Purpose
Post-operative pelvic irradiation in patients with cervical cancer has been reported to reduce pelvic recurrence and improve survival [1, 2]. However, the incidences of pelvic and vaginal recurrences in the radiation field are 8.7-21.6% and 2.2%, respectively [1-3], and there is no standard treatment for previously irradiated pelvic recurrence. Several reports have indicated that re-irradiation with interstitial brachytherapy (ISBT) results in a local control rate of 36.3-76.9%. Late adverse events of grade 3 or higher were observed in 7.4-25.0% of gynecological cancer patients, mainly comprising cervical cancer patients who were previously treated with definitive radiotherapy [4-6]. However, there are few reports on re-irradiation in uterine cancer patients with vaginal recurrence after post-operative pelvic irradiation. Furthermore, while total dose administered is lower in patients with vaginal recurrence after post-operative pelvic irradiation than in patients undergoing definitive radiotherapy (including external beam radiotherapy [EBRT] and brachytherapy) as initial treatment, brachytherapy generally delivers a higher dose to organs at risk (OARs; namely the bladder, rectum, sigmoid colon, and small intestine) in patients with vaginal recurrence after post-operative radiotherapy.
Thus, this study aimed to evaluate the doses for OARs and outcomes of re-irradiation with ISBT in uterine cancer patients with vaginal recurrence after post-operative pelvic irradiation.
Material and methods
Study design and patients
Overall, 27 gynecological cancer patients with local recurrence after prior radiotherapy, including definitive radiotherapy (comprising EBRT and brachytherapy) and post-operative pelvic irradiation (comprising EBRT and/or brachytherapy), received re-irradiation with high-dose-rate (HDR) ISBT between January, 2012 and December, 2016 at the Tsukuba University Hospital. In this study, we included six uterine cancer patients who met the following criteria: 1. Post-operative pelvic irradiation performed as initial radiotherapy; 2. Initial radiotherapy did not include brachytherapy; and 3. No recurrence other than vaginal recurrence. Protocol was approved by the institutional review board of Tsukuba University Hospital (IRB No. H30-338), and requirement for informed consent was waived because of the retrospective nature of the study.
Radiotherapy
Previous whole-pelvis EBRT after initial surgery was delivered using a three-dimensional conformal technique (3D-CRT) or intensity-modulated radiotherapy (IMRT) using a linear accelerator photon energy higher than 10 MV. Whole-pelvis EBRT was administered at a dose of 45.0-50.4 Gy in 25-28 fractions. Concurrent chemoradiotherapy with weekly cisplatin was administered to cervical cancer patients with tumors of pathological stage ≥ T2b and/or tumors of pathological lymph node status N1. Whole-pelvis EBRT was principally applied within 8 weeks post-operatively.
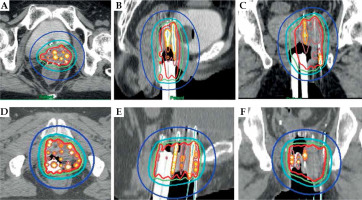
All patients underwent re-irradiation with HDR-ISBT. Brachytherapy was performed using an iridium-192 (192Ir) remote afterloading system (Micro-Selectron HDR-TM; Nucletron, Veenendaal, The Netherlands). For all patients, planning was based on computed tomography (CT) images of 2.5-mm slice thickness, and dose calculations were performed using Oncentra (Nucletron, Veenendaal; The Netherlands) without heterogeneity correction, in accordance with the TG-43 protocol. Martinez universal perineal interstitial template was used during ISBT. Stainless and/or plastic needles were inserted under CT guidance under spinal anesthesia and epidural anesthesia, and irradiation was performed twice a day for 3-4 days. Clinical target volume (CTV) included only macroscopic tumors identified on gynecological examination and based on CT and/or T2-weighted magnetic resonance imaging (MRI). An ISBT dose of 38-42 Gy in 6-7 fractions for 3-4 days was prescribed for CTV (Figure 1), and CTV D90 was evaluated.
