Purpose
The management of superficial non-melanoma skin neoplasms is commonly performed by kilovoltage radiation. However, this approach has significant limitations, particularly in dose distribution over curved surfaces and anatomically complex areas. In addition, electron beams are used in these treatments, but their application may be sub-optimal in specific situations, such as in small lesions located in anatomically difficult-to-access areas [1]. Given this context, high-dose-rate brachytherapy (HDR-BT) has emerged as an alternative for the management of cutaneous neoplasms, with guidelines and consensus standardizing indications, planning, and procedures [2-4]. This treatment is widely used in the control of various types of cancer, including cervical, prostate, breast, and skin [2, 3, 5, 6]. One of the main advantages of HDR-BT is its ability to effectively eradicate tumors while preserving functionality of the treated area. However, its successful implementation requires specialized tools, such as molds or applicators, which ensure reproducibility, adaptation, and accessibility to the area of interest. This allows correct dose distribution in the target volume and decreases complications [3, 4].
Since the beginning of radiotherapy, the use of molds and applicators for brachytherapy has proven to be a fundamental tool for obtaining positive clinical results [7, 8]. Accordingly, recent clinical series and analyses have consolidated the role of superficial techniques in various anatomical locations and surface/interstitial combinations when the anatomy requires their use [9-12]. In recent years, the development of technologies, such as high-resolution imaging, artificial intelligence, and custom-made applicators using 3D printing, has significantly transformed the practice of brachytherapy in both low- and high-rate modalities [6]. A wide spectrum of printable materials is available in this area, including thermoplastic filaments (e.g., PLA, ABS, PET-G), light curing resins, and flexible polymers, such as TPU. The choice depends on anatomical and dosimetric requirements [13, 14]. In relation to elastomers, silicone rubber (usually RTV) is employed in surface molds or bolus, customized for its adaptability, structural consistency, and adequate skin biocompatibility. In the context of surface HDR, clinical guidelines recognize this material as appropriate to optimize the contact, reduce air gaps, and ensure positioning reproducibility [2, 3, 15].
In clinical applications, devices manufactured by 3D printing have demonstrated repeated utility in several areas of clinical application, providing customization and improvements in clinical logistics [13, 15, 16]. However, high-concavity regions, e.g., fingers, still pose specific challenges, as the absence of applicators designed for these scenarios can induce dose variations when air-tissue interfaces are present, generating hot spots and cold spots. Therefore, the manufacturing of devices that are optimally adapted to the patient’s anatomy is essential to ensure proper dose distribution in the target volume [17]. In addition, previous evidences demonstrate that the administration of HDR-BT by superficial molds or applicators can offer local control with favorable esthetic and functional results, particularly in selected skin lesions [9, 18-20].
In this context, applicators manufactured by 3D printing represent an innovative solution compared with traditional devices. These allow for an anatomical design that optimizes both treatment safety and efficacy by minimizing air gaps between the applicator and the patient’s surface [13].
Material and methods
The present research was developed in three stages. First, the design and manufacturing of an assemblable negative mold for a cylindrical finger applicator using 3D printing technologies. Second, the elaboration of applicator by pouring RTV silicone into a mold; and third, the realization of tomographic simulations and dosimetric planning of the applicator in a handheld anatomical phantom. In order to identify the developed device, the AppliFinger name was assigned to this applicator.
Prototype design
The applicator design consisted of a cylindrical structure made of RTV silicone rubber, designed to house the finger inside. This structure included a hollow central cavity, aligned with the cylinder shaft, intended for direct contact with the finger’s surface. In addition, an open segment was designed that spans a 93.5-degree angle, allowing the finger’s thickness to comfortably fit the inner surface of the applicator. The cylinder’s walls were designed with a uniform thickness throughout the structure to confirm functionality.
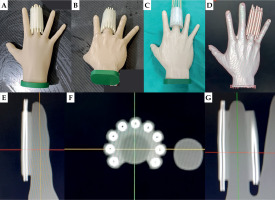
The design incorporated nine circular perforations distributed equidistantly along an arc in the upper sector of the cylinder. These perforations were surrounded by an additional wall that acts as a support for the catheters, through which the radioactive sources would be positioned. Dimensions of the prototype were set at 5.5 cm in height, with a distance of 6.7 mm between each perforation, and a thickness of 5 mm between central cavity of each hole and inner surface of the applicator, as visualized in Figure 1.
