A 30-year-old pregnant woman developed erythema with pruritus on the trunk and limbs 1 month before the first visit to the previous clinic. The initial diagnosis was prurigo and she was treated with a topical steroid, which did not relieve her symptoms. Thereafter, blisters appeared on the hands and feet and spread to the entire body, and she was referred to our hospital.
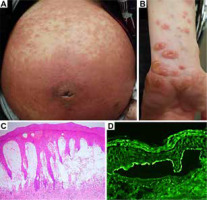
The patient had marked annular erythema on the face, diffuse palpable erythema on the trunk (Figure 1 A), and tense bullae at the peripheries of erythema on the limbs (Figure 1 B). The results of laboratory examinations were within normal limits, except for leukocytosis with eosinophilia (white blood cell count, 19.86 × 109/l; eosinophil count, 2.68 × 109/l). The chemiluminescent enzyme immunoassay for BP180 was positive (671 U/ml, cut-off < 20). Histopathology of a biopsy from skin lesion with tense bulla demonstrated subepidermal bullae and infiltrates of eosinophils and neutrophils (Figure 1 C). Direct immunofluorescence (IF) revealed linear deposits of IgG (Figure 1 D) and C3 along the basement membrane zone (BMZ). Indirect IF showed IgG anti-BMZ antibodies, which reacted with the epidermal side of 1 mol/l NaCl-split skin. Complement IF of split skin for C3 was also positive. Immunoblotting of the normal human epidermal extract showed that IgG antibodies in the patient serum reacted with BP180, but not BP230. Patient serum showed positive IgG reactivity with the recombinant protein (RP) of the BP180 NC16a domain, but not with the RP of the BP180 C-terminal domain. Immunoblotting of concentrated culture supernatant of HaCaT cells demonstrated that IgG antibodies reacted with the 120-kDa LAD-1.
Figure 1
Clinical and histopathological features and results of immunofluorescence. A, B – Diffuse and palpable erythema on the abdomen (A) and tense bullae 8 mm in size on the peripheries of exudative erythema on the limbs (B). C – Histopathology for skin biopsy showed a subepidermal bulla (haematoxylin-eosin, original magnification ×40). D – Direct immunofluorescence showed a linear deposition of IgG along the epidermal side of the blister
A diagnosis of pemphigoid gestationis was established on the basis of these histopathological and immunological findings. Treatment was started with oral prednisolone at a dose of 30 mg/day, which was then increased to 50 mg/day. However, her condition did not improve. Concomitant plasmapheresis was considered, but she declined the treatment. Considering the adverse effect of high-dose prednisolone on the mother and foetus, the dose of prednisolone was reduced to 40 mg/day and the infant was delivered at 38 weeks of gestation by forceps. Unexpectedly, her skin rash did not improve after delivery. Therefore, the prednisolone dose was increased to 60 mg/day, and intravenous immunoglobulin (IVIG) was administered. Then, the prednisolone dose was reduced, using the skin rash and anti-BP180 antibody titre as indicators, and a second course of IVIG was administered. The anti-BP180 antibody titre, which reached a maximum of 2,820 U/ml during pregnancy (approximately 33 weeks of gestation), was reduced to 104 U/ml. Eventually, the patient went into remission with a combination of mizoribine and a third course of IVIG.
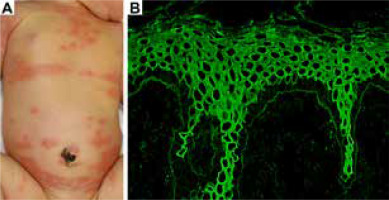
At the age of 3 days, the infant developed erythema and blisters similar to those of the mother (Figure 2 A). Indirect IF of the umbilical cord blood serum showed IgG reactivity with BMZ (Figure 2 B). The skin condition in the infant resolved approximately 1 week after the topical application of hydrocortisone butyrate. The rash did not recur.
Figure 2
Clinical features in the neonate and the result of indirect immunofluorescence for umbilical cord blood serum. A – At the age of 3 days, the infant developed erythema and blisters similar to those of the mother. B – Indirect immunofluorescence of umbilical cord blood serum shows IgG reactivity along the basement membrane zone (magnification ×100)
Pemphigoid gestationis occurs in approximately 1 in 60,000 pregnancies, and it commonly develops during mid-to-late pregnancy. The clinical features of pemphigoid gestationis resemble bullous pemphigoid and the causative antibodies react with the BP180 NC16a domain, similar to bullous pemphigoid antibodies. BP180 produced by chorion derived from the foetus is thought to be exposed to the maternal immune system and induce antibody production [1].
Skin lesions usually resolve spontaneously at 1 week to 1 month postpartum, but are prolonged after delivery or develop during the puerperal period in some cases. Our case also showed a prolonged and intractable disease course. The reason why skin lesions are prolonged or worsen after delivery and require high-dose prednisolone and additional treatment in some pemphigoid gestationis cases is unknown. One possibility is that more amount of BP180 protein is released into the maternal circulation from weakened chorion during pregnancy and at delivery [2].
The standard treatment for pemphigoid gestationis is similar to that for bullous pemphigoid and systemic steroid administration is the mainstay of the therapy. Because high-dose steroid treatment during pregnancy can affect the foetus and the condition may spontaneously resolve after delivery, physicians are often reluctant to treat pemphigoid gestationis with the same dose of steroids as that used for bullous pemphigoid. Currently, there is no established protocol for the treatment of pemphigoid gestationis, and a number of different therapies, including various immunosuppressants, diaminodiphenyl sulfone, IVIG, plasmapheresis, and steroid pulse therapy are used [3–9].
Systemic steroids (over 20 mg/day) for 4 weeks or longer must be administered with caution during pregnancy, because high-dose steroids increase the risk of gestational diabetes and preterm birth [10]. To reduce these risks, adjunctive therapies, which are also safe during pregnancy, are recommended to be used for the early reduction of steroids. These therapies might also help to reduce prolonged conditions at postpartum. IVIG is commonly administered during pregnancy and in the postpartum period. A few pemphigoid gestationis cases were reported to be treated with plasmapheresis [11], although plasmapheresis requires the monitoring of hemodynamics of the placenta. Selective plasmapheresis, which efficiently removes IgG antibodies, is assumed to have a lower risk with respect to pregnancy than conventional plasmapheresis.
Although our case was treated with up to 50 mg/day of systemic prednisolone prior to delivery, the symptoms were prolonged after delivery. Although the reason for this intractable disease course is currently unknown, safer additional treatments, such as IVIG and plasmapheresis, might reduce the disease severity during pregnancy, which could avoid the prolonged condition.







