Introduction
Indocyanine green (ICG) is a water-soluble fluorescent dye that, after intravenous administration, rapidly binds to plasma proteins and is almost completely excreted in bile; its half-life is about 3–4 minutes [1, 2]. When excited with light of wavelength 750–800 nm, ICG emits fluorescence in the near-infrared range (~830–840 nm), which is captured in real time by NIR cameras [1, 3]. This enables dynamic assessment of perfusion and visualization of anatomical structures “invisible” in white light (e.g. anatomical planes, small lesions, vessels and lymphatic pathways) without disturbing the standard operative field [4–6].
Compared to visible dyes (e.g. methylene blue), NIR imaging is characterized by deeper tissue light penetration (about 1–1.5 cm), better contrast, and the ability to simultaneously view the operative field under white light and fluorescence [5, 6]. ICG binds to plasma lipoproteins, which confines it to the intravascular space and makes the signal intensity reflect local blood flow [4, 7]. In clinical practice, the dye serves as a navigation tool: it facilitates detection of small lesions, provides objective assessment of perfusion, and supports identification of lymphatic pathways [4, 7].
The safety profile of ICG is favourable: adverse events, including anaphylactic reactions, are rare (about 1 : 10,000), and the doses used in thoracic surgery remain much lower than the permissible daily limits (approx. 2–5 mg/kg/day) [8–10]. In patients with severe liver failure, caution is advised due to its route of elimination [9].
The development of minimally invasive thoracic surgery techniques – video-assisted thoracic surgery (VATS), robotic-assisted thoracic surgery (RATS) has limited the ability to palpate and directly assess perfusion, making localization of small subpleural lesions, precise delineation of the intersegmental plane, and assessment of gastric conduit perfusion before anastomosis more difficult [6, 11]. The use of ICG with an NIR camera effectively compensates for these limitations, providing the surgeon with real-time anatomical-functional information [4, 6, 11–13].
In thoracic surgery, ICG is currently used primarily in four areas: (1) delineation of the intersegmental plane (ISP) in segmentectomy, (2) localization of small lung nodules, (3) perfusion and anastomosis assessment of the gastric conduit in esophagectomy, and (4) visualization/identification of the thoracic duct [11–20]. A summary of doses, routes of administration, and main clinical indications is presented in Table I [14–37].
Table I
ICG dosing, administration and clinical goals in thoracic surgery (with representative references)
| Clinical scenario/goal | Administration route | Typical dose/concentration | Timing | Key intraoperative read-out | Practical notes | References |
| Anatomical segmentectomy – intersegmental plane identification (NEGATIVE staining) | Intravenous (IV) bolus | 0.05–0.1 mg/kg (3–5 mg); optional micro-bolus 1–2 mg if demarcation fades | After ligation/division of the target segmental artery; visualize within 10–60 s | Perfused segments fluoresce; the target segment remains dark clear intersegmental demarcation | Consider adjunct endobronchial ICG for complex/multisegment resections; optimize NIR settings and camera distance | [10, 11, 24] |
| Anatomical segmentectomy – intersegmental plane identification (POSITIVE staining) | Endobronchial (bronchoscopic) injection | 0.5–1.0 ml at 1.25–2.5 mg/ml (diluted) into the target segmental bronchus | Before parenchymal division; allow short dispersion time | Target segment fluoresces; adjacent parenchyma relatively dark | May be combined with IV bolus (dual visualization); requires accurate bronchoscopic deposition | [22, 25, 26] |
| Localization of nonpalpable pulmonary nodule (CT-guided marking) | Percutaneous injection under CT guidance | 0.2–0.5 ml at 1.25–2.5 mg/ml; deposit 5–15 mm from the lesion | Same-day surgery; direct transfer to the OR after marking | Fluorescent spot guides lesion exposure and margin control for wedge/segmentectomy | Optional radiopaque contrast to verify position on CT; avoid pleural transgression near bullae | [13, 29, 31] |
| Localization of nonpalpable pulmonary nodule (navigational bronchoscopy – ENB/VNB) | Endobronchial (bronchoscopic) injection | 0.5–1.0 ml at 1.25–2.5 mg/ml | Immediately pre-op under general anesthesia or in a hybrid suite | Persistent fluorescence at/near the target facilitating resection with adequate margin | Useful for deeper/central lesions; avoids pleural puncture; coordinate with anesthesia and OR schedule | [14, 32] |
| Localization of nonpalpable pulmonary nodule (navigational bronchoscopy – ENB/VNB) | Endobronchial (bronchoscopic) injection | 0.5–1.0 ml at 1.25–2.5 mg/ml | Immediately pre-op under general anesthesia or in a hybrid suite | Persistent fluorescence at/near the target facilitating resection with adequate margin | Useful for deeper/central lesions; avoids pleural puncture; coordinate with anesthesia and OR schedule | [14, 32] |
| Thoracic duct mapping/chyle leak management | Intradermal/subcutaneous injections into both groins (± pedal) | 0.2–0.5 ml per groin at 2.5 mg/ml (bilateral) | Wait 30–90 min for transit; gentle massage at injection sites | Fluorescent thoracic duct course and/or 'hot spot' of leakage under NIR | If no signal, add depot or assessment; adjust to local protocol and NIR system sensitivity | [18–20, 37] |
To date, no comprehensive thoracic surgery – focused narrative review encompassing all four thoracic indications for indocyanine green has been published. This review addresses that gap by synthesizing recent evidence, situating the evidence within current international guidance, and offering pragmatic, procedure-oriented recommendations for routine clinical practice.
