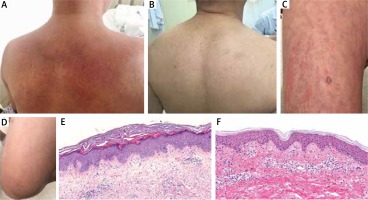
A 54-year-old man admitted to our department presented with recurrent fever for 18 months with arthralgia and skin rash in May 2018. The patient appeared to have run a fever with no obvious cause since December 2016. His highest body temperature was 41°C, accompanied by arthralgia and myalgia, the laboratory examination showed the following: white blood cells (WBC) 12.59 × 109/l (3.5–9.5 × 109/l) with neutrophils 75.3% (40–75%); C-reactive protein (CRP) 65.8 mg/l (0–8 mg/l), erythrocyte sedimentation rate (ESR) 87 mm/h (0–15 mm/h), negative for cytomegalovirus (CMV), EBV-DNA, TORCH, ANA, ds-DNA, SSA, SSB, anti-Sm, RF, ANCA, mycoplasma, and chlamydia. Procalcitonin (PCT) 0.11 ng/ml (< 0.05 ng/ml). Pulmonary computed tomography (CT) showed nodules and chordal shadows in the right upper lung with partial calcification and mediastinal lymphadenopathy. T-spot result showed 247.4 pg/ml (< 14 pg/ml), but was negative for acid-fast staining of sputum. The patient had been in remission from pulmonary tuberculosis history for 30 years. The local hospital diagnosed him with pulmonary tuberculosis, treated him with RHZE and intermittent non-steroid anti-inflammatory drugs (NSAIDs), but his body temperature continued to fluctuate, then he had 30 mg glucocorticoids per day added, after which the fever subsided. After 3 months, glucocorticoid and anti-tuberculosis drug treatment was stopped, the patient self-administered Chinese traditional herbal therapy, but his symptoms persisted, with body temperature fluctuations at 37–37.5°C, and long-term fatigue, lost 5–10 kg in 1 year. His fever returned with no obvious cause on 21 May 2018, at which time his body temperature fluctuated at up to 38.5–39°C, accompanied by pharyngalgia and arthralgia, the polyarthritis involving bilateral hands proximal interphalangeal joints. At his physical examination, the patient presented with typical evanescent salmon-pink maculopapular skin rash and atypical persistent pruritic linear skin eruption on his chest, upper back, shoulders, arms, and thighs (Figures 1 A, C). Lymph nodes in the neck and axilla became enlarged. The laboratory examination results are given in Table 1, including Ferritin of 1918 ng/ml (30–400 ng/ml). Both typical and atypical skin rash was biopsied. For typical evanescent rashes, the histopathologic results indicated that the epidermis was normal, a mix of inflammatory cells, including neutrophils and eosinophils, shows slight infiltration into the superficial dermis and mid perivascular area (Figure 1 F). For atypical persistent skin eruption, scattered and aggregated necrotic keratinocytes were distributed in the upper third layer of epidermis and keratin whorls in the stratum corneum, mixed lymphocytes and neutrophils infiltrated the papillary and mid dermis with slight dermal papilla fibrosis (Figure 1 E). Bone marrow biopsy excluded the presence of haematological malignancy. He met the adult onset Still’s disease (AOSD) criterion, and initially received Nimesulide 100 mg twice per day and colchicine 0.5 mg twice per day. Ten days later, the patient had a mild fever (37–38°C), the pain of his throat was relieved, and the skin rashes and the arthritis improved slightly. We then ceased Nimesulide and continued 0.5 mg colchicine twice per day. After 2 months, the skin rash had subsided completely and the arthralgia disappeared (Figures 1 B, D). Ferritin, ESR, CRP, leukocyte, and neutrophils returned to normal. One month later, we withdrew colchicine, and there has been no recurrence of the disease for nearly 2 years.