Fig. 1
Dose distribution for ISBT. Axial (A), sagittal (B), and coronal sections (C) for the vaginal recurrence at the vaginal stump after post-operative irradiation. Axial (D), sagittal (E), and coronal sections (F) for the vaginal recurrence in the vaginal wall after post-operative irradiation. For vaginal recurrence at the vaginal stump, 13 stainless steel needles were inserted using Martinez universal perineal interstitial template, and a radiation dose of 6 Gy per fraction was prescribed for CTV (A-C). For vaginal recurrence in the vaginal wall, 18 stainless steel needles and five plastic needles were inserted using Martinez universal perineal interstitial template, and a radiation dose of 6 Gy per fraction was prescribed for CTV (D-F). Isodose line: blue line, 25% of the prescribed dose; light blue line, 50%; green line, 75%; red line, 100%; brown line, 150%; orange line, 200%; yellow line, 250%; white line, 300%. ISBT – interstitial brachytherapy; CTV – clinical target volume
Follow-up
Treatment responses were assessed 2-3 months after HDR-ISBT completion based on response evaluation criteria in solid tumors, using histopathology and/or MRI [7]. Late adverse events were graded based on the National Cancer Institute’s common terminology criteria for adverse events version 4.0. Follow-up visits comprised gynecological examination, cytology, blood tests, and imaging using CT or MRI every 2-6 months for 5 years.
Statistical analysis
For dose summation of brachytherapy and post-operative pelvic irradiation, equivalent dose in 2 Gy fractions (EQD2) was calculated based on a linear-quadratic model [8]. α/β ratio was assumed to be 3 Gy when calculating dose-volume parameters of OARs (i.e., D0.1cc and D2cc) [9].
Times to local recurrence, distant metastasis, and late adverse events were defined as periods from the completion of ISBT to the diagnosis of local recurrence, diagnosis of distant metastasis, and occurrence of adverse events, respectively. Follow-up duration was defined as the period from the completion of ISBT to the last medical examination.
Results
Patients and treatment characteristics are presented in Table 1. The median age of the patients was 57.5 years (range, 42-60 years). Five of the six patients had cervical cancer, while one had endometrial cancer. Post-operative pelvic irradiation was delivered to five and one patients using 3D-CRT and IMRT, respectively. The median time from initial surgery to vaginal recurrence was 25.7 months (range, 7.1-86.8 months). The median time from the completion of pelvic irradiation to ISBT initiation was 24.1 months (range, 9.0-104.5 months). The median tumor size, as measured using MRI just before ISBT, was 3.3 cm (range, 1.5-6.3 cm).
Table 1
Patient and treatment characteristics
[i] SCC – squamous cell carcinoma; RH – radical hysterectomy; TAH – total abdominal hysterectomy; SRH – semi-radical hysterectomy; BSO – bilateral salpingo-oophorectomy; PLN – pelvic lymph node dissection; RT – radiotherapy; CCRT – chemoradiotherapy; EBRT – external beam radiotherapy; 3D-CRT – three-dimensional conformal radiation therapy; IMRT – intensity-modulated radiation therapy; CT – chemotherapy; ISBT – interstitial brachytherapy
Table 2 summarizes CTV volumes, D90 for CTV and D2cc for OARs at ISBT, and total EQD2 of D2cc for OARs (including EBRT and ISBT). The mean CTV volume was 49.5 cc. The mean doses per fraction of ISBT for CTV D90, D2cc for the bladder, and D2cc for the rectum were 6.1 Gy, 4.4 Gy, and 3.8 Gy, respectively. D2cc for the sigmoid for patient number 1, 4, and 5 were < 0.1 Gy because of the distance between the vaginal tumor and the sigmoid. The mean total EQD2, including EBRT and ISBT, for D2cc for the bladder, sigmoid, and rectum were 92.1 Gy, 50.4 Gy, and 81.6 Gy, respectively.
