Introduction
Surgical aortic valve replacement remains the mainstay of treatment for aortic valvular disease in a large population of patients. Conventional stented, rapid deployment and new-generation stented biological valves are now available for surgical aortic valve replacement. New generation devices are characterized by advanced tissue treatment for theoretical prolonged durability and new stent design able to expand in case of future transcatheter valve-in-valve procedures, in a lifelong perspective treatment. On the other hand, in recent years, the micro-invasive cardiac procedure, i.e., transcatheter aortic valve replacement, has undergone significant technological progress with a reduction in peri-procedural complications, with the aim to be able to treat effectively also patients over 75 years of age and affected by more concomitant pathologies, i.e., chronic pulmonary disease, obesity, and in the presence of a greater incidence of worse general conditions due to frailty or multi-organ pathology. Perceval sutureless bioprosthesis (Corcym Inc., Milan, Italy) have been developed to combine the advantage of a rapid deployment device with that of the traditional surgical approach, thus reducing extracorporeal circulation and aortic clamping times, and, consequently, the potential risk of postoperative complications. Introduced by 2007, the Perceval prosthesis is a pericardial surgical heart valve with a sutureless and collapsible design that simplifies the surgical implantation. This prosthesis has shown a stable hemodynamic performance with satisfactory postoperative gradients as well as early and long-term follow-up results. Moreover, the Perceval may enable lower peak and mean pressure gradients than conventional biological prostheses, a larger effective orifice area, and therefore lower risk of patient-prosthesis mismatch, which is one of the main predictors of structural deterioration during follow-up. Compared with sutured bioprostheses, the Perceval valve has the disadvantages of selective implantation criteria, which are not applicable to all patients, i.e., with respect to the annular and the sino-tubular junction diameters [1–3]. In the retrospective single-center study started in the year 2022 and previously published (the Perfecta – Perceval vs. Trifecta [St. Jude Medical, Abbot Vascular, previously St. Jude Medical, Inc., St. Paul MN, USA] – study), we evaluated whether the implantation of the Perceval prosthesis could shorten surgical times, allowing for better in-hospital outcomes, especially in patients with comorbidities [4].
Aim
In light of recent data published in the literature, which have highlighted a greater risk of structural valve deterioration (SVD) of the Trifecta prosthesis and, consequently, worse late outcomes which led to the withdrawal of the prosthesis from the market, and since we have implanted the Trifecta prosthesis for a long period of surgical activity, we wanted to carefully evaluate the hemodynamic performance and the long-term results of this prosthesis by comparing them with those obtained with the Perceval prosthesis implanted in the same period of surgical activity. In particular, to accurately assess long-term outcomes, we focused on comparing the following end-points: 1) late survival; 2) event-free survival rates; 3) freedom from SVD and its impact on redo operation risk; 4) the incidence and impact of patient-prosthesis mismatch (PPM) on long-term outcomes.
Material and methods
From December 2014 to June 2023, at the Cardiac Surgery Division of the Tor Vergata University Polyclinic, we collected data from 280 consecutive patients affected by aortic valvular disease with predominantly severe stenosis, who underwent surgical aortic valve replacement (SAVR) with implantation of the third-generation Trifecta stented biological aortic valve prosthesis and the Perceval sutureless biological aortic valve prosthesis.
In comparison with the sutured bioprosthesis, for implantation of the Perceval prosthesis we followed the implantation criteria recommended by the company: that is, age over 75 years (even if we subsequently extended the indication to patients up to 70 years of age), specific aortic annulus dimension, i.e., between 19 and 27 mm and a healthy aortic root anatomy, with the exclusion of the ratio of the sinus of Valsalva to the superior annulus greater than 1.3, in order to avoid incorrect positioning.
The Perfecta study was approved by the Institutional Review Board – Ethics Committee of the Tor Vergata Polyclinic (study No. 107/22, date of approval June 14, 2022).
