Introduction
Lung cancer is the second most common cancer in the United Kingdom and the leading cause of cancer-related death [1, 2]. Between 2017 and 2019, the incidence rates for lung cancer were highest among people aged 80 to 84. Each year, approximately 45% of all new lung cancer cases in the UK are diagnosed in individuals aged 75 and older [3]. Due to improvements in the healthcare system and lifestyle changes over the past 40 years, life expectancy in the UK has increased.
Therefore, it is important to re-evaluate management options for this population of patients. In the last few decades, there has been an evolution in approaches to thoracic surgery with minimal invasive techniques (video-assisted thoracoscopic surgery (VATS) and robotic-assisted thoracoscopic surgery (RATS)) and the adoption of enhanced recovery after surgery (ERAS) protocols. These advances facilitate faster recovery, earlier return to a routine lifestyle, and reduced complication and morbidity rates [4, 5]. In addition, recent trials suggest that sublobar resection is not inferior to lobectomy and has additional benefits, with preservation of more lung tissue and a decreased risk of postoperative dyspnoea [6, 7]. Furthermore, small trials have compared the use of stereotactic ablative radiotherapy (SABR) to surgery in the treatment of early-stage lung cancer. Despite the small number of patients recruited in these trials, they concluded that SABR could be an alternative to surgery in this group of patients [8, 9].
Accordingly, due to the lack of studies that discuss the octogenarian group of patients (≥ 80 years old), we focused on this patient population to evaluate the outcomes of both modalities of treatment for early-stage lung cancer (SABR vs. surgery) to guide surgeons and physicians in making a shared decision with patients about the best treatment options for them.
Material and methods
The data for this study were collected by the Scottish Cancer Network (SCAN) from the South-East of Scotland over the past 10 years. The South-East Scotland region has a population of 1.4 million people, accounting for 26% of Scotland’s total population [10, 11]. SCAN data were retrieved after permission was granted from the SCAN audit office to access, review and publish findings from the dataset.
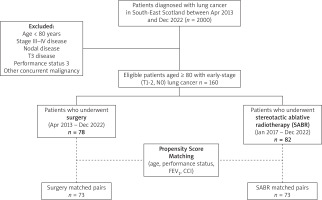
This study assessed 2,000 patients diagnosed with lung cancer retrospectively between April 2013 and December 2022. We included only patients aged 80 years and older. Specifically, we included 78 patients who underwent surgical resection (Group 1) between April 2013 and December 2022 and 82 patients who received SABR for early-stage lung cancer (T1-T2 N0) between January 2017 and December 2022. It is important to note that SABR was only introduced in this region in 2017, and by December 2022, a total of 353 patients had been treated with SABR (Figure 1).
In our exclusion criteria, we omitted any patients with nodal disease, PS 3, T3 disease, stage IV lung cancer, or other types of cancer. The Charlson Comorbidity Index (CCI) was used to assess patients’ fragility. The baseline score for this population (≥ 80 years old with lung cancer) was 6 points; therefore, we classified these patients into two groups: 6–8 or above 8.
Only patients with stage I non-small cell lung cancer (NSCLC) were eligible for SABR treatment. The treatment protocols for SABR included 54 Gy delivered in three fractions, 55 Gy in five fractions, or 60 Gy in eight fractions.
Any missing information from the SCAN data was obtained from the patients’ digital medical records.
Clinical notes explaining decisions for patients with good PS and pulmonary lung function being treated by SABR were unavailable, but all cases were discussed in the multidisciplinary team (MDT).
This study is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines, and the completed STROBE checklist pertinent to this study is provided in the supplementary materials [12].
Statistical analysis
Statistical analyses were conducted via Python (v3.13.1). For continuous variables, the two groups were compared using Student’s t-test or the Mann-Whitney U test for parametric and non-parametric data, respectively. For categorical variables, the χ2 test or Fisher’s exact test was used. A Kaplan-Meier survival analysis was conducted on the unmatched cohort to assess 5-year survival. A propensity-score matched analysis was conducted to account for differences in preoperative variables, using logistic regression modelling and matching by the variables age, PS, forced expiratory volume in the first second (FEV1), and CCI. This yielded 73 matched pairs in a 1 : 1 ratio using the nearest matching with replacement. Covariate balance after matching was assessed using standardised mean differences, with values < 0.1 being indicative of negligible imbalance and values > 0.2 reflecting potentially important residual imbalance. Matching with replacement was deemed appropriate due to the small sample size of the cohort; however, it means that the log-rank p-values associated with Kaplan-Meier analysis in the matched cohort should be treated cautiously and only used as an adjunct to the whole cohort analysis rather than interpreted independently. Overall survival (OS) was calculated using Kaplan-Meier survival analysis; log-rank tests and confidence intervals were calculated when appropriate.
