Introduction
Despite the constant progress in medicine, lymphoedema still remains a serious diagnostic and therapeutic issue, reducing the quality of life of patients themselves and of their families [1]. The clinical picture of this condition develops in consequence of congenital or acquired lymph transport disorders in underdeveloped or damaged vessels and lymph nodes [2]. Lymph retention results in functional disorders and the development of unfavourable structural changes in numerous systems, comprising – without limitations – skin, subcutaneous tissue, fascia, as well as the immune, vascular, and nervous system. The clinical picture is dominated by progressing deformations of limbs, resulting in difficulty in attempting physical activity and the reduction of the scope thereof, as well as deterioration of the patient’s general health condition, with such ailments as obesity, trophic changes of skin, chronic leg ulcers, and recurrent inflammations. Distant consequences include deterioration of patients’ social situation, with their growing social alienation. Additionally, chronically incurred treatment costs and limitations in terms of undertaking professional activity deteriorate the financial situation of patients and their families [1].
Considering the above deliberations, it seems clear that patients suffering from all forms of lymphoedema need to be covered by an effective treatment which would limit as much as possible the adverse consequences of this disease. Recognised patterns of an effective treatment in the form of the comprehensive antioedema therapy are available in the world literature [3, 4]. Sadly, its implementation – as the everyday practice demonstrates – encounters numerous obstacles resulting from – without limitations – flaws of the healthcare staff training system and limited availability of medical services financed from the National Healthcare Fund. Difficulty in accessing full treatment, observed in our country, raises questions concerning the usefulness of individual therapeutic techniques and of the application thereof with a 2-day weekend break in the reduction of the severity of symptoms.
Aim
The objective of this trial was to compare the results of treating patients with lymphoedema with the application of an isolated multilayer compression therapy and the comprehensive antioedema therapy.
Material and methods
The study involved a group of 34 female patients aged 50–80, treated in 2015–2016 in the Daily Rehabilitation Centre of the Palium Hospice in Poznan due to 2nd stage primary lymphoedema according to Olszewski’s classification [1].
All subjects demonstrated clinical oedema symptoms, such as thickening and fibrosis of the subcutaneous tissue, an additional Stemmer’s sign, and a 20-second compression test. Patients with a positive history suggestive of true or dissecting aneurysms of any artery, neoplastic, neurological, inflammatory, and allergic diseases, chronic venous insufficiency, renal failure requiring dialysis, neurogenic diabetic foot, unregulated or untreated hypertension, diffuse dermatological lesions, taking phlebotropic, steroid, and immunosuppressive drugs, as well as patients after any types of surgical procedures carried out on arteries, veins, or lymphatic vessels in the examined limb were excluded from the trial. In all patients included in the trial the value of the Toe Brachial Index oscillated between 0.9 and 1.1.
During the study, the therapy of comorbidities, i.e. angina pectoris, diabetes, and hypertension, was continued. No phlebotropic or diuretic therapy was introduced. All patients were advised to quit smoking completely and to increase their physical activity.
Research groups
The patients were randomly assigned to two research groups, 17 subjects in each. Patients from group 1 were subjected to multilayer bandaging. The remaining patients (group 2) were administered the comprehensive antioedema therapy, comprising the manual lymphatic drainage, the compression band therapy, and the exercise therapy. Patients from both groups were encouraged to intensify their physical activity and received instructions on appropriate skin hygiene of their affected limbs. The procedures were carried out over 10 days, with a 2-day weekend break.
Manual lymphatic drainage (MLD)
MLD was applied in patients from group 2 in the supine position, with slightly elevated lower limbs, bent at the angle of 15–20° in the knee joints. The procedure was carried out without any lubricants, in compliance with Vodder’s rules [2]. The duration of each drainage session was 60 min.
Compression therapy
In patients from both groups, the treatment was commenced with separate bandaging of individual toes with a highly stretchy bandage (Matplast by Matopat). Next, a cotton stocking was put on the limb (Tubula Cotton by Matopat) along with a synthetic lining (Matosoft synthetic by Matopat) and elastic bandages of low extensibility (Rosidal K by Lohmann&Rauscher). The foot and the shank were dressed with 4-5 8 cm- and 10 cm-wide bandages. Four 12 cm-wide bandages were applied on the thigh. The compression bandages were each time applied by experienced medical staff. The pressure of 50–70 mm Hg measured at the ankle level was obtained by means of the bandages; the pressure decreased accordingly in the proximal direction.
The bandages were maintained for 22 h. The compression was applied for 5 weekdays, with a 2-day break during the weekend. During the break in treatment, the patients put on compression stockings they had used on their own before being qualified for the trial.
Exercise therapy
Patients from group 1 were advised to undertake an active lifestyle, with a special emphasis put on walking and avoiding the sitting position.
After bandaging of the lower limbs, patients from group 2 were subjected to a controlled exercise therapy on a stationary bike or a magnetic cross trainer. The duration of the exercises and the load applied were adjusted to the current status of each patient.