All the variables analyzed in the Perfecta study and the criteria for defining the preoperative and intraoperative clinical variables have been reported in the previously published manuscript [4] and in the reference studies [5, 6]. In the present study, both preoperative clinical and intraoperative variables, as well as echocardiographic variables, were analyzed to evaluate the follow-up outcomes and hemodynamic performance.
In particular, for both groups of patients, the clinical variables analyzed were age, gender, the Euro-SCORE II evaluation system for operative risk (European System for Cardiac Operative Risk Evaluation), NYHA class, cardiovascular risk factors, i.e., smoking, diabetes mellitus, dyslipidemia, hypertension, presence and severity of concomitant coronary artery disease (CAD), and left main stem disease (LMSD). Concomitant pathologies analyzed were chronic obstructive pulmonary disease, arterial peripheral vascular disease, and obesity. Ultrasound variables examined at discharge from hospital and during follow-up were the peak and mean transprosthetic valve gradients, the left ventricular ejection fraction, and the degree of patient-prosthesis mismatch (PPM), defined as mild-to-moderate when indexed effective orifice area (iEOA) was greater than 0.80 cm2/m2 and lower than ≤ 0.85 cm2/m2, and moderate in presence of iEOA > 0.65 cm2/m2 and ≤ 0.80 cm2/m2 [7].
Definitions and data analysis during follow-up
The follow-up was performed by clinical evaluation and transthoracic echocardiograms, scheduled at 6 months, 12 months, and then every 12 months. When direct contact with patients was not possible, clinical information was collected by telephone interview with family members or by physicians. Adverse events were classified according to the standardized definitions established by the European Societies ESC and EACTS Guidelines for the management of heart valvular disease [8].
Specifically, in the actuarial survival estimate, we evaluated long-term mortality for all causes, including freedom from cardiac mortality, sudden death, and mortality due to heart failure or myocardial infarction. Structural valve deterioration (or degeneration) (SVD) was assessed by ultrasound according to the standardized definition for surgical and transcatheter bioprosthetic aortic valves [9]: initial SVD was defined as stages 1 and 2S, and advanced or moderate-to-severe SVD was defined as stages 2SR and 3. We defined PPM as 1 for mild-to-moderate grade and 2 for moderate grade.
Statistical analysis
Statistical analysis was performed using StatView 4.5 (SAS Institute Inc., Abacus Concepts, Berkeley, CA). Contingency tables with raw data were analyzed using χ2, G-squared, and Fisher’s exact tests for categorical variables and the unpaired Student’s t-test for continuous variables to compare the two groups of patients receiving Perceval or Trifecta prostheses. Actuarial survival, freedom from cardiac death, freedom from prosthetic valve-related events, i.e., redo operation due to SVD, endocarditis and redo operation due to endocarditis, thromboembolism, and cumulative event-free survival, were expressed as mean values plus or minus one standard deviation, and computed using the Kaplan-Meier method. The Mantel-Cox log-rank test was used to compare survival estimates among subgroups, i.e., SAVR for isolated aortic disease versus SAVR plus coronary artery bypass grafting (CABG), as well as according to the presence and severity of PPM. The Cox proportional hazards method was used to evaluate the influence of variables on time to death and to adverse events. Those variables with a p-value less than or equal to 0.1 in the Cox linear analysis were then entered into the regression model to identify risk factors predictive of events over time. All other values were expressed as mean plus or minus one standard deviation of the mean. The calculated echocardiographic parameters at the follow-up were compared with those at discharge from hospital. P-values less than 0.05 were considered statistically significant.
Results
Two hundred and eighty patients (mean age: 75.8 ±6.4 years, 151 males, 54%, 129 females, 46%) underwent SAVR: the St. Jude Trifecta sutured bioprosthesis was implanted in 220 (78.6%) patients, the Perceval sutureless bioprosthesis in 60 (21.4%) patients. Associated CABG was performed in 100 (35.7%) patients. Baseline and preoperative characteristics are reported in Table I.
