Purpose
Image-based adaptive brachytherapy (IGABT) is now the standard of care for patients with locally advanced cervical cancer. The prospective EMBRACE-2 magnetic resonance imaging (MRI)-based brachytherapy study utilized hybrid applicators in ~74% of cases, demonstrating that conformal dosimetry (Table 1) can be achieved in majority of cases. This individualized approach translated into excellent local control rates (> 90%) and lower grade ≥ 3 morbidities than prior techniques [1, 2].
Table 1
Goals of dosimetry based on the EMBRACE-2 study
| EMBRACE-2 | D90 HR-CTV | D98 GTV | D2cc rectum | D2cc bladder | D2cc sigmoid |
|---|---|---|---|---|---|
| Aim | 90-95 Gy | ≤ 95 Gy | ≤ 65 Gy | ≤ 80 Gy | ≤ 70 Gy |
| Limit | ≥ 85 | ≥ 90 | < 75 | < 90 | < 75 |
Compared with conventional perineal interstitial techniques, most hybrid applicators penetrate less tissue, use fewer needles, and facilitate more reproducible needle positioning [1, 3]. These improved clinical outcomes and technological advancements have led to increased utilization of hybrid applicators [3]. Commercially available hybrid applicators now provide a lower barrier to implementation, broadening their use at centers without the ability to manufacture custom devices.
Despite these advantages, practical guidance on applicator selection based on clinical stage and extent of residual disease is lacking. Therefore, this manuscript aimed to guide applicator selection from one commercial vendor (Varian Medical Systems, Palo Alto, CA, USA), while procedure details or treatment planning were beyond the scope of this paper.
Selection planning steps
At our institutions, we utilize Bravos high-dose-rate (HDR) afterloader with wide range of Varian brachytherapy applicators:
intracavitary brachytherapy (ICBT): Tandem and ring (T&R) or tandem and ovoid (T&O);
hybrid ICBT with interstitial needles: T&O with needles or Aarhus hybrid vaginal template with straight and oblique needles;
multichannel and interstitial cylinders (with or without Kelowna template).
This paper highlighted case-based examples using hybrid applicators for IGABT, aiming to assist clinicians in appropriate selection of applicators based on clinical context. Ultimately, applicator selection depends on individual experience and judgement.
Proper applicator selection is based on clinical examination and MRI, both before the initiation of external beam radiation therapy (EBRT) and pre-implantation (MRI performed typically during week 4-5 of EBRT), to allow time for procedure planning. MRI at this stage is critical for guiding applicator choice, allowing the measurement of cervical and uterine length and residual disease. Imaging findings suggesting that ICBT alone may not be enough to meet tumor coverage and organs at risk (OAR) are as follows [1, 4]:
large volume residual disease, e.g., cervical width > 4 cm or high-risk clinical target volume (HR-CTV) > 30 cc;
eccentric location of tumor;
residual medial or lateral parametrial involvement, or vaginal extension beyond the proximal 1/3rd.
As a general principle, a single intrauterine tandem provides adequate dose coverage for approximately 2 cm of tissue on either side of the uterine canal (4 cm in total if disease is symmetric) [5].
Applicators’ overview
Aarhus tandem and ring hybrid applicator, and T&O hybrid applicator
The Aarhus hybrid tandem and ring applicator has a ring design, but lacks the ring channel. When the Aarhus template is utilized, needles must be positioned on both sides of the ring to approximate a conventional T&R isodose distribution. Straight needles can further extend coverage by additional ~1 cm laterally (total 3 cm from the midline), based on the number of needles and weightings of dwell positions used in the needles on the side of their placing [6, 7]. Oblique needles can add another ~1 cm laterally (total of up to 4 cm from the midline when used in combination with straight needles), based on the location of target, number of needles, and dwell position weighting [8]. Insertion depth of needles is generally 2-4 cm above the surface of ring or ovoids, depending on the length of cervical canal and vertical dimension of disease identified on imaging [5]. Caution is advised with the insertional depth of oblique needles, as they can be close to parametrial vessels and increasing the risk of vascular injury. Image guidance (transabdominal or transrectal ultrasound) is strongly recommended to ensure accurate needle placement.
The T&O hybrid applicator allows standard ovoid loading; interstitial needles can be added unilaterally or bilaterally as needed to extend isodose coverage similarly to straight and oblique needles in the Aarhus hybrid T&R.
In various cases, MRI-guided ICBT alone may be employed for the first fraction of treatment. If the resulting dosimetry shows inadequate coverage of HR-CTV or excessive organs at risk (OARs) doses, continuing with ICBT alone for subsequent fractions may not be appropriate. For these clinical situations, a hybrid applicator can be considered for the remaining fractions. In that case, a simulated plan can be created using the first fraction MRI with applicator in situ, given geometric compatibility with tandem and ring and Aarhus hybrid tandem and ring. BrachyVision planning system allows planners to replace standard ring with the same size of Aarhus ring, adding interstitial needles for simulated planning. This simulated plan can help determine the appropriate number of needles, needle placement, and depth of insertion for the remaining fractions to optimize target coverage and OARs sparing as well as improves clinicians’ confidence in adequate needle insertion and placement.