All the procedures, i.e. lymphatic drainage, compression therapy, and exercise therapy were performed/supervised by the same experienced physiotherapist.
Limb volume measurement
The assessment of the therapy results employed linear measurements of the lower limb perimeter by means of a non-stretchy soft measuring tape. The limb volume was determined on the basis of measurements performed at the same levels, at 4 cm intervals, and at the same time of the day, using the formula for the volume of a frustum of a cone. Limb measurements were performed by another individual, with the blinding the manner of conducting the therapy. The measurement was carried out on the treatment onset date, prior to the therapy commencement, and on the day following the treatment completion.
Duplex-Doppler ultrasound scan of veins and arteries of lower limbs
The Doppler ultrasound scan of veins and arteries of lower limbs was performed in all the patients in order to exclude any vascular pathologies, which might reduce the effectiveness of the therapy applied (venous insufficiency) or constitute a contraindication for its implementation (advanced atherosclerotic lesions). The duplex ultrasound tests were based on linear heads with the frequency of 5–12 MHz and 5–9 MHz, apparatus Mindray DC-8. The evaluation was performed by a radiologist and a vascular surgeon with more than 10 years of diagnostic experience, in compliance with the recommendations of the Polish Society of Phlebology and the Polish Society of Vascular Surgery [5, 6].
Approval of the ethics committee
The trial was approved by the Bioethics Committee at the Poznań University of Medical Sciences (No. 270/17).
Statistical analysis
Calculations were carried out by means of Statistica 12 software by StatSoft. The adopted level of significance was α = 0.05. The result was recognised as statistically significant when p < α. In order to compare variables measured on the interval scale due to the lack of compatibility with the normal distribution or variables measured on the ordinal scale, the Kruskal-Wallis test was applied. Additionally, the Dunn’s multiple comparison test was used so as to determine any differences between which groups. In order to assess whether changes at the level of the parameters analysed caused by the therapies applied were statistically significant, for variables measured on the interval scale due to the lack of compatibility with the normal distribution or for variables measured on the ordinal scale, the Wilcox test or the Friedman test was used, respectively. The Dunn’s multiple comparison test was additionally applied in order to determine which measurements differed from each other.
Results
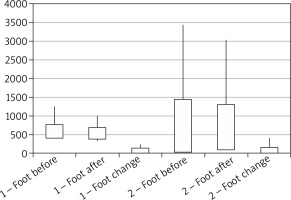
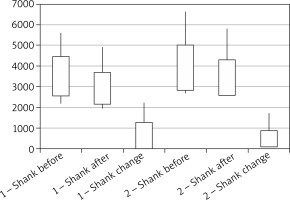
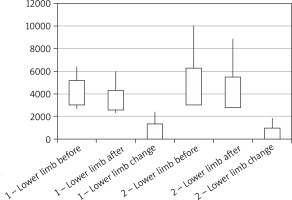
From the commencement of the trial the volume of the parts of lower limbs affected with lymphoedema was comparable in both groups and it was 4094.6 ±1056.38 ml and 4655.7 ±1634.2 ml in group 1 and group 2, respectively. The volume of feet in both groups was 590.2 ±179.2 ml and 740.9 ±698.2 ml (p = 0.62), respectively, whereas the volume of shanks was 3504.4 ±949.6 ml and 3914.8 ±1094.8 ml (p = 0.30), respectively.
After 2 weeks the volume of lower limbs was 3441.7 ±876.9 ml and 4132.6 ±1358.8 ml, respectively (p = 0.20). The volume of feet and shanks in individual groups was 529.4 ±145.5 ml and 689.1 ±607.7 ml (p = 0.34), 2921.3 ±776.9 ml and 3443.5 ±851.1 ml, respectively (p = 0.12).
In patients from both groups a significant reduction in the volume of lower limbs as a result of the therapies applied could be observed. The range of changes in terms of the volumes of lower limbs was comparable in both groups and it was 652.9 ±712.2 ml (reduction by 15.9%) and 523.1 ±448.1 ml (reduction by 11.2%), respectively (p = 0.77). In relation to feet and shanks, the oedema reduction range in individual groups amounted to 69.76 ±58.3 ml (reduction by 10.2%) and 51.78 ±99.17 ml (reduction by 6.9%) (p = 0.06), and 577.3 ±684.4 ml (reduction by 16.6%) and 471.3 ±392.8 ml (reduction by 12%) (p = 0.52) (Figures 1–3).
