether topical vancomycin paste was applied to the sternal edge (n = 455) or not (n = 1239).
The vancomycin group received topical application of vancomycin paste, which was prepared by mixing 1 to 3 g of powder with saline until a paste formed. This paste was applied twice: once after sternotomy and again before sternal closure. Bone wax was used in both groups. Intravenous cefazolin was used as perioperative prophylaxis in most patients across all centers. In cases of documented beta-lactam allergy, vancomycin or clindamycin was administered intravenously. Patients were excluded from the final analysis if they died within 48 hours of CABG (n = 13) or if a topical antibiotic other than vancomycin was used (n = 36). Figure 1 represents a diagram of the patient flow chart.
Study endpoint and definition
The primary outcome was defined as the development of DSWI involving the muscle, bone, and/or mediastinum requiring surgical intervention and treatment with antibiotics. Surgical management of DSWI consisted of either irrigation–suction drainage (used in the early years of the study period) or negative pressure wound therapy.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation (SD), and categorical variables as counts and percentages. Between-group comparisons for continuous variables were performed using Student’s t-test, while categorical variables were compared using the Pearson χ² test.
To identify independent predictors of DSWI, a multivariable logistic regression model was constructed for the entire cohort using all preoperative and operative variables listed in Table I. Multivariable analysis was performed using stepwise backward selection, including only variables with a p-value ≤ 0.05 in univariate analysis.
Table I
Baseline and operative characteristics of the study population before and after propensity score matching
[i] Data are expressed as mean ± standard deviation or n (%). AF – atrial fibrillation, BMI – body mass index, CLD – chronic lung disease, CVAEs – cerebrovascular adverse events, EF – ejection fraction, ITA – internal thoracic artery, LM – left main coronary artery, MI – myocardial infarction, NYHA – New York Heart Association functional classification, OPCAB – off-pump coronary artery bypass, PAD – peripheral artery disease, PCI – percutaneous coronary interventions, PSM – propensity score matching, RI – renal impairment, SMD – standardized mean difference. Moderate and severe renal impairment defined as eGFR ≥ 50 and < 85 ml/min/1.73 m2 and eGFR < 50 ml/min/1.73 m2, respectively. CVAEs defined as any history of stroke or transient ischemic attack.
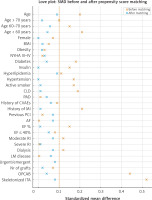
To mitigate the confounding inherent to observational data, propensity score (PS) matching was applied. Propensity scores were estimated using a multivariable logistic regression model incorporating all variables listed in Table I. Patients were matched in a 1 : 1 ratio using caliper matching without replacement, with a caliper width equal to 0.2 of the standard deviation of the logit of the PS. Covariate balance between groups was assessed using standardized mean differences (SMD), with SMD ≤ 0.1 considered indicative of adequate balance (Figure 2). Matched data were analyzed using appropriate methods for paired observations, including conditional logistic regression to estimate odds ratios (ORs) [23–28].
Figure 2
Love plot showing changes in standardized mean difference before (orange) and after (blue) matching
AF – atrial fibrillation, BMI – body mass index, CLD – chronic lung disease, CVAEs – cerebrovascular adverse events, EF – ejection fraction, ITA – internal thoracic artery, LM – left main coronary artery, MI – myocardial infarction, NYHA – New York Heart Association functional classification, PAD – peripheral artery disease, PCI – percutaneous coronary interventions, PSM – propensity score matching, RI – renal impairment, SMD – standardized mean difference.
A sensitivity analysis using the E-value was performed to evaluate the potential impact of unmeasured confounding. The E-value quantifies the minimum strength of association that an unmeasured confounder would need to have with both the exposure and the outcome, beyond the measured covariates, to explain away the observed association fully. The E-value was calculated for the primary endpoint and interpreted relative to the magnitude of known predictors of DSWI [29–32].
All statistical analyses were conducted using R (R Core Team, 2021. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL: https://www.R-project.org/). Propensity score matching was performed using the MatchIt and optmatch packages.
Results
Between January 2006 and December 2024, a total of 1,694 patients met the inclusion criteria and were stratified according to whether topical vancomycin paste was applied to the sternal edges (n = 455) or not (n = 1,239). Before matching, the two groups were comparable with respect to sex, body mass index, history of cerebrovascular events, left main coronary artery disease, previous percutaneous coronary intervention, peripheral vascular disease, heart failure symptoms, and atrial fibrillation. However, patients who received topical vancomycin paste were more likely to be older, diabetic, on insulin therapy, have a history of myocardial infarction, undergo off-pump CABG, and receive skeletonized internal thoracic arteries. Conversely, patients who did not receive topical vancomycin paste were more frequently active smokers and more commonly had chronic lung disease and renal impairment. Baseline and operative characteristics are summarized in Table I.
In the entire cohort, multivariable logistic regression analysis demonstrated that the use of topical vancomycin paste was independently associated with a 63% reduction in the incidence of DSWI (OR = 0.37; 95% CI: 0.14–0.97; p = 0.045). Skeletonization of the internal thoracic arteries was also independently associated with a 62% reduction in DSWI incidence (OR = 0.38; 95% CI: 0.21–0.69; p = 0.0015). Other independent predictors associated with an increased risk of DSWI included age ≥ 60 years, obesity, chronic lung disease, insulin therapy, female sex, and heart failure symptoms (Table II).