The aim of this review is to analyse the current literature on the use of indocyanine green (ICG) in thoracic surgery, with a particular emphasis on the four main areas of application mentioned above. International guidelines and proposed management algorithms were also taken into account.
Methods
This is a narrative review. Literature searches were conducted in PubMed, Scopus, and Web of Science. Review articles, original studies, and meta-analyses from 2020 to 2025 concerning the use of ICG in thoracic surgery were included. Publications describing clinical applications, technical aspects of fluorescence, efficacy and safety of ICG in segmentectomies, nodule localization, gastric conduit perfusion assessment, and thoracic duct mapping were analysed. Society guidelines and position statements of the European Association for Endoscopic Surgery (EAES), the European Society of Thoracic Surgeons (ESTS), and the International Society for Fluorescence Guided Surgery (ISFGS) were also reviewed. Exclusion criteria: conference abstracts, letters and editorials, single-patient case reports, and studies outside the predefined thoracic indications of ICG.
Results
Anatomic lung segmentectomy – delineation of the intersegmental plane
With the increasing detection of small tumour foci, segmentectomy is increasingly regarded as an oncologically equivalent alternative to lobectomy in carefully selected patients, with the added benefit of preserving lung parenchyma [21]. Reliable determination of the intersegmental plane (ISP) is crucial for success, which in thoracoscopy can be difficult, especially when the classical inflation–deflation method produces uneven inflation or fails in patients with emphysema [5, 11]. The use of indocyanine green fluorescence has improved the predictability of this step [22–24].
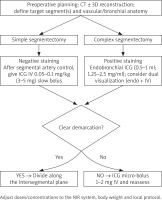
The so-called “negative staining” technique is most commonly used: after ligating or transecting the segmental artery, a small intravenous bolus of ICG is administered (typically 0.05–0.1 mg/kg, about 3–5 mg). Within a few tens of seconds, all perfused segments become fluorescent under NIR imaging, while the target segment remains dark. A sharp, well-demarcated boundary then appears, usually lasting 1.5–3 minutes; if necessary, a micro-bolus can be given to refresh the contrast [10, 21, 24, 25]. An alternative is “positive staining” – administration of small volumes of ICG bronchoscopically into the segment to be resected; this technique can also be combined with intravenous injection as part of “dual visualization”, especially in complex, multisegment resections [22, 25, 26]. A simplified algorithm of the procedure is shown in Figure 1.
The effectiveness of ISP visualization with ICG reaches about 90–95% in available meta-analyses and multicentre series. This translates into shorter identification time and a higher chance of obtaining a negative margin while maximizing parenchymal preservation, which is particularly important in ground-glass opacity (GGO) lesions and patients with limited respiratory reserve [23–25, 27, 28]. However, systemic and technical factors affect image quality: severe emphysema, presence of adhesions, delayed ICG injection relative to vessel ligation, or improper NIR camera settings can reduce contrast; in bronchoscopic marking, success depends on deposit precision and the team’s experience [3, 5, 22, 26, 27]. The 2023 ESTS consensus recommends using ICG/NIR as an adjunct tool for ISP identification in VATS/RATS segmentectomy (strength of recommendation: moderate) [28].
Localization of nonpalpable lung nodules
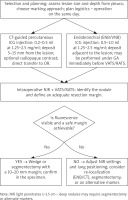
In the era of widespread computed tomography, increasingly smaller subpleural lesions or lesions located deeper than 5–10 mm from the pleura are selected for resection, which cannot be detected by palpation in VATS/RATS. To perform a parenchyma-sparing resection with an adequate margin, reliable localization of the lesion is necessary. ICG fluorescence has proven to be an effective and safe tissue marker which – unlike hookwire or coils – leaves no foreign body and is less likely to migrate [29–32]. The dye is most often administered locally: percutaneously under CT guidance (0.2–0.5 ml of ICG at a concentration of 1.25–2.5 mg/ml) just before surgery, or bronchoscopically using navigation (0.5–1.0 ml of similar concentration). Both methods provide very high detectability (usually > 90–95%; ~98% in meta-analyses) with a low complication rate, which is more related to the puncture technique than to the properties of the dye [13, 14, 30–32]. A decision-making scheme for localization is shown in Figure 2.