Table I
Clinical preoperative characteristics of patients undergoing aortic valve replacement with Perceval sutureless or St. Jude Trifecta bioprostheses
Brief summary of in-hospital results
Associated CABG to SAVR was performed in 73 (33%) patients in the Trifecta group, and in 27 (45%) patients in the Perceval group (p = 0.11). Extracorporeal circulation and cross-clamp times were significantly longer in the Trifecta group in all SAVR (96 ±36 and 67 ±21 minutes vs. 61 ±23 and 49 ±18 minutes) and in isolated SAVR (84 ±28 and 66 ±21 minutes vs. 54 ±10 and 43 ±8 minutes) (p < 0.001). Operative mortality was 2.7% (n = 6) in the Trifecta group vs. 0% in the Perceval group (p = 0.02), stroke 0.9% (n = 2) vs. 1.6% (n = 1) (p > 0.9), permanent pacemaker implantation 4.5% (n = 10) vs. 4.9% (n = 3) (p = 0.4). Postoperative complications were significantly higher in Trifecta patients (18.6% vs. 6.6%) (p = 0.01). Independent predictors of in-hospital death and postoperative complications were concomitant presence of preoperative multi-vessel coronary artery disease and chronic obstructive pulmonary disease, rather than implantation of the Trifecta prosthesis itself [4]. Echocardiography at discharge showed similar peak and mean trans-aortic valve gradients of the two studied devices overall (21.6 ±7.3 and 11.6 ±4.3 mm Hg in the Trifecta group vs. 22.6 ±7.9 and 12.6 ±4.8 mm Hg in the Perceval group; p > 0.1) [4].
Results of present study
Two hundred and seventy-four survivors, i.e., 214 patients who received St. Jude Trifecta implants and 60 patients who received Perceval implants, were analyzed. Sizes of prostheses implanted and analyzed during follow-up of the Trifecta valve were 19 in 61 patients (28.5%), 21 in 87 (40.7%), 23 in 49 (22.9%), 25 in 17 (7.9%); and those of the Perceval valve were S (small) in 16 (26.6%), M (medium) in 34 (56.6%), L (large) in 8 (13.3%), and XL in 2 (3.3%) patients. At discharge, moderate PPM was more frequently observed in the Trifecta group in comparison with the Perceval group (17.3%, [n = 38] vs. 8.2% [n = 5]; p = 0.004). In relation to the prosthesis sizes, the incidence of PPM 2 was as follows: Trifecta 19: 22 out of 61 patients (36.1%); Trifecta 21: 21 out of 88 patients (17%); Trifecta 23: 1 case out of 52 (1.9%); Trifecta 25: absent (0/19); Perceval S: 3 out of 16 patients (18.7%); Perceval M: 2 out of 34 (5.9%), Perceval sizes L-XL: absent (0/10) (p < 0.001).
Survival rates
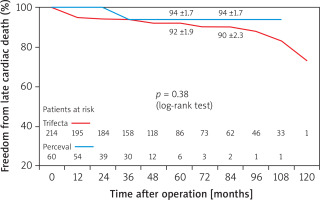
Mean follow-up was 58.9 ±34.1 and 43.5 ±21.1 months after Trifecta and Perceval valve implantations, respectively. Follow-up was 96% complete; 12 patients (11 in Trifecta group, 1 in Perceval group) were lost or did not complete the follow-up. At 7 years, actuarial survival was 68 ±4.1% in the Trifecta group vs. 89 ±4.8% in the Perceval group (p = 0.19) (Figure 1), and freedom from late cardiac death was 90 ±2.3% vs. 94 ±1.7% (p = 0.38) (Figure 2). There were 67 late deaths (24.4%) out of 274 patients who survived after SAVR; 23 of them (34%) were due to cardiac causes. Echocardiographic data collected during follow-up for the evaluation of transprosthetic gradients and SVD were complete in 180 patients.