Table 1
The laboratory examinations of the patient
Figure 1
A, C – Atypical persistent pruritic eruption: red, violaceous, or brownish linear urticarial oedematous erythematous plaques on the back and legs. B, D – 3 months later after treatment with colchicine. E – Scattered and aggregated necrotic keratinocytes were distributed in the upper third layer of epidermis and stratum corneum. Mixed lymphocytes and neutrophils infiltrated the papillary and mid dermis with slight dermal papilla fibrosis. F – Typical evanescent rash, a mix of inflammatory cells, including neutrophils and eosinophils, shows slight infiltration into the superficial dermis and mid perivascular area
Adult onset Still’s disease is a rare multisystemic inflammatory disease, which is characterized by spiking fever, recurrent polyarthralgia, evanescent eruption associated with neutrophils, hepatosplenomegaly, and lymphadenopathy [1, 2]. Atypical persistent skin eruptions (APSEs) as the new skin manifestation of AOSD have been documented [3, 4]. Biopsy specimens from such lesions also revealed characteristic changes, the unique pathological feature that necrotic keratinocytes in the upper third layer of the epidermis and keratin whorls in the stratum corneum are the most important clues to establish the correct diagnosis. Our latest study shows that interleukin-1β (IL-1β) and interferon-γ (IFN-γ) were specifically expressed in the upper third layer of the epidermis and were juxtaposed to the loci of the necrotic keratinocytes. It implies that IL-1β and IFN-γ might be involved in the formation of APSE [5]. We noticed that general physicians and rheumatologists had misdiagnosed this patient for 18 months, all of them neglected the characteristic of skin eruptions, not to speak of the biopsy. It is worth noting the importance of the rash and its histopathological characteristics of AOSD.
Scientists believe that the key to the pathogenesis of AOSD is the intense activation of innate immune cells and overproduction of several pro-inflammatory cytokines including IL-1, IL-6 and IL-18. The treatments for AOSD have usually been empirical. Oral corticosteroids, non-steroidal anti-inflammatory drugs, disease modifying-anti-rheumatic drugs, sulfasalazine, methotrexate, cyclosporine, minocycline, azathioprine, and biological agents such as IL-6 blocker, IL-1 blocker have been used to treat this rare disease [1]. It has been reported that skin rash is a negative predictor of anakinra withdrawal related to sustained remission, which could be explained by IL-1β overproduction in AOSD patients with skin lesions [6].
Colchicine is an inexpensive and widely-available drug. It can be used in the treatment of a range of autoinflammatory diseases like gouty arthritis and, classically, familial Mediterranean fever. Colchicine could impair neutrophil recruitment by decreasing neutrophil L-selection expression and modulating E-selection expression on the surface of endothelial cells. This leads to subsequent downregulation of multiple inflammatory pathways and to neutrophil activation [7]. It can suppress the NLRP3 inflammasome [3] and the activation of caspase-1 [8], thereby blocking the conversion of pro-IL-1β to active IL-1β, reducing the release of IL-1β and a range of other interleukins, including IL-6, TNF-α [8]. Considering this mechanism, it could be a good choice for AOSD treatment especially for patients with APSEs. Asano et al. have reported a case that a biologic agent against AOSD patient was successfully treated by colchicine [9]. There has also been another case that an AOSD patient with mutations of the MEFV gene was partially responsive to colchicine [10].
Through our patient’s history of tuberculosis, the usage of glucocorticoids, immunosuppressive agents, and biological agents involved different levels of contraindications. For the economical reason, colchicine is the first choice for our patient, and, not surprised, his clinical manifestations, such as fever, arthropathy, and skin rash, were gradually improved. Many of his laboratory findings, specifically serum levels of ESR, CRP, IL-1β, tumor necrosis factor-α (TNF-α), and ferritin, became normal in 3 months. We believe that colchicine can be considered as a cheap therapeutic option for AOSD, especially for patients who cannot be treated with glucocorticoids or immunosuppressive agents, because of complications like tuberculosis or HBV infection, and those cannot afford biological agents such as IL-1β blocker.