Results
In the non-matched group, the median age in the surgical group was 81 years, and in the SABR group was 83 years (p < 0.001). The PS was significantly better in patients who underwent surgery compared to those who had SABR. There was no significant difference between groups in the CCI, but there was in the forced expiratory volume in one second (FEV1). Approximately half (53.8%) of the patients who underwent lung resection had a biopsy proving malignancy before surgery, but only 18.3% of patients treated with SABR had a biopsy proving malignancy. In the surgical group, most of the patients (85.9%) had minimally invasive surgery with VATS, and 77% of patients had a lobectomy. Only 3.8% of patients had R1 resection, whereas the rest had complete resection (Table I).
Table I
Patient characteristics and pathological outcomes
It is noteworthy that patients who underwent surgery had a post-operative upstage in T and N stages (32% and 11.5%, respectively). There were 3.8% of patients who were staged after surgery as N2 disease (Table I).
In patients who had surgery, we used the Clavien-Dindo Score [13] to classify the post-operative complications (Table II).
Table II
Clavien-Dindo score
Regarding the postoperative complications, there was no 30-day mortality, but there was 1 case of 90-day mortality. In the SABR group, there was no 30-day or 90-day mortality (Table III).
Table III
Postoperative complications
Approximately 45% of patients had no complications. The average length of stay (LOS) was 7.6 days. The most common complications were chest infection (34.6%), atrial fibrillation (AF) (15.3%), and prolonged air leak (more than 5 days) (10.2%). There were no cases of bleeding, reoperation, empyema, bronchopleural fistula (BPF), or myocardial infarction (MI) (Table III).
The OS was significantly better in patients who underwent surgery than in those who underwent SABR (52.6 months vs. 36.7 months, p < 0.001). There was no significant difference in 1-year or 3-year survival between groups, but 5-year survival differed significantly (surgery: 52.5% vs. SABR: 21.1%, p = 0.001) between groups. There was no significant difference in recurrence or disease-free survival (Table IV).
Table IV
Survival and recurrence in the non-matched groups
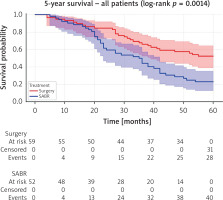
Prior to propensity score matching, a 5-year Kaplan-Meier survival analysis was conducted on the unmatched cohort with 59 patients in the surgery group and 52 in the SABR group. Median follow-up was 38.0 months (IQR: 24.0, 61.2) overall, with the surgical group median follow-up being 51.5 months (IQR: 30.2, 76.0) and the SABR group being 32.5 months (IQR: 22.0, 45.0). There was a statistically significant difference (log-rank p = 0.0014) in survival outcomes in 5-year survival, with surgery (52.5%) showing superior survival outcomes as compared to SABR (23.1%).
After undertaking propensity score matching (PSM) by age, PS, FEV1 and CCI, there were 73 patients in each group. The median age was 81 years in both the surgical and SABR cohorts (Table V). Assessment of balance using standardised mean differences (SMD) demonstrated residual imbalance for several baseline characteristics, importantly in certain lung function categories (FEV1 60-80% SMD = 0.40, FEV1> 80% SMD = 0.26). Accordingly, the matched cohort analyses were interpreted as supportive of the findings from the full unmatched cohort.