Figure 1
Survival in surgical aortic valve replacement (SAVR) patients receiving Perceval sutureless and Trifecta bioprosthesis implants
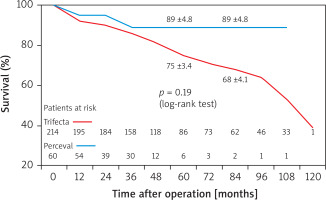
Figure 2
Freedom from late cardiac death in SAVR patients receiving Perceval and Trifecta bioprosthesis implants
At the Cox regression analysis, independent predictors of all-causes late death were preoperative chronic pulmonary disease (HR = 2.3; p = 0.05) and advanced age (HR = 1.2; p = 0.06) (Table II). Concomitant presence of three-vessel CAD and/or LMSD was detected as a risk factor in the Cox linear analysis for late survival, and as an independent predictor in the regression analysis of late cardiac death (HR = 2.9; p = 0.04) (Table III). At 7 years, in patients requiring CABG associated with SAVR affected by three-vessel CAD and/or LMSD in comparison with the other patients, the rank-test analyses showed significantly worse survival (47 ±13% vs. 68 ±8.0%, p = 0.009) and reduced freedom from cardiac death (78 ±9.0% vs. 92 ±2.3%, p = 0.04).
Table II
Independent predictors of late survival (Cox model)
Table III
Independent predictors of late cardiac death (Cox model)
| Cox linear analysis | Regression | |||
|---|---|---|---|---|
| Variables | P-value | Hazard ratio | 95% CI | P-value |
| Three-vessel CAD and/or LMSD | 0.01 | 2.9 | 0.97–8.61 | 0.04 |
| Chronic pulmonary disease | 0.11 | 2.1 | 0.69–7.23 | 0.18 |
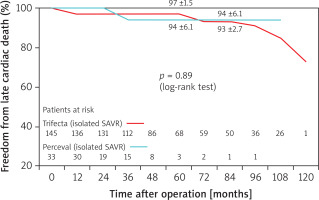
Considering the patient population undergoing only isolated SAVR, survival and freedom from late cardiac death were found to be comparable for the two types of prostheses, i.e., Trifecta 145 patients, Perceval 33 patients (Figures 3 and 4).
Figure 3
Survival in patients undergoing isolated SAVR with Perceval and Trifecta bioprosthesis implants
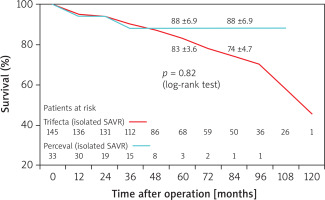
Figure 4
Freedom from cardiac death in patients undergoing isolated SAVR with Perceval and Trifecta bioprosthesis implants
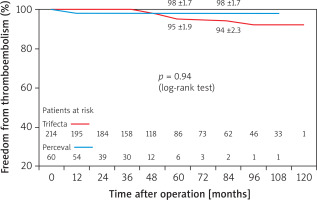
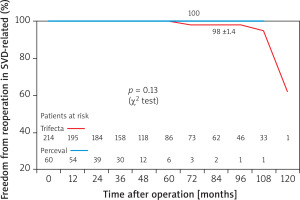
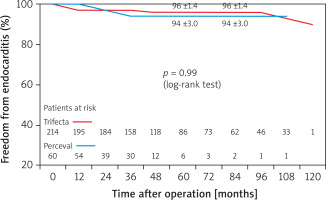
Freedom from endocarditis and redo operation due to endocarditis (94 ±3.0% vs. 96 ±4.0%) (Figure 5), from thromboembolism (94 ±2.3% vs. 98 ±1.7%) (Figure 6), and from redo operation due to SVD (98 ±1.4% vs. 100%) (Figure 7) were similar in both groups (Trifecta vs. Perceval) of patients. There was a non-significant tendency for freedom from redo operation in favor of the Perceval prosthesis (no patients re-operated in the Perceval group, 8 patients [3.7%] in the Trifecta group [surgical redo operations = 2, valve-in valve procedures = 6]) (p = 0.13).
