Introduction
Chronic venous disorders (CVD) cover a wide range of pathologies ranging from small changes in the nature of reticular veins and telangiectasia not significantly affecting the quality of life of patients, to extensive venous ulcers leading to severe disability. They are associated with a great medical and socioeconomic impact [1]. It is estimated that in Poland they affect about 47% of females and 37% of males [2]. They are a significant problem, not only from a medical point of view, yet also economical. CVD accounts for around 362,000 sick leaves per year, representing 6.4 millions of work days lost. It is estimated that CVD constitutes 1–3% of all health expenses [3]. Treatment of venous ulcers only in the UK costs 400–600 million pounds a year, and in the USA over 1 billion dollars a year [4]. The total social costs in the UK, France and Germany are estimated at over one billion dollars in each of these countries [5]. Unfortunately, there are no specific reports on costs of treatment of CVD and associated conditions in Poland.
Although a holistic pathophysiology of CVD remains unknown, venous skin changes and ulceration are predominant manifestations of CVD. Telangiectasias and varicose veins are reported to be extremely common, with a prevalence of about 80% and 20–64%, respectively [6]. Other characteristic symptoms include oedema and skin changes, hyperpigmentation, eczema, atrophie blanche and lipodermatosclerosis [7–9].
Aetiology of the disease is multifactorial and results from complex interactions of genetic and environmental factors. Risk factors include age, gender, obesity, family history and ethnicity [10–12]. Older people are affected more often, with a higher prevalence among females [13]. A body mass index (BMI) greater than 30 increases the risk for CVD significantly, and so does the positive family history [14].
Main pathomechanisms that lead to CVD include reflux, obstruction or combination of both [15]. Apart from those factors, also failure of calf and foot muscles (by decreased mobility or neuromuscular problems) can lead to inadequate venous return. This obstruction can lead to venous claudication, described by the patient as bursting pain while walking, that is relieved by rest or elevation of legs.
Primary symptoms as heaviness and aching of legs, especially during prolonged standing, can worsen the quality of everyday life [16]. This may consequently lead to aggravation of daily activities and professional work. Nevertheless, the early symptoms can be alleviated by regular exercise, leg elevation and avoidance of prolonged steady standing.
Colour Doppler ultrasound imagining, also known as triplex US, is currently the gold standard test for diagnosis and following up CVD [17]. It enables to study both the wall and the lumen of deep and superficial veins of lower extremities and analysis of venous flow. It should be done systematically in comparative, bilateral way. Computed tomography of the lower extremities is widely used to study venopulmonary thromboembolism. Nevertheless it is not generally used in diagnosis of the superficial venous system, yet may be complementary to Doppler ultrasound scan in particular cases with an unexpected anatomic source.
The gold standard treatment option is physical therapy, which includes compression therapy and vascular exercises programmes, which are recommended to be started early. Compression therapy is the most efficient and cost-effective basic therapy for CVD [18]. Due to better understanding of CVD’s pathomechanisms and pathophysiology of venous ulceration, new treatment options are constantly being discovered and implemented [19]. In all cases the treatment is directed toward prevention of the retrograde blood flow and venous pooling. New types of wound dressing, surgical modalities, matrix materials and growth factors should all be used a supplement to compressive therapy.
Aim
The European guidelines emphasize the lack of data on the percentage of population that have the first symptoms of CVD. The purpose of the study was to determine the level of public awareness about CVD and analysis of CVD symptoms frequency in a randomly selected group of patients.
Material and method
One hundred and seventy-five patients took part in the research. The mean age was 44, median 41 years old. The group consisted of randomly selected patients who participated in a preventive assessment of nevi.
The participants were informed about the purpose of research and the way the test was carried out. Patients were informed about the purpose of our research and its objectives. They could ask questions concerning the questionnaire and if they did not fully understand any terms, the physician was explaining those.
Patient participation in the study was voluntary, all study participants were informed of its details and signed informed consent for subsequent use of data for educational and commercial purposes. All patients had the right to refuse to participate in the study.
Patients more than 18 years old, from whom we received informed consent were included in our research. Whereas patients who were unable to understand the questions in our questionnaire and from whom we could not obtain informed consent were excluded.
The examination was performed under the supervision of a specialist dermatologist who assessed the occurrence of the first signs and symptoms of CVD. A questionnaire, which consisted of two sections has been made, one part to be completed by the patient and the other to be completed by the doctor. The patient’s part contained detailed questions on patient’s knowledge on CVD. It begins with yes-no questions if the patient knows the disease and thinks that it is common. Then it proceeds to complex questions: if the patient can indicate from listed signs and symptoms (leg fatigue, leg oedema, coldness of legs, leg ulcers, varicose veins, vomiting, easy fatigability) the ones that refer to CVD. The same applied to methods of prophylaxis and treatment. Patients could choose from compressive stockings, by-pass surgery, phlebectomy, sclerotherapy, ulcer treatment and dietary supplements. The last complex task was to point the procedure through which diagnosis of CVD can be made. The choice consisted of X-ray, USG duplex scan, computed tomography and complete blood count.
The other part’s aim was to determine if the patient has the first symptoms of CVD and is aware of this. To both rule out situations when the patient reports false symptoms or does not report them, we created a simple table to be completed by the doctor. The table consisted of main primary CVD signs and symptoms (leg oedema, telangiectasias, hemosiderosis, lipodermatosclerosis and varicose veins) to be examined by the doctor.
We received an approval of the Independent Bioethics Commission for Research at the Medical University of Gdansk to conduct our research.