Cylinder-based applicators and Kelowna template
Multichannel and interstitial cylinder
One limitation of the Aarhus and hybrid T&O applicator is the inability to adequately treat a disease extending the proximal part of vagina. In these situations, the use of multichannel or interstitial cylinders with tandem is recommended. If vaginal wall thickness is approximately 5 mm or less, adequate coverage can be achieved with a multichannel cylinder or interstitial cylinder alone. However, as described by Glaser et al., coverage up to 7 mm of vaginal depth may be feasible with these techniques [9]. When the vaginal wall is thicker than 7 mm, template-guided (Kelowna) needle insertion is typically required to provide adequate dose coverage. Interstitial cylinder can be used in combination with central tandem. The interstitial cylinder has oblique needles, which can help treat supra-vaginal disease in the cervix and/or parametrium. It can also be employed to treat asymmetrical vaginal recurrences in endometrial cancer.
Kelowna template is necessary when using multichannel or interstitial cylinder with tandem for extended lateral, posterior, or distal vaginal disease coverage. The template allows more customizable needle distribution through the perineal template, enabling improved conformality and dose escalation in the areas of residual disease while sparing OARs. It should be noted that only the 2.5 cm cylinder is compatible with the Kelowna template.
Practical applications
Case 1: Stage IIB cervical cancer treated with Aarhus tandem and ring hybrid applicator with straight needles
Patient with stage IIB cervical cancer completed concurrent chemotherapy and EBRT. Post-EBRT MRI pelvis showed residual cervical lesion measuring 4.3 cm in width (Figure 1). For residual disease with a width larger than 4 cm, the use of additional straight interstitial needles is needed to achieve adequate target coverage. It can be accomplished using either the Aarhus applicator with straight needles (Figure 1B), or tandem and ovoid with straight needles. The choice between these applicators depends on individual clinician preference. Needle position is determined based on MRI images (Figures 1, 2). In cases where the vaginal apex is narrow, small ring of the Aarhus applicator may be easier to insert. Conversely, in patients with a more capacious vaginal canal, larger ovoids may offer better anatomical conformity. Overall, both types of applicators can achieve target coverage and help reduce dose to OARs (Figures 1, 2). Figure 2 shows the hybrid T&O that is not MRI-compatible, and a newer version of T&O hybrid applicator that is MRI-compatible with similar straight and oblique needles.
Fig. 1
Patient treated with Aarhus tandem and ring hybrid applicator with straight interstitial needles. A) Post-EBRT MRI pel vis showing 4.3 cm residual width of cancer. B) Aarhus applicator with potential straight hole position marked with red circle to be used to cover the disease. C) CT planning scan axial view showing central tandem with four straight needles covering the tumor. D) CT planning scan with coronal view displaying depth of needles up to lower uterine segment. E) Clinical goals showing dosimetry to achieve EMBRACE-2 goals
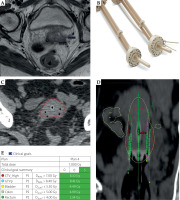
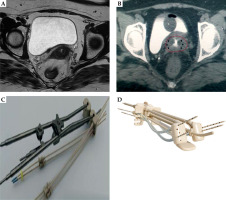
Fig. 2
Patient treated with hybrid tandem and ovoid and straight interstitial needles. A) Post-EBRT MRI pelvis revealing resid ual disease involving medial parametria. B) CT planning scan axial view showing tandem with six straight needles. C) Tandem and ovoid applicator with guide tube indicating straight and oblique needles. D) MRI-compatible tandem and ovoid applicator
Case 2: Stage IIB cervical cancer treated with ICBT with straight and oblique needles
Stage IIB cervical cancer patient completed concurrent chemotherapy and EBRT. Post-treatment MRI pelvis revealed moderate treatment response, with residual cervical lesion measuring 5.6 cm in width and extending to the left posterior and lateral parametria (Figure 3). Given that the target width was more than 5 cm, a second row of oblique interstitial needles were required to adequately extend isodose line. This can be achieved with either the Aarhus tandem and ring hybrid applicator, or ovoids with straight and oblique needles. Specifically, in the utilization of posterior and lateral oblique needle channels, it is critical to cover the residual disease (Figures 3, 4). Either applicator can achieve the desired dosimetry, and the choice is driven by user preference and anatomical considerations.