Logistics influence the outcome. Ideally, the operation should be scheduled on the same day in order to take advantage of the period of maximal fluorescence and minimize marker diffusion. For precise margin control, it is advisable to place the dye deposit 5–15 mm from the lesion; adding a radiopaque contrast medium to the ICG solution allows intraoperative CT verification of the deposit. Deep tumour location (> 20–30 mm from the pleura) and obesity may weaken detection due to limited NIR light penetration (~1–1.5 cm) [13, 14, 31, 32]. Intravenous administration of ICG alone to “find” the nodule, although studied, has low specificity and is not recommended as the sole method – it is only reasonable in the context of anatomical resection of the entire segment containing the lesion [30, 32].
Gastric conduit perfusion assessment and anastomosis in esophagectomy
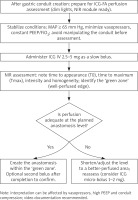
Anastomotic leak remains one of the most serious complications of esophagectomy; the perfusion of the distal gastric conduit segment plays a key role. ICG fluorescence angiography allows intraoperative, objective assessment of perfusion in real time and is a valuable complement to subjective visual evaluation [15–17, 33–35]. After creating the conduit and before performing the anastomosis, 2.5–5 mg of ICG is administered intravenously. The time to signal appearance (T0), time to peak (Tmax), intensity, and homogeneity of fluorescence are analysed. Areas with delayed or weaker fluorescence are considered ischemic; in such situations, conduit trimming or changing the level of the anastomosis may be indicated. Some teams use a second bolus after creating the anastomosis to confirm its perfusion [15–17, 33–35]. An intraoperative algorithm for gastric conduit perfusion assessment is shown in Figure 3.
In many series, the inclusion of ICG fluorescence angiography (ICG-FA) has led to modifications in the operation plan and has been associated with a reduction in anastomotic leak rates. Although assessment methods are still varied and some analyses are nonrandomized, the direction of the effect is consistent: a lower incidence of anastomotic leak after routine implementation of fluorescence [16, 17, 33–36]. The 2023 EAES recommendations advocate the use of fluorescent techniques for perfusion assessment in gastrointestinal surgery, including esophagectomy [17]. Hemodynamic factors (vasopressors, positive pressure ventilation) influence interpretation, so the assessment should be performed under stable conditions and documented on video.
Visualization and identification of the thoracic duct
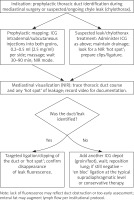
Injury to the thoracic duct during extensive mediastinal procedures is rare but clinically significant; in cases of evident chylothorax, rapid identification of the leak site is crucial for successful management. ICG lymphatic fluorescence facilitates both prophylactic identification of the duct course (e.g. in minimally invasive esophagectomy (MIE) with extended lymphadenectomy) and targeted ligation during reoperation [18–20, 37]. The technique is simple: after injection into the groin tissues (0.2–0.5 ml of ICG per side at 2.5 mg/ml), a signal appears in the mediastinum within 30–90 minutes, making the duct and the “hot spot” of the leak easily identifiable under the NIR camera. The method shortens search time, increases confidence in repair, and has a good safety profile; its limitations include the need to wait for dye transit and the absence of fluorescence in complete vessel occlusion. Experience from various centres confirms the usefulness of this technique in both the prevention and treatment of chylothorax [18–20, 37, 38]. A simplified management scheme for intraoperative prophylaxis and confirmed chylothorax is shown in Figure 4.
Discussion
The use of ICG in thoracic surgery fits into the trend of minimally invasive “intelligent surgery”, where the anatomical image is supplemented by functional information. In segmentectomy, the dye allows one to “see” the boundaries that were previously reconstructed indirectly – resulting in a more predictable course of the resection, preservation of healthy parenchyma, and higher chances of a negative margin. In many centres, ICG has become standard in segmental procedures performed via VATS/RATS, and the ESTS consensus formalizes this practice [24–26, 28]. In nodule localization, ICG solves typical problems of mechanical techniques: nothing is displaced, nothing needs to be removed from the lung, and the surgeon has a continuous guiding signal both to the lesion and during margin assessment [29–32]. Meanwhile, in esophagectomy, ICG serves as a final objective test before constructing/performing the anastomosis – allowing one more, objective assessment of whether the conduit end has a chance to heal properly. This explains the observed reduction in anastomotic leaks (AL) in the literature after routine implementation of fluorescence [16, 17, 33–36]. Finally, in thoracic duct mapping, fluorescence turns tedious, often fruitless searches into a targeted procedure and facilitates both the prophylaxis and treatment of chylothorax [18–20, 37, 38].