Figure 5
Freedom from endocarditis in SAVR patients receiving Perceval and Trifecta bioprosthesis implants
Freedom from SVD and impact of PPM on SVD
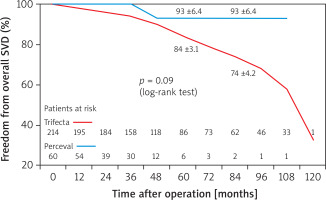
At 7 years, freedom from overall SVD, including all stages of prosthesis deterioration, was 74 ±4.2% in the Trifecta group vs. 93 ±6.4% in the Perceval group (Figure 8), with a non-significant tendency for a greater freedom from events in favor of the Perceval prosthesis (p = 0.09).
Figure 8
Freedom from overall (all stages) SVD in SAVR patients receiving Perceval and Trifecta bioprosthesis implants
Overall SVD was observed in 61 patients (1 in the Perceval group, 60 in the Trifecta group); initial SVD was observed in 53 out of 61 patients (1 Perceval, 52 Trifecta).
The incidence of SVD according to the Trifecta valve prosthesis size was as follows: n = 19, 23 out of 61 patients (37.7%), n = 21, 20 out of 87 (22.9%), n = 23, 12 out of 49 (24.5%), n = 25, 5 out of 17 (29.4%). In the Perceval group, SVD was observed in only 1 patient with size M.
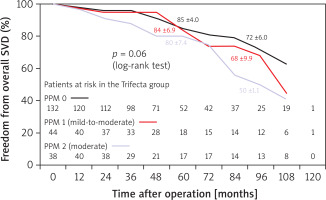
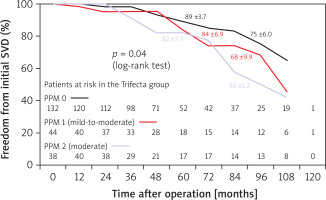
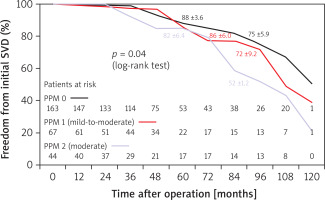
In the Cox model analysis, independent predictors of SVD were the Trifecta prosthesis itself (HR = 22.4; p = 0.002) and moderate grade PPM, i.e., PPM 2 (HR = 2.3; p = 0.03) (Table IV). The Trifecta valve (HR = 20.5; p = 0.003) and moderate grade PPM (HR = 2.5; p = 0.02) were also independent predictors of early stages, i.e., 1 and 2S, of SVD (Table V). Moderate PPM showed in the medium and long-term follow-up a negative impact on the freedom from overall SVD and from its initial stages also in the Mantel-Cox test (Figures 9, 10). Given the higher incidence of SVD in the Trifecta group, we reported overall and initial freedom from SVD analyzed only in this patient group (Figures 11, 12).
Table IV
Independent predictors of overall SVD (stages 1-2S, 2SR-3) (Cox model)
| Cox linear analysis | Regression | |||
|---|---|---|---|---|
| Variables | P-value | Hazard ratio | 95% CI | P-value |
| Trifecta prosthesis | < 0.0001 | 22.4 | 3.01–166 | 0.002 |
| PPM 2 | 0.02 | 2.3 | 1.06–4.90 | 0.03 |
| PPM 1 | 0.13 | 1.6 | 0.89–4.14 | 0.23 |
Table V
Independent predictors of initial stages (1-2S) of SVD (Cox model)
Figure 9
Freedom from overall SVD in SAVR patients receiving Perceval and Trifecta bioprosthesis implants stratified by patientprosthesis mismatch (PPM) presence and degree (mild-to-moderate [PPM 1] and moderate [PPM 2])
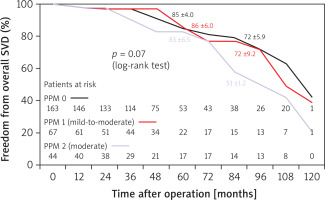
Figure 10
Freedom from initial (stages 1-2S) SVD in SAVR patients receiving Perceval and Trifecta bioprosthesis implants stratified by PPM presence and degree (absent [PPM 0], mild-to-moderate [PPM 1] and moderate [PPM 2])
Cumulative event-free survival
Cumulative event-free survival, including all-cause death, freedom from endocarditis, from thromboembolism, from redo operation and from overall SVD, was 55 ±4.2% in the Trifecta group vs. 55 ±2.3% in the Perceval group, and there were no statistically significant differences between the two types of prosthesis either in the Mantel-Cox test or in the Cox model regression.
