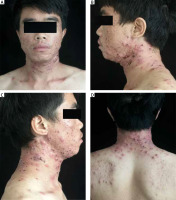
A 30-year-old man presented to our hospital with a 2-year history of massive inflammatory, nodulocystic, and painful lesions as well as an aggravation for 2 months. Clinical examinations revealed that there were massive red papules, pustules, nodules, and cysts scattered over the skin of his whole cheeks and neck, and part of his shoulder and back. Most of the pustules were filled with white secretion, and some were damaged with crust (Figure 1). A rectal temperature was 37°C.
Figure 1
A – The anterior view of a 30-year-old man with acne conglobata. B – The left side view of a 30-year-old man with acne conglobata. C – The right side view of a 30-year-old man with acne conglobata. D – The posterior view of a 30-year-old man with acne conglobata
He has taken oral isotretinoin (20 mg daily for 3 months) and nadifloxacin ointment for the therapy, but there was no significant improvement on him and the number of his skin lesions has increased.
During the hospitalization, he was diagnosed with acne conglobata through his physical examination. In his laboratory examinations, the white blood cell count (15.15 × 109/l) and neutrophil count (10.60 × 109/l) in blood routine tests were increased. The erythrocyte sedimentation rate (ESR) (40 mm/h) and C-reactive protein (CRP) (27.60 mg/l) were also significantly elevated (Figure 2).
Figure 2
Colony morphologies of C. avidum of the facial pustules of this patient on Schaedler agar plates after incubation
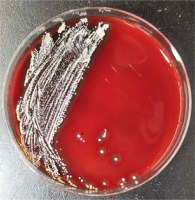
We got the secretion sample for microbiological detection. However, no bacteria and fungi were found in the secretions in the microscopic examination. The Gram stain and acid-fast staining were also negative. But in the secretion culture of his facial secretions, the Cutibacterium (formerly Propionibacterium) avidum (C. avidum) colonies were found in the Brucella blood agar plate.
Then we used a daily dose of intravenous moxifloxacin hydrochloride (400 mg), oral isotretinoin (10 mg three times), and topical use of mupirocin ointment and nadifloxacin ointment. After a week of therapy, the skin lesions improved and he was discharged with the therapy of a daily dose of oral amoxicillin (1.5 g), isotretinoin (10 mg three times), and aforementioned external medicines. Finally, this patient had complete resolution of his symptoms at 4 months’ follow-up.
Acne conglobata is a rare and severe type of acne with interconnecting inflammatory and grouped nodules, cysts, and abscesses [1]. Acne conglobata is often resistant to conventional therapies for acne and heals with disfiguring scarring, resulting in a big impact on the mental health and patients’ quality of life [1]. Although this patient had an elevated white blood cell count, ESR and CRP, the 2-year medical history as well as numerous persistent polyporus comedones and non-inflammatory cysts suggested that the diagnosis was dominated by acne conglobata rather than acne fulminans [2]. In decades, microorganisms on the skin surface have been considered to be one important factor of acne conglobata [1–3]. According to previous studies, C. acnes is believed to be involved in the chronic inflammatory state, leading to the presence of acne vulgaris [2]. However, it is not the proliferation of C. acnes but C. avidum that has been observed in the facial secretion of this patient. Thus, we supposed that this case may be caused by C. avidum.
C. avidum and C. acnes are prevalent species colonized on the skin tissues in both healthy people and patients with acne. C. acnes tends to reside in the skin regions with excessive sebum and is the main pathogen of acne. While C. avidum is a type of human cutaneous commensals in maintaining skin homeostasis and usually colonized in regions with eccrine sweat glands, such as nares, axilla, groin, and the perianal region [4]. It is a low pathogenic form but can be an opportunistic deep infection, instrumentation procedures, and immunodeficiency [4]. In previous reports, the C. avidum infections usually occurred in patients after the surgery or instrumentation procedures, such as breast implant infection and skin abscesses [4].
Interestingly, we firstly reported a rare case of acne conglobata caused by C. avidum infection. To date, there have been few reports in the literature or researches about the pathogenetic role of C. avidum in acne. We speculate that the possible pathogenic mechanisms of C. avidum in the acne conglobata are as follows: Firstly, C. avidum belongs to cutaneous commensals and the trauma, surgery, device, as well as immunosuppressant intake can be the contributors of C. avidum infections in previous evidence. Here, we speculated that the destruction of the skin barrier and microenvironment may promote the proliferation of C. avidum in this patient. Besides, C. avidum is capable of hydrolysing triacylglycerols and thereby increasing short chain fatty acids, which are thought to be the key factors in promoting inflammation in acne [5]. In addition, the formation of the biofilm in C. acnes leads to the amplification of inflammation and activation of innate immune in acne. Thus, the biofilm formation in C. avidum which can contribute to the persistent infections may play a role in the pathogenesis of acne conglobata [3]. Moreover, the C. avidum have high levels of catalase activity that cause oxidative stress in the pathogenesis of acne [4, 6]. What is more, C. avidum has more tryptamine-like activity and the tryptamine produced by C. avidum may lead to sebaceous inflammation [4]. Moreover, some bioactive substances produced by C. avidum, such as neuraminidase, propionicins, and lecithinase may also participate in the progression of acne conglobata [4]. Although C. avidum is a potential pathogen of acne conglobate, more attention and investigations about the functions of C. avidum are further needed.
As for the antimicrobial susceptibility of C. avidum, the β-lactams, rifampin, fluoroquinolones, and macrolides have a good antibacterial activity, whereas the erythromycin and clindamycin have low activity [4]. Recently, clarithromycin, and ciprofloxacin resistance were observed in C. avidum strains [7, 8]. In previous studies, benzoyl peroxide may be anti-inflammatory and antibacterial treatment and can prevent the occurrence of antibiotic resistance by resistant mutant selection [5, 9].
In conclusion, this is the first case presented as acne conglobata caused by C. avidum infection. It is necessary for the dermatologist to confirm the microorganism isolated from the facial secretions of patients with acne conglobate and select the sensitive antibiotic treatment. The good news is that C. avidum remains sensitive to most antibiotics so far. However, since there are few studies on the C. avidum infections in acne conglobate, more researches on C. avidum are still urgently needed.