At the same time, limitations must be noted. Most data come from observational, often single-centre studies, and the protocols used are heterogeneous: ICG doses, concentrations, timing of administration and interpretation criteria differ between centres. In esophagectomy, there are no agreed quantitative thresholds (e.g. specific T0/Tmax values) that could serve as a basis for firm recommendations; similarly, in segmentectomy there is no universal, validated metric of “demarcation quality”. Efforts in the coming years should focus on standardization: description of equipment (NIR platform and its settings), dosing (mg, mg/kg, concentration, route), hemodynamic conditions at the time of assessment, and consistent definitions of outcomes (micro-/macro-margin, conversion, AL, chylothorax). Extending practice to semi-quantitative and quantitative methods (quantitative ICG – Q-ICG) may increase decision reproducibility and facilitate comparability of studies [5, 17, 33–36].
Experience from various centres confirms the usefulness of this technique in both the prevention and treatment of chylothorax. Implementation typically prolongs the procedure by a few to several minutes, but after an adaptation period the additional time becomes marginal. The cost of ICG and the NIR module is relatively small compared to the potential systemic benefits: fewer conversions, fewer leaks, fewer reoperations, and shorter hospital stays [7, 16, 31]. The safety profile of ICG is very favourable [7–9] – serious reactions are extremely rare – and the elimination of radiographic markers in nodule localization reduces radiation exposure and eliminates the problem of marker dislodgement/dislocation [31, 32]. Notably, commercial ICG formulations contain sodium iodide (as an excipient/stabilizer). Accordingly, caution is warranted in patients with known hypersensitivity to iodide salts due to the potential risk of hypersensitivity reactions, including rare anaphylaxis. Although true allergic reactions to ICG are rare, isolated cases of severe hypersensitivity have been reported. When using ICG, clinicians should be aware of its sodium iodide content and be prepared to manage potential allergic reactions, including anaphylaxis.
In the near future, further developments can be expected, such as quantitative ICG (Q-ICG) methods, integration of imaging with 3D reconstructions, and augmented reality to assist in planning complex multisegment resections and education; concurrently, work is underway on targeted molecular tracers (e.g. for sentinel lymph node mapping) [39]. Regardless of the direction of further development, standardization of practice and consistent reporting remain essential.
Conclusions
Fluorescence using indocyanine green has revolutionized many areas of modern thoracic surgery. In lung segmentectomy, ICG enables precise and rapid delineation of resection boundaries, which facilitates safer lung-sparing surgery. In the localization of nonpalpable lung nodules, this method significantly increases detection efficacy while reducing invasiveness and complications compared to traditional markers. In esophageal surgery, ICG fluorescence angiography has become a valuable tool for perfusion assessment as it allows identification of patients at risk of anastomotic ischemia and helps prevent some complications by changing the surgical strategy. Finally, the use of ICG for thoracic duct visualization helps surgeons avoid damaging it or effectively manage any injury, which translates to a lower incidence of chylothorax.
The current state of knowledge, supported by numerous publications from the last 5 years, confirms the utility and safety of indocyanine green in these applications. Moreover, many of the cited studies indicate improved treatment outcomes with the use of this technology (e.g. lower recurrence risk after segmentectomy, fewer conversions, reduced anastomotic leaks in esophageal surgery, and fewer lymphatic complications). However, it should be remembered that optimal use of ICG requires appropriate equipment (NIR camera), team experience, and – as experts emphasize – standardization of protocols. Ongoing clinical studies (including randomized trials) will help refine decision algorithms, for example by determining clear criteria for assessing gastric conduit perfusion or indications for prophylactic ligation of the thoracic duct. In parallel, new technologies are being developed, such as quantitative fluorescence analyses and targeted molecular contrast agents, which may make ICG assessment even more precise.
In summary, indocyanine green has established its position as a versatile tool supporting the thoracic surgeon. In the four areas discussed – pulmonary segmentectomy, nodule localization, gastric conduit perfusion, and thoracic duct procedures – the use of ICG aligns with current trends in minimally invasive “intelligent” surgery, providing valuable real-time information. As a result, procedures become more predictable and safer, ultimately translating to better patient care.