Statistical analysis
The obtained test results were subjected to statistical analysis using the Statistica 1.2 program, determining the mean values, median and standard deviation. Compatibility with the normal distribution was determined using the Shapiro-Wilk test. The probability density function was 1. In the case of compliance with the normal distribution, a t-test for independent samples was used. The t test values for women and men who know/do not know and are aware that the disease is common/uncommon were around 0.0001. Statistically significant results were those for which p < 0.05. Those results directly imply that there were statistically relevant differences between men and women.
Results
Median age of the patients was 41, whereas mean age 44 ±15.4 years. One hundred and eight (61.75%) women and 67 (38.28%) men took part in the research, which gives 175 people in total.
62.85% know what CVD is, 72.27% of whom are women, which means that 78.70% of all women tested and only 37.31% of men tested claim to know what CVD is. 73.14% of people tested claimed that CVD is a common disease, 68.75 % of whom were women, which means that only 59.7% of men, and 81.48% of women claim it is common.
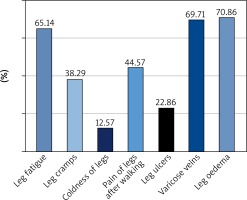
As subjective symptoms and objective signs of CVD they could mainly associate oedema (70.86%), leg fatigue (65.14%) and varicose veins (69.71%) (Figure 1).
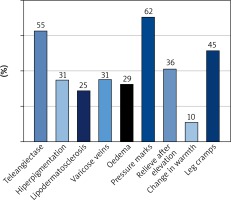
As it comes to subjective symptoms and objective signs of CVD (Figure 2), 54.86 of patients claimed to have telangiectasias, 61.71% have pressure marks (impressions by socks in the evening), 30.86% have varicose veins.
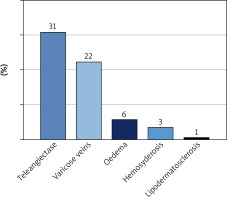
Dermatologists confirmed varicose veins in 15.43% of the patients, while 7.43% of the patients did not recognize any of the symptoms recognized by the physician. On the other hand, physicians ruled out varicose veins in 15.43% of the patients. This directly implies that further measures should be undertaken to spread the knowledge on CVD among the society.
The examination is a key to identifying early symptoms of CVD. Every physician should be able to notice the visible signs and take the medical history on CVD symptoms. Most of the early CVD symptoms and objective signs, which physicians recognized in the randomly selected group of patients tested (Figure 3) were telangiectasias (30.86% of the patients) and varicose veins (22.28% of the patients).
Discussion
Physicians should regularly and thoroughly examine both legs. When strong evidence of CVD presence is visible, abdomen should be examined for venous collaterals presence as well. It should be an ultimate aim to share the knowledge on CVD among the society and encourage people to undergo further diagnostics. It may be easily done by general practitioners. The ultimate purpose of those measures would be to avoid high-cost treatment of ulcers and varicose veins.
According to 64.57% of the patients, duplex ultrasound (DUS) examination is currently a gold standard for diagnosis of CVD [20]. It is based on a combination of ultrasound imaging and pulsed Doppler wave. Both anatomy and hemodynamic features of the venous system can be examined. Owing to DUS invention, invasive techniques such as phlebography lost their applicability. Currently phlebography is mostly used to evaluate congenital vein problems or to identify a vein for arterial bypass grafting.
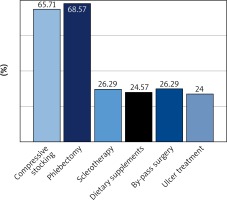
As seen in Figure 4, most of the patients recognize phlebectomy (68.57%) and use of compression stockings (65.71%) as main therapeutic measures for CVD. This is an uplifting conclusion since compression therapy is a significant part of conservative treatment. It is widely used because of its accessibility, non-invasiveness and high efficacy in increasing venous flow.
Nevertheless, 13.71% of the patients claim that they do not know any therapeutic measures for CVD. 24.57% of the patients stated that dietary supplementation is effective for CVD treatment. There are no evidence-based premises to support this thesis. What is more, 26.29% of them point out by-pass grafting. This is obviously not routinely recommended therapeutic measure for CVD.
Furthermore, surgery called phlebectomy, which was recognized by 68.57% of the patients, is a safe and effective procedure that may be performed under local anaesthesia [21]. It consists of removal or avulsion of varicose veins through small incisions.
Unfortunately only 26.29% of the patients identified sclerotherapy as a highly effective measure to treat CVD [22]. Sclerotherapy, which involves injection of liquid or foam agents to dilated veins. This leads to damage of the endothelium and consequently ablation of the veins. It is an easily repeatable treatment, which has good long-term results [23]. Not to mention that it is less invasive than surgery.
Leg elevation, physiotherapy and leg massage are highly recommended to all patients with CVD as they significantly reduce venous stasis and leg oedema.
As ulceration occurs, compressive dressings with povidone iodine or hydrocolloid dressings should be applied in order to achieve wound healing [24]. Twenty-four percent of the patients recognised treatment of ulcers as a significant aid to manage life-threatening consequences of CVD.
Conclusions
Although the study revealed that most of the people are not acquainted with CVD and its first symptoms, the majority of tested patients presented symptoms of initial stages of CVD. It should be an ultimate aim to spread the knowledge on CVD among the society and encourage people to undergo further diagnostics. The ultimate purpose of those measures would be to avoid high-cost treatment of ulcers and varicose veins.