Table 2
Doses for clinical target volumes (CTVs) and organs at risk (OARs)
Outcomes and adverse events are presented in Table 3. The median follow-up duration was 53.3 months (range, 17.7-71.8 months). A complete response (CR) was achieved in five of the six patients. Local recurrence was observed in two patients who experienced vaginal recurrence infiltrating the adjacent organs, such as the bladder and the rectum. For patient number 1, local recurrence, which was located above the vaginal stump, was observed at 26.5 months after re-irradiation with ISBT; the tumor grew rapidly, resulting in an enterovaginal fistula. The total EQD2 for D0.1cc of the bladder and rectum were 169.0 Gy and 91.9 Gy, respectively. For patient number 5, CR was not achieved, and vaginal recurrence, which was located above the vaginal stump, had re-growth at 7.4 months after re-irradiation with ISBT, and led to the formation of vesico-vaginal and recto-vaginal fistulas at 2 months after re-growth of vaginal recurrence. The total EQD2 for D0.1cc of the bladder and rectum were 141.1 Gy and 94.2 Gy, respectively. These two patients were managed by colostomy due to the fistulas. At the time of analysis, four of the six patients were alive. Grade 2 late rectal complications occurred in two patients (patient number 2 and patient number 3; 33.3%). In particular, one patient received iron for mild anemia secondary to a persistent small amount of rectal bleeding, while the other patient underwent argon plasma coagulation for mild rectal bleeding that did not cause anemia. No late complications of grade ≥ 3 were observed in four alive patients. Grade 4 complications were observed in patients number 1 and 5 who had local recurrence infiltrating to adjacent organs (i.e., the bladder and rectum) after re-irradiation with ISBT.
Table 3
Outcomes and adverse events
Discussion
For vaginal recurrence in uterine cancer patients without previous pelvic irradiation, definitive radiotherapy (including brachytherapy) has been the standard treatment, with a good local control rate of approximately 95% [10-12]. In guidelines reported by the American Brachytherapy Society (ABS), the use of ISBT was recommended for vaginal tumors > 5 mm to deliver a sufficient dose to tumor while sparing OARs [13]. However, there is no standard treatment for patients with vaginal recurrence who have undergone pelvic irradiation previously.
Chemotherapy is not a definitive treatment option for gynecological cancer patients with pelvic recurrence primarily treated with surgery [14, 15]. Furthermore, the response rates of chemotherapy for recurrence in an irradiated field (30-33%) were lower than those for recurrence outside an irradiated field (60-75%) [16-18]. Additionally, it has been reported that pelvic exenteration leads to overall survival rates of 26.7-65.8% in cervical cancer patients with local recurrence after radiotherapy [19-22]. Shingleton et al. showed that pelvic control after pelvic exenteration was achieved in 99 (69.2%) of the 143 cervical cancer patients with local recurrence after pelvic irradiation [22]. However, mortality and complications caused by pelvic exenteration were very high (4.8-6.3% and 56.9-65.3%, respectively) [19, 20, 22].
Recent reports have shown that re-irradiation with ISBT could lead to 2-year survival, with local control rates of 54.7-78.0% and 50.0-71.4%, respectively, in gynecological cancer patients who have received pelvic irradiation previously. The incidence rate of grade ≥ 3 late complications, including bowel obstruction, recto-vaginal fistula, and vesico-vaginal fistula was 7.7-25.0% [4-6, 23]. When comparing to pelvic exenteration, re-irradiation with ISBT could also achieve a lower incidence of grade ≥ 3 late complications. Xiang et al. reported their results of re-irradiation with ISBT for vaginal recurrence after definitive radiotherapy, with EBRT and brachytherapy, in cervical cancer patients [23]. Other recent reports have included patients receiving definitive radiotherapy and those treated with post-operative radiotherapy as initial treatment [5, 6]. However, few studies have included only patients with vaginal recurrence after post-operative pelvic irradiation [24]. In our study, four (66.7%) of the six patients who previously received post-operative pelvic irradiation achieved local control, and the median survival period was 53.3 months (range, 17.7-71.8 months). In the remaining two patients with local recurrence, fistulas were observed after re-irradiation with ISBT, and the total EQD2 for D0.1cc of the bladder and rectum were > 140 Gy and > 91 Gy, respectively, which was extremely high. Therefore, there was a possibility that the fistulas were caused by radiotherapy. However, in both patients, local recurrences were diagnosed by cytological analysis after re-irradiation with ISBT (one patient; CR not achieved), and CT revealed that the fistulas had been formed by growing recurrences. Thus, grade ≥ 3 late complications were not observed in four alive patients, who achieved local control after re-irradiation with ISBT, at the time of analysis.