Only the χ2 test analysis highlighted a higher and statistically significant incidence of adverse events in the Trifecta group (103 out of 214 [48%] vs. 7 out of 60 [11.7%], p < 0.001), mainly due to the greater number of patients who presented the different degrees of SVD mentioned above.
Ultrasound variables
In patients undergoing Perceval implantation, was observed a lower average value of transprosthetic peak gradient in comparison with the Trifecta valve (Table VI). Also, the mean value of the maximum transprosthetic gradient of the Perceval size S valves was significantly lower than that calculated for the Trifecta size 19 valves (Table VII).
Table VI
Ultrasound variables of the two biological prostheses at discharge from hospital and during follow-up (not including patients with severe SVD)
| Parameter | Discharge from hospital | Follow-up | ||
|---|---|---|---|---|
| Perceval (all patients) | Trifecta (all patients) | Perceval (all patients) | Trifecta (all patients) | |
| Transprosthetic peak gradient [mm Hg] (mean ± SD) | 22.6 ±7.9 | 21.6 ±7.3 | 17.8 ±4.3* | 22.8 ±8.3* |
| Transprosthetic mean gradient [mm Hg] (mean ± SD) | 12.6 ±4.8 | 11.6 ±4.3 | 12.0 ±3.9 | 12.9 ±4.9 |
| LVEF, % (mean ± SD) | 58.7 ±4.9 | 56.9 ±6.8 | 58.4 ±4.9 | 56.5 ±6.9 |
Table VII
Transprosthetic peak and mean gradient ultrasound measurements based on the caliber of the two biological prostheses at discharge from hospital and during follow-up (not including patients with severe SVD)
| Variable | Peak gradients | Mean gradients | ||
|---|---|---|---|---|
| Discharge from hospital | Follow-up | Discharge from hospital | Follow-up | |
| Trifecta size 19 | 23.2 ±7.6 | 26.0 ±7.7* | 12.8 ±4.5 | 14.9 ±4.3 |
| Trifecta size 21 | 21.6 ±7.4 | 21.8 ±7.8 | 11.7 ±4.5 | 12.4 ±4.8 |
| Trifecta size 23 | 19.7 ±6.7 | 19.8 ±8.4 | 10.7 ±3.8 | 11.6 ±5.5 |
| Trifecta size 25 | 16.3 ±6.5 | 19.9 ±7.5 | 8.5 ±3.6‡ | 11.8 ±4.5‡ |
| Perceval size S | 24.8 ±9.2 | 17.0 ±1.4* | 15.0 ±6.4 | 13.3 ±5.5 |
| Perceval size M | 22.5 ±7.4 | 18.6 ±4.8 | 12.0 ±4.1 | 10.6 ±2.6 |
| Perceval size L | 17.8 ±6.9 | 20.0 ±2.8 | 10.0 ±3.5 | 11.0 ±0.7 |
NYHA class
NYHA class significantly improved in comparison with the preoperative status both in the Trifecta group (NYHA class I–II in 79.6% [168 out of 211 analyzed patients] vs. 46% [101 out of 220 patients]; p < 0.001) and in the Perceval group (NYHA class I–II in 98.3% [58 out of 59] vs. 40% [24 out of 60]; p < 0.001).