Table II
Multivariable logistic regression analysis of independent predictors of deep sternal wound infection in the entire cohort
Propensity score matching yielded 368 matched pairs for the final comparison. The two matched groups were well balanced, with all standardized mean differences < 0.1 (Figure 2). In the matched cohort, the use of topical vancomycin paste was associated with an 83% reduction in the incidence of DSWI (conditional OR = 0.17; 95% CI: 0.05–0.60; p = 0.001).
Sensitivity analysis
The E-value for the association between topical vancomycin paste and reduced DSWI in the matched cohort was 11.2. In comparison, the odds ratios of the measured risk factors ranged from 1.88 to 4.19 (Table II). Because these observed associations are substantially weaker than the E-value, an unmeasured confounder would need to exert a much stronger effect than any known risk factor to fully account for the observed treatment effect. This suggests that the findings are robust and unlikely to be explained by residual confounding.
Discussion
In this large cohort of patients undergoing primary isolated CABG with BITA, the use of topical vancomycin paste was associated with a marked reduction in DSWI. The consistency of the effect – demonstrated both in the overall population and after propensity-score matching – supports the value of local antibiotic prophylaxis as a simple and effective adjunct to conventional perioperative measures.
Patients undergoing BITA grafting constitute a group at intrinsically elevated risk for sternal complications. In addition to well-recognized patient-related factors such as diabetes, obesity, advanced age, female sex, pulmonary disease, renal impairment, and postoperative re-exploration, BITA harvesting itself substantially contributes to DSWI risk [33–36]. Importantly, the clinical implications of DSWI extend far beyond the immediate postoperative period. Recent evidence from the Polish National Registry (KROK registry) demonstrated that DSWI independently predicts increased late mortality after CABG, irrespective of the revascularization strategy employed [36]. Earlier work by Gaudino et al. similarly showed that postoperative mediastinitis is associated with a sustained excess mortality risk up to a decade after surgery [33]. These findings emphasize that preventing DSWI is not merely an effort to avoid early morbidity but a determinant of long-term survival.
The observed benefit of topical vancomycin paste aligns with the microbiology of post-sternotomy mediastinitis. Staphylococcus aureus and coagulase-negative staphylococci account for most DSWI events [9, 37, 38]. Local antibiotic application achieves high tissue concentrations at the sternal edges, surpassing levels obtainable through systemic prophylaxis alone, particularly during the period of impaired sternal perfusion following BITA harvesting. This targeted exposure likely contributes to the substantial risk reduction demonstrated in our analysis.
The BITA harvesting technique is another important determinant of DSWI. Skeletonization of the internal thoracic arteries improves preservation of sternal blood flow and has repeatedly been associated with fewer sternal complications [39]. Our findings corroborate the protective effect of skeletonization. Nonetheless, the technique requires additional operative skill and has historically raised concerns about graft integrity and long-term patency [40, 41]. For these reasons, many surgeons continue to rely on pedicled BITA grafts, accepting an inherently higher sternal risk. The present study suggests that topical vancomycin paste may provide an important adjunctive measure in such cases, offering risk mitigation without mandating changes in surgical technique. This could facilitate broader and safer adoption of BITA revascularization and, by extension, multiple arterial grafting (MAG).
This is particularly relevant given the ongoing underuse of MAG despite compelling evidence of its long-term survival benefit [12–20]. Surveys from Canada, Europe, and North America consistently identify concern over DSWI as one of the leading reasons surgeons avoid BITA grafting, especially in higher-risk patients such as those with diabetes or obesity [42, 43]. By demonstrating a meaningful reduction in DSWI with a simple, inexpensive intervention, our findings directly address one of the most persistent barriers to wider MAG utilization.
The biological rationale for this protective effect further strengthens the argument for broader adoption. Following BITA harvest, especially when the pedicled technique is used, sternal perfusion can be significantly diminished. Local vancomycin administration ensures that bactericidal antibiotic levels are present precisely during this vulnerable interval. When combined with surgical techniques that preserve chest wall vascularity, such as skeletonization, this approach provides complementary pathways to reduce sternal complications.
Several limitations of this study merit acknowledgement. As a retrospective analysis, unmeasured confounding cannot be fully excluded despite extensive adjustment, propensity matching, and sensitivity analyses. Variations across centers in perioperative practices, such as glycemic control, wound surveillance protocols, and antibiotic stewardship, may have influenced outcomes. The decision to use topical vancomycin was not randomized and may reflect surgeon preference or perceived patient risk. Furthermore, the BITA harvesting technique was not standardized across surgeons, and technical nuances that affect sternal perfusion could not be captured. Microbiological data were incomplete for all DSWI cases, limiting assessment of pathogen-specific treatment effects. Finally, graft patency, an essential aspect of BITA outcomes, was not evaluated. The effectiveness of other topical antibiotics or alternative delivery systems was also beyond the scope of this analysis.
Conclusions
The present study provides robust multicenter evidence supporting the use of topical vancomycin paste as an effective adjunct for reducing deep sternal wound infection in patients undergoing coronary artery bypass grafting with bilateral internal thoracic arteries. By improving the safety profile of bilateral internal thoracic artery grafting, this strategy may help remove a significant barrier to broader adoption of multiple arterial grafting and ultimately contribute to improved long-term patient outcomes.