A prospective multi-institutional European study on MRI-guided brachytherapy in patients with locally advanced cervical cancer showed that grade 1, 2, 3, and 4 late rectal complications were observed in 20.1%, 6.0%, 1.6%, and 0.1% of the patients, respectively. EQD2 for D2cc for a 10% probability of overall rectal grade ≥ 2 toxicity was 69.5 Gy, with more severe rectal complications and higher frequencies occurring with a D2cc value of ≥ 75 Gy [25]. Liu et al. reported no serious complications in cervical cancer patients treated with ISBT for vaginal recurrence after post-operative pelvic irradiation with a total dose of 45 Gy in 25 fractions, although the observation period was short [24]. In their study, mean total EQD2 for the bladder, sigmoid, and rectum D2cc for dose summation of ISBT and post-operative pelvic irradiation were 85.6 ±5.8 Gy, 69.6 ±5.9 Gy, and 71.6 ±6.4 Gy, respectively. In our study, the mean total EQD2 for the bladder, sigmoid, and rectum D2cc were 92.1 ±20.5 Gy, 50.4 ±19.4 Gy, and 81.6 ±10.9 Gy, respectively. The total EQD2 for D2cc for the bladder and rectum in four and five of the six patients, respectively, exceeded the dose recommended by the ABS (bladder, ≤ 90 Gy; rectum and sigmoid, ≤ 75 Gy) [13]. In two patients, who were managed by colostomy due to fistula caused by local recurrence infiltrating to adjacent organs after re-irradiation with ISBT, grade 4 late complications were observed. However, in these two patients, EQD2 of D0.1cc for the bladder and rectum were > 140 Gy and > 91 Gy, respectively. Therefore, the possibility of fistula caused by radiation could not be ruled out. At least, in four alive patients without local recurrence after re-irradiation with ISBT, it could be mentioned that grade ≥ 3 late complications were not observed during analysis (median follow-up duration, 64.4 months; range, 47.4-71.8 months). Ang et al. suggested the possibility that occult injuries induced by irradiation in rhesus monkeys substantially resolved within 2 years after radiotherapy [26]. In our study, a break between the completion of pelvic irradiation and ISBT initiation (median, 24.1 months; range, 9.0-104.5 months) may be associated with the low incidence of severe late complications.
In patients treated with pelvic exenteration for pelvic recurrence after pelvic irradiation, poor survival was associated with large recurrent tumors (≥ 3 cm), recurrence with pelvic wall infiltration, and short interval between completion of initial treatment and pelvic recurrence (≤ 1 year) [20-22]. Moreover, in patients undergoing re-irradiation with ISBT for pelvic recurrence after pelvic irradiation, a large recurrent tumor (≥ 4 cm) was a poor prognostic factor for survival and local control, and a short interval between the completion of initial treatment and pelvic recurrence (≤ 6 months) was associated with poor survival [5, 23]. In our study, local recurrence and subsequent death were observed in two patients; one had vaginal recurrence with a large tumor (4.5 cm) and pelvic wall infiltration, while the other had a large tumor (6.3 cm). The remaining four patients without local recurrence at the time of analysis had small vaginal recurrence < 4.0 cm without pelvic wall infiltration. These results might encourage regular gynecological examination for early detection of vaginal recurrence and consideration of re-irradiation with ISBT as the first-line treatment for vaginal recurrence after post-operative pelvic irradiation.
The limitations of our study included its’ retrospective design and small group of the patients. Moreover, bias was present in the patients’ clinical backgrounds. Thus, further multicenter studies that involve a larger patient population with uniform clinical backgrounds are required to verify our findings. However, the outcomes of this study suggested that re-irradiation with ISBT could be a treatment option for small vaginal recurrence after post-operative pelvic irradiation.
Conclusions
Re-irradiation with ISBT achieved good local control and a low-rate of late complications after post-operative irradiation among patients with small vaginal recurrence < 4.0 cm without pelvic wall infiltration. Although the total doses of OARs, which were delivered by post-operative pelvic irradiation and ISBT, exceeded the doses recommended by the ABS for almost all patients, a long interval between completion of pelvic irradiation and ISBT initiation may be associated with a low incidence of severe late complications. Re-irradiation with ISBT may be an effective treatment strategy for gynecological cancer patients with small vaginal recurrence after post-operative pelvic irradiation.