Discussion
Trifecta and Perceval prostheses have shown equivalent early clinical results and hemodynamic performance when compared with each other [10–20]. The main problem observed with implantation of the Trifecta prosthesis has been highlighted in terms of SVD and increased risk of reoperation, so much so that, at the end of 2022, the manufacturing company Abbot has started the withdrawal of the Trifecta valves. Furthermore, in order to treat the Trifecta prosthetic dysfunction with the percutaneous valve-in-valve technique, the risk of occlusion of the coronary ostia is quite high. For these reasons, since we have implanted a rather large number of Trifecta prostheses over a period of 5 years (December 2014 to July 2019), we have evaluated the clinical and echocardiographic results during the follow-up. Since in the same period we have also frequently implanted the Perceval prosthesis, we wanted to compare the results of this prosthesis with those of the Trifecta. Obviously, the number of patients with the Perceval implant was lower, because its implantation requires compliance with the established anatomical criteria regarding the aortic root and annulus dimensions.
The main findings of our study were as follows: 1) similar survival and event-free survival rates for both types of prostheses; 2) higher incidence of SVD after implantation of the Trifecta prosthesis not associated, at least in the medium-to-long term, with a higher incidence of redo operation; 3) higher incidence of moderate-grade PPM in the Trifecta group.
In our study, survival and freedom from late cardiac mortality and major adverse events were equally satisfactory and comparable with single or multicenter studies recently reported in the literature in patient populations with a mean age similar to our patients, for both the Trifecta prosthesis and the Perceval prosthesis [21–26]. These results were also found to be equally comparable in the two groups of patients we analyzed, both as an entire population and as isolated SAVR interventions. Specifically, in our analysis, as previously reported by us in 177 Trifecta implantations [27], concomitant severe CAD was shown even in this larger patient population to be a predictor of reduced event-free survival.
Structural valve deterioration
Compared to internally mounted bioprostheses with supporting stents, the Trifecta externally mounted prosthesis exhibits superior early hemodynamic performance, but it has been shown that it presents an unexpected rapid deterioration in the medium term. This may derive from two physio-pathological mechanisms that occur for the leaflets of the prosthesis mounted outside the supporting stents. The first determines, especially with the systolic movement, the tears of the leaflets themselves leading to relevant aortic insufficiency and advanced degrees of SVD, i.e., stages 2S-R and 3. The other is related to progressive inflammatory phenomena causing thickening and calcification of the cusps already early, leading to the initial stages of deterioration in which a high degree of aortic stenosis prevails due to reduction of the EOA of the prosthesis. Yokoyama et al., who analyzed 15 observational studies that enrolled 23,539 patients undergoing SAVR with externally mounted prostheses (Trifecta valves = 6146, Mitroflow valves = 3192) and internally mounted Perimount prostheses (n = 14,201), found higher reoperation rates for SVD in the Trifecta and Mitroflow groups (p < 0.001) [28]. Fukuhara et al., comparing a Trifecta group (n = 508) with a non-Trifecta bioprosthesis group (n = 550), reported an approximately 4-fold higher incidence of SVD in the former (13.3% vs. 4.4%; p = 0.01), and even higher for patients under 65 years of age (27.9% vs. 6.9%; p = 0.004), with a notably elevated adverse event rate after 5 years of follow-up [29]. Werner reported at 5 years an incidence of SVD of 4.49% for the Trifecta and 1.04% for the Intuity prostheses, which increased further for the Trifecta valve at 7 years up to 23.78% [22]. Similar to other reported findings, in our study we observed an overall incidence of SVD in 61 Trifecta valves (28.5%) vs. 1 Perceval valve (1.7%), and an incidence of initial SVD in 52 Trifecta valves (24.3%) vs. 1 case (1.7%). In particular, as shown in Figure 8, the lowering of the SVD-free survival curve became evident after 48 months of follow-up, with a further marked decline after 7 years.