Table V
Baseline characteristics for matched cohort
| Parameter | Surgery (73 patients) | SABR (73 patients) | Standardised mean difference (SMD) |
|---|---|---|---|
| Age, median (IQR) | 81.0 (2.00) | 81.0 (4.00) | 0.251 |
| Sex | 0.31 | ||
| Male | 34 (46.6%) | 23 (31.5%) | |
| Female | 39 (53.4%) | 50 (68.5%) | |
| Performance status | |||
| 0 | 24 (32.9%) | 25 (34.2%) | PS0: 0.03 |
| 1 | 40 (54.8%) | 43 (58.9%) | PS1: 0.08 |
| 2 | 9 (12.3%) | 5 (6.85%) | PS2: 0.18 |
| Smoking | 0.45 | ||
| Yes | 60 (82.2%) | 70 (95.9%) | |
| No | 13 (17.8%) | 3 (4.1%) | |
| Charlson Comorbidity Index (CCI) | 0.23 | ||
| 6–8 | 65 (89.0%) | 59 (80.8%) | |
| > 8 | 8 (11.0%) | 14 (19.2%) | |
| Forced expiratory volume in first second (FEV1) | |||
| < 60% | 2 (2.7%) | 5 (6.8%) | FEV1< 60%: 0.19 |
| 60–80% | 19 (26.0%) | 8 (11.0%) | FEV1 60–80%: 0.40 |
| > 80% | 52 (71.2%) | 60 (82.2%) | FEV1> 80%: 0.26 |
| Pulmonary disease | 0.06 | ||
| Yes | 27 (37.0%) | 29 (39.7%) | |
| No | 46 (63.0%) | 44 (60.3%) | |
| Cardiac disease | 0.23 | ||
| Yes | 23 (31.5%) | 16 (21.9%) | |
| No | 50 (68.5%) | 57 (78.1%) | |
| Hypertension | 0.46 | ||
| Yes | 41 (56.2%) | 25 (34.2%) | |
| No | 32 (43.8%) | 48 (65.8%) | |
| Diabetes | 0.63 | ||
| Yes | 11 (15.1%) | 0 (0%) | |
| No | 62 (84.9%) | 73 (100%) | |
| History of cancer | 0.18 | ||
| Yes | 25 (34.2%) | 19 (26.0%) | |
| No | 48 (65.8%) | 54 (74.0%) | |
| Peripheral vascular disease | 0.30 | ||
| Yes | 12 (16.4%) | 4 (5.48%) | |
| No | 61 (83.6%) | 69 (94.5%) | |
| Stroke/transient ischaemic attack | 0.10 | ||
| Yes | 5 (6.85%) | 7 (9.59%) | |
| No | 68 (93.2%) | 66 (90.4%) | |
| Renal disease | 0.05 | ||
| Yes | 5 (6.85%) | 4 (5.48%) | |
| No | 68 (93.2%) | 69 (94.5%) |
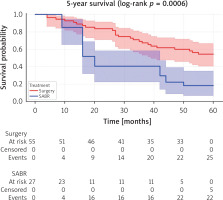
After Kaplan-Meier survival curve analysis, there was a statistically significant difference in survival outcomes after 5 years in the matched cohort (surgery: 54.5%, SABR: 18.5%, log-rank p < 0.001). No significant difference was found between the matched groups regarding recurrence after a period of disease-free survival (Figures 2, 3).
Discussion
With the rapid evolution and advancement of technology in thoracic surgery, treatment options, and the adoption of screening programmes in the United Kingdom (UK), it has become essential to discuss treatment options for octogenarian patients, particularly with the improvements in quality of life and survival rates within this population. There is limited literature addressing the treatment of lung cancer in this age group, and strong evidence comparing SABR to surgery for early-stage lung cancer is lacking.
Two randomised controlled trials (RCTs), ROSEL and STARS, involved a small number of patients and were both closed early. Despite these limitations, they concluded that SABR is significantly more effective than surgery concerning 1-year and 3-year OS rates. However, these trials have faced several critiques. For instance, confirmation of malignancy was not mandatory in the ROSEL trial, potentially allowing benign lesions to be included. Moreover, in the surgical group, 19 patients underwent open lobectomy, even though current evidence supports sublobar resection and the standard of care has shifted toward minimally invasive surgery [8, 9].
In our opinion, the 1-year survival rate should not be considered a measure of success in early-stage lung cancer, as the expected survival for this patient group without treatment has been reported to be approximately 13 months [14, 15]. Therefore, we believe that our study, which is based on propensity-matched analysis from a national database and includes long-term follow-up, can contribute valuable evidence when discussing treatment options for these patients.
During our literature review, we identified two matched studies on the same topic. Park et al. published a propensity score-matched analysis involving 48 patients aged 75 years and older with stage I lung cancer who were treated with either SABR or surgery. The findings revealed that OS was significantly better in the surgical group (surgery vs. SABR: 65.9% vs. 40.3%; p = 0.034). However, there was no significant difference in the cumulative incidence of cancer-related deaths (p = 0.089) or recurrence rates (p = 0.111) between the two groups. The authors concluded that surgery should be the preferred treatment option for patients with stage I lung cancer aged 75 years or older [16].