Figure 1
Volumes of lower limbs affected with oedema prior to and after the completion of the therapeutic cycle and the range of changes in terms of volumes of lower limbs in both groups
Discussion
Lymphoedema is a chronic condition, which reduces patient’s physical fitness considerably and contributes to deterioration of their mental state [3]. Although numerous scientific societies recommend the administration of the comprehensive antioedema therapy, many authors take a critical view of the usefulness of individual methods applied as a monotherapy [4, 7–9]. This refers also to the MLD. According to some authors, the usefulness of this method is minor as compared to the control group applying the comprehensive antioedema therapy [6]. On the other hand, however, there are authors who note that MLD is a valuable addition to the comprehensive antioedema therapy, but they admit that it should not be applied independently, separately from other physiotherapy methods [10]. MLD itself should also be performed with due care and precision, starting with stimulating terminuses and the abdominal cavity [11]. In our trial MLD was performed by means of Vodder’s grip. As it was demonstrated in the studies by Foldi and Wolf, the application of lubricants and creams before MLD reduces the effectiveness of circular and pumping grips, which eventually contributes to weakening of the effectiveness of the entire MLD [11, 12]. The task of MLD is not only to stimulate lymphangiomotoricity, but also to improve elasticity of skin, the subcutaneous tissue, as well as to reduce the oedema volume, the latter being a measurable effect of the procedure. Frequently, advanced stages of oedema give an effect of hard, inelastic tissue resistance. The application of MLD techniques allows to observe enhanced “softness” of the tissue, which may have a positive effect on the application of subsequent pillars of the comprehensive antioedema therapy [13]. In 2017 the European Expert Group recognised the compression therapy as the most important factor controlling the oedema volume, and in doing so it recommended that it should be included in the international standard of the lymphoedema treatment, both in its initial and advanced stages [14]. Research on the effectiveness of the compression therapy has been undertaken all over the world in recent years [15–18]. As in the case of MLD, a properly applied compression dressing is of key importance (multilayer bandaging, taking measurements in order to make compression clothing). Inaccurate or altogether inappropriate bandaging, e.g. leaving the toes out, may even cause the oedema expansion and increase [19, 20].
In our trial we applied the compression therapy by means of elastic bandages with low extensibility. Previous studies had confirmed their advantage at the initial stage of the treatment, as compared to hosiery. In the period of 24 weeks Badger achieved an oedema reduction by 31% when elastic bandages and compression stockings were used, as compared to 15.8% when only compression stockings were applied [21].
Johansson compared the MLD with the sequential pneumatic compression (SPC) in the treatment of unilateral lymphoedema in 28 women after breast cancer. After 2 weeks he observed a reduction in the oedema volume by 15% and 7%, respectively (p < 0.03). According to this research, MLD and SPC had a significant effect on the reduction of the oedema of patients’ arms, whereas no significant difference between these two treatment methods was demonstrated [22]. Similar results of the therapy of women after mastectomy were achieved by Ochałek and Grądalski, who compared the multilayer bandaging compression therapy with the comprehensive antioedema therapy. During 10 treatment sessions they achieved an oedema reduction by 11.7% and 10.5%, respectively [23].
Johansson et al. in their other work compared the effectiveness of the compression therapy with low extensibility bandages applied as a monotherapy in combination with MLD in patients with postoperative oedema of lower limbs. In both cases they achieved a considerable reduction of oedema volumes (30–37%), and the volume differences were insignificant [24]. Likewise, Pawlaczyk did not observe any differences in terms of reduction of the postoperative oedema volume, as well as the degree and range of normalisation of skin microcirculation in patients after the femoropopliteal bypass, in which the sequential pneumatic compression, low extensibility bandages, and manual lymphatic drainage were applied in the postoperative period in combination with compression products [25].
Different results were obtained by McNeely et al., who compared a 4-week-long bandaging compression therapy applied as a monotherapy or in combination with MLD. They achieved significant oedema reductions in both cases; nevertheless, the reduction range was significantly higher for the procedures applied in combination [26]. In both cases referred to above the authors observed a higher oedema reduction than we did in our trial. This difference may result first of all from a longer duration of the therapy – in our trial it was 2 weeks, whereas in the trials of Johansson and McNeely it was 6 and 4 weeks, respectively.
Our observations of both groups are suggestive of the fact that multilayer bandaging applied as a monotherapy has similar effectiveness to that of the comprehensive antioedema therapy comprising MLD, multilayer bandaging, and physical exercise. Although we achieved a similar effect in all the groups, we do not know what motor reactions of blood and lymphatic vessels take place when the multilayer bandaging is applied alone as compared to stimulating the lymphatic system by means of MLD techniques only. It is possible that when MLD as well as the exercise therapy i.e. the comprehensive antioedema therapy is applied, besides the mechanical squeezing of the oedema there may also occur reflex lymphangiomotoric reactions, and in the long-term perspective there arises a question whether the application of the comprehensive antioedema therapy does not affect a better therapeutic effect than the multilayer bandaging alone, without MLD techniques and the exercise therapy. It seems to be advisable to conduct research towards a better understanding of vascular and tissue reactions during the application of the multilayer bandaging as a monotherapy.
Conclusions
The obtained results suggest that in patients deprived of access to the comprehensive antioedema therapy, comparable results can be achieved by implementing an adequate compression therapy. Consequently, it is advisable to implement the multilayer compression therapy in patients with lymphoedema who wait for the administration of the comprehensive compression therapy or who for various reasons do not have this option.