However, at least from what was observed in our case series, the high incidence of SVD detected by ultrasound examination did not translate into a high clinical risk of reoperation. This aspect highlighted in our study may depend, at least in part, on the fact that the patients after surgery were carefully followed both clinically and with echocardiographic examinations at our valvular heart disease clinic. Considering the age at surgery and the 5–9-year increase in patients’ age at follow-up, targeted medical therapy often improved clinical symptom control. Furthermore, as observed with echocardiography, the higher incidence of SVD in the Trifecta group compared to the Perceval group was related to its early stages, which do not necessarily require reoperation but can be monitored and treated with medical therapy alone. Indeed, the NYHA functional class was found to be generally satisfactory, even in the Trifecta group. The possibility of being closely monitored in the outpatient clinic allowed for the careful selection of cases for which a redo or valve-in-valve procedure was strictly necessary.
Patient-prosthesis mismatch
In patients undergoing SAVR, especially with a biological prosthesis, the incidence of PPM may not be negligible. Its main consequence is to determine high transprosthetic gradients through a normally functioning valve [7]. The negative impact of PPM on all-cause mortality and late cardiac death, as well as on freedom from SVD, has been reported in several studies. The incidence of moderate PPM ranges from 20% to 70%, whereas severe PPM occurs more rarely, its incidence ranging from 2% to 10%. Both types of prostheses have been shown to be effective in reducing the incidence of moderate and severe PPM. Hernandez-Vaquero et al., analyzing 516 SAVR with Trifecta and Perceval, reported an incidence of moderate PPM of 8.6% and 21.4%, and 2.1% and 3.8%, respectively [30]. Wakami et al. in 110 Trifecta implantations reported a significantly higher incidence of 7-year SVD in patients affected by moderate PPM, i.e., 12.6% vs. 2.8% (p = 0.005) [31]. In our study, by dividing a milder grade of PPM, i.e., 0.85 < iEOA > 0.80 cm2/m2 vs. a moderate grade, i.e., iEOA value less than 0.80 cm2/m2, we observed that during follow-up a lower freedom from total SVD and from initial stages of SVD was evident after 72 months of follow-up (Figures 9, 10). In particular, in the Cox model analysis, moderate PPM in association with the Trifecta prosthesis implantation itself represented an independent predictive factor of SVD (Tables IV, V), with markedly higher hazard ratios for the Trifecta prosthesis itself, i.e., > 20 compared to those measured for the PPM, i.e., > 2. The two predictive factors were probably related to each other, since moderate PPM, i.e., PPM 2, as reported above, was found more frequently in patients with Trifecta prostheses (17.3%) than in those with Perceval prostheses (8.2%), and was found more frequently in the Trifecta prosthesis sizes 19 (36.1%) and 21 (17.1%) in comparison with the Perceval size S (18.7%). Therefore, the higher incidence of PPM 2 in the Trifecta group could explain, at least in part, the higher risk of late SVD observed during the follow-up, although, as highlighted in the figures, it appears more evident that the moderate PPM compared to the Trifecta prosthesis itself had a lesser and later influence on the reduced freedom from SVD.
Strengths and limitations of the study
The primary interest of the study is that we compared two types of latest generation prostheses, Perceval vs.Trifecta, to evaluate their late outcomes, and we observed that with them, the major late outcomes appear to be satisfactory. The main limitations of the study are related to its retrospective observational nature; furthermore, during the follow-up, given its length, from the interviews with the family members some data could have been lost or not analyzed correctly, underestimating the real incidence of some prosthesis-related adverse events or need for reoperation. In any case, we tried to carefully analyze the clinical and echocardiographic data, especially to estimate major outcomes and to analyze freedom from SVD.
Conclusions
Trifecta and Perceval bioprostheses appear to be associated with similar and equally satisfactory late outcomes, in terms of survival and freedom from cardiac death, from reoperation, and from prosthesis-related adverse events.
The Trifecta prosthesis carries a higher risk of SVD, starting from medium-term follow-up, even if this risk does not seem to be significantly associated with the need for redo operation, at least during a medium-long term period. Close clinical and outpatient echocardiographic monitoring with appropriate medical therapy may help, at least in part, to prevent the progression of SVD over time, thus reducing the risk of reoperation.
The Perceval prosthesis can be considered a viable option among the latest-generation, most promising biological prostheses for the lifelong management of aortic valve disease.