Ni et al. published a PSM analysis on patients aged 80 years or older with clinical stage Ia (T1N0M0) NSCLC between 2012 and 2017. Their findings indicated that radiotherapy was associated with significantly worse OS compared to surgery (p < 0.001). The OS rates at 1, 3, and 5 years for the surgical group were 90.0%, 76.9%, and 59.9%, respectively, while the radiotherapy group had OS rates of 86.0%, 54.3%, and 28.0%, respectively. They concluded that surgery with lymph node dissection should be the treatment of choice for stage Ia in octogenarians [17].
Despite octogenarians often having good PS, pulmonary function tests (PFT), and an acceptable CCI, there remains a concern regarding the risk of comorbidities after surgery and the potential impact of surgery on these patients. In our study, we observed no 30-day mortality and only 1 case of 90-day mortality. The average LOS was 7.6 days, and the most common complications included chest infections, AF, prolonged air leaks, and acute kidney injury (AKI).
It is important that this information is clearly communicated to patients during discussions about treatment options. We should engage in shared decision-making with the patient, explaining the risks of mortality and comorbidities in the post-operative period while also highlighting the better long-term survival outcomes that surgery offers. Furthermore, surgery provides the opportunity for better staging and lymph node clearance. Our results indicated that there was an 11.5% rate of unexpected upstaging in nodal disease. For patients who tolerate surgery well, there may be the option of adjuvant treatment in cases of unexpected lymph node involvement.
Patients should discuss the potential side effects and toxicity associated with SABR. Cannon et al. conducted a retrospective study involving 30 patients aged 80 and older who received SABR, with a median total dose of 54 Gy delivered in 3 fractions. In this study, 3 patients experienced late-grade 3 pulmonary dyspnoea, but there were no cases of grade 4 or 5 toxicities. Additionally, 1 patient developed late-grade 2 pneumonitis, and 3 patients had late-grade 1 pneumonitis. Three patients reported grade 1 chest wall pain. The authors concluded that the octogenarian group can tolerate SABR [18].
In another retrospective study by Brooks et al. from the University of Texas MD Anderson Cancer Center, the effectiveness of SABR was evaluated in elderly patients with early-stage lung cancer. This study included 330 patients aged 75 and older, treated with SABR at doses of either 50 Gy in 4 fractions or 70 Gy in 10 fractions, with a median follow-up of approximately 55 months. They found that the median OS rates for patients aged 75 and older were 86% at 1 year, 57.5% at 2 years, and 39.5% at 5 years. Importantly, no patients aged 75 or older experienced grade 4 or 5 toxicity. The most common grade 2 and 3 complications in this group included fatigue, pneumonitis, chest wall pain, and rib fractures, with only 8 reported cases of cardiac events [19].
The topic of SABR versus surgery in the treatment of early-stage lung cancer requires more evidence to guide discussions in MDT meetings. This evidence will help surgeons and patients make shared decisions about the most appropriate treatment. Currently, the ongoing phase III randomised controlled trial known as the STABLE-MATES Trial aims to compare sublobar resection to SABR in patients with stage I lung cancer (where the tumour size is 4 cm or less). The primary endpoint of this trial is OS at 3 years, while the secondary endpoints include progression-free survival at 5 years and toxicity [20].
One limitation of our study is the small sample size in the matched groups, along with a lack of data regarding why some patients with good PS and PFT were treated with SABR instead of surgery. Additionally, PSM with replacement introduces a larger variance, which therefore may reflect less precise estimates [21]. For this reason, the Kaplan-Meier analysis results produced in the matched cohort must be treated with caution when interpreting their significance. We considered this approach appropriate due to the small sample size and the nature of the PSM analysis being used as an adjunct to the whole cohort analysis. Furthermore, the data acquired were only representative of one region and therefore may not be generalisable to the United Kingdom or worldwide. Another limitation is a lack of detail on quality-of-life outcomes, which is outside the scope of this study but warrants further research. Finally, a key limitation of this study is the fact that there were non-overlapping eras between groups (surgical patients from 2013–2022 vs. SABR from 2017 onwards). This not only affects the Kaplan-Meier survival analysis, but also means that temporal changes in perioperative care and oncological management may have influenced outcomes independently of treatment modality.