Fig. 3
Patient treated with Aarhus tandem and ring hybrid applicator with straight and oblique needles. A) Post-EBRT MRI pelvis showing 5.6 cm residual width of cancer with eccentric disease on the left. B) Aarhus applicator with potential straight and oblique hole position marked in red to be used to cover the disease. C) MRI axial scan showing central tandem with four straight needles and one oblique needle covering the disease. D) CT scan with coronal view demonstrating depth needle up to lower uterine segment. E) Clinical goals showing dosimetry to achieve EMBRACE-2 ideal goals
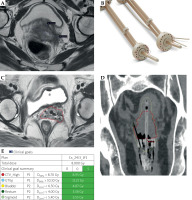
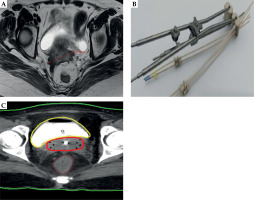
Fig. 4
Patient treated using tandem and ovoid with straight and oblique interstitial needles. A) Post-EBRT MRI showing residual disease involving lateral parametria. B) Tandem and ovoid applicator with guide tube showing straight and oblique needles. C) CT planning scan axial view of tandem with 7 interstitial needles (straight and oblique)
Case 3: Stage IIIC1 cervical cancer treated with ICBT with interstitial cylinder
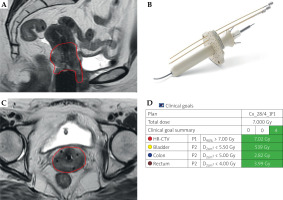
Patient with stage IIIC1 cervical cancer with extension into the upper vagina completed concurrent chemotherapy and EBRT. Post-EBRT MRI pelvis demonstrated a good response, with residual disease noted in the upper part of vagina. Given the extent of the disease into the upper one-third of vagina, adequate coverage would not be obtained with ovoids or ring alone. Additionally, the lateral extent of disease measured 4.6 cm, thus an interstitial multichannel cylinder with central tandem was utilized (Figure 5).
Fig. 5
Patient treated with tandem, interstitial cylinder, and interstitial needles. A) Post-EBRT MRI demonstrating residual disease extending into upper vagina in sagittal view. B) Interstitial cylinder. C) Post-EBRT MRI showing 4.6 cm × 1.7 cm residual disease in axial view. D) CT planning scan with coronal view of depth needle up to lower uterine segment. E) CT planning scan axial view of tandem with seven interstitial needles. F) Clinical goals showing dosimetry to achieve EMBRACE-2 ideal goals
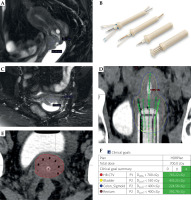
Case 4: Stage IIB cervical cancer treated with ICBT with multichannel cylinder and Kelowna template
Patient with stage IIB cervical cancer with extension to the upper 4 cm of vagina completed concurrent chemotherapy and EBRT. Post-EBRT MRI pelvis showed marked response, with residual induration noted in the upper part of vagina. Given the extent of disease involving more than one-third of the vagina, adequate coverage could not be achieved by ovoids and ring alone. Therefore, a multichannel cylinder with central tandem and Kelowna template was employed (Figure 6). The multichannel cylinder allowed improved coverage of the vaginal extent of disease, measured to be approximately 6 mm. In this case, the addition of Kelowna template facilitated the insertion of lateral needles into the cervix and medial parametria, ensuring adequate dose coverage of the residual disease (Figure 6).
Fig. 6
Patient treated with multichannel cylinder and Kelowna applicator. A) MRI of multichannel cylinder with central tandem in place to cover HR-CTV (red) extending to vagina. B) Multichannel applicator with central tandem and Kelowna applicator MRI axial scan showing central tandem with four needles through Kelowna applicator to cover parametrial disease. C) Coronal MRI views of oblique needles to help cover cervix and parametria. D) Clinical goals showing dosimetry to achieve EMBRACE-2 goals
Case 5: Stage IIIB cervical cancer treated with ICBT with interstitial cylinder and Kelowna template
Patient with stage IIIB cervical cancer extending to the upper 4 cm of vagina completed chemotherapy and EBRT. Post-EBRT MRI pelvis demonstrated good response, with residual induration in the upper part of vagina. As the disease extended to more than one-third of the vagina, coverage with a standard ovoid or ring would be insufficient. The plan was to treat using interstitial cylinder with central tandem and Kelowna template. The residual vaginal disease had a thickness of approximately 5 mm, therefore, oblique needles were inserted through the cylinder (Figure 7).
Fig. 7
Patient treated with tandem, interstitial cylinder, Kelowna applicator, and interstitial needles. A) MRI showing residual disease measuring 6.2 cm in axial view and B) 4.2 cm in sagittal view. C) Interstitial cylinder applicator with central tandem and Kelowna applicator. D) CT scan axial of central tandem with 11 needles through interstitial cylinder and Kelowna applicator to cover lateral parametrial disease. E) CT planning scan axial view of 11 interstitial needles. F) Clinical goals showing dosimetry to achieve EMBRACE-2 ideal goals
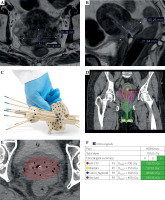
The interstitial cylinder helped cover vaginal extent of the residual disease, as the thickness of 5 mm plus oblique needles through interstitial cylinder facilitated covering of the cervix and parametria without the need of template involvement.
Conclusions
Image-guided adaptive brachytherapy is now considered the standard of care for cervical cancer patients. This paper aimed to provide a practical guide for clinicians by outlining the flexibility of applicators available, offering recommendations on their selection based on clinical scenarios. It was not intended to replace clinical judgement or individualized decision-making, rather to guide and support physicians in choosing the most appropriate applicators available to optimize brachytherapy outcomes.