The process of prototype’s manufacturing started with creating an assemblable negative mold by Autodesk Fusion 360 3D modelling software (version 2.0.20981). The prototype consisted of four parts. First two pieces corresponded to the base and upper lid designed as discs, with central cavities and nine perforations distributed equidistantly around cavities. These two pieces included grooves in both the center and periphery to ensure correct fitting with the other parts of the set. The third piece was an outer cylinder that defines outer walls of the silicone structure, while the fourth piece consisted of a central cylinder that aligns with the base and top cap to complete the mold. For lamination process, a 3D model was processed in a Bambu Studio software (version 1.9.1), generating extrusion paths (G-code) required for layer-by-layer fabrication of the mold. Configuration excluded support structures and adhesion base, thus optimizing print time and surface finish. Printing was performed with a Bambu Lab X1 Carbon printer using PLA (polylactic acid) filament. A layer resolution of 0.2 mm, infill density of 30%, extrusion temperature of 220°C, and bed temperature of 60°C were employed.
Prototype manufacturing
For this process, the four previously printed parts, nine cylindrical and straight metal rods of the same diameter as the catheters, and a two-component RTV silicone elastomer composed of a silicone base (component A) and a curing agent (component B), which react chemically when mixed, were utilized to form the final solid material.
The process was carried out as follows: the third and fourth pieces of the mold were placed on the base (lower structure), along with the nine cylindrical metal rods, which acted as guides to form channels corresponding to catheters in the final silicone prototype. At the same time, two-component RTV silicone was prepared by mixing the base component with curing agent, following proportion recommended by the manufacturer. The resulting mixture was placed in a vacuum chamber to eliminate air bubbles generated during the mixing process. This procedure eliminates internal irregularities, favors homogeneous distribution of the material, and contributes to obtaining a silicone mould with better mechanical properties and a higher quality surface finish. The cavity was then filled with the prepared silicone rubber, so that it would comprehensively cover all spaces around the structures and metal bars. Finally, the top cover was placed over the assembly, adjusting it to fit correctly with the other pieces. This step allows the mold to be shaped and stable while the silicone goes through the curing process. After completing the curing time (8 hours), demolding was carried out, in which tests were performed to verify flexibility and adaptability of the design to fingers with different anatomies. In addition, the pressure exerted between the holes and catheters was evaluated, ensuring it was acceptable. Optimal pressure allows the catheters to remain firmly inside the applicator, preventing from shifting.
Simulation and planning
Tomographic simulation was performed using two handheld phantoms, one male and one female. The applicator was placed on two different fingers of each hand to carry out the study. When adhesive tape was applied to the outer surface of the applicator to attach it to the phantom’s finger, a deformation occurred in the openings intended for the catheters. To avoid this effect, guides were placed to ensure the integrity of these openings during the tomographic simulation. Owing to its cylindrical design, the applicator easily adapts to the morphology of the finger. However, it was necessary to bring the ends of the longitudinal part of the applicator closer together to confirm that the internal surfaces come into direct contact with the surface of the finger, thus optimizing the fit. This simulation study was performed with a Discovery CT590 tomograph (GE Medical Systems) in helical mode using an energy of 120 kV and 1.25 mm cuts. The acquisition encompassed an anatomical field that extended from the distal tip of the middle finger to the end of the hand.
Treatment planning was carried out with a HDRplus 3.0.6.0 system (Bebig GmbH) using flexible LAA1400-GYN catheters. To simulate the treatment area, a volume under the surface of the phantom with a thickness of 3 mm was defined and consequently, the prescription depth was set at 3 mm from the surface. In each catheter, 8 stop points were configured, evenly spaced every 5 mm. Dwell times were individually adjusted to optimize target volume coverage (PTV), and duration of stopping points was modulated to ensure that 100% of PTV volume receive the prescribed dose.
Results
Design and printing of the prototype
During the development of the model, various modifications were implemented to solve several challenges. One of the main difficulties was to achieve a proper fit between the parts, maintaining the necessary pressure for assembly. After several adjustments, the optimal print settings were defined, allowing to obtain a functional fit. Likewise, the design of the holes for the metal bars was optimized, slightly increasing their diameter to facilitate insertion. These adjustments struck a balance between the ease of assembly of the parts and proper pressure that assured the firmness of the metal bars in position. In Figure 2, the 4 pieces that were designed are illustrated.
In the printed prototype, the walls were of the right hardness, although small accumulations of material were observed at the edges. Likewise, both the base and lid showed slight irregularities on their surfaces. However, these drawbacks were easily solved by a light wear process with fine-grit sandpaper number 150, which allowed the finish to improve without compromising critical dimensions of the design. On the other hand, by adjusting the thicknesses of the sockets, optimal pressure was achieved for both the assembly of the parts and insertion of the metal bars, ensuring functionality in the final design, as can be seen in Figure 3A.
Demolding and mechanical testing
After the curing process was completed, the silicone experienced a slight contraction, which generated pressure between the walls of the structure. However, the design of the assembly allowed a proper demolding of the prototype without major inconveniences. A minimal remnant of silicone was observed in the joints, which was easily removed, as presented in Figure 3B and C. Regarding the mechanical tests carried out on the silicone applicator, the following results were obtained: the flexibility of the material allowed optimal adaptability to fingers of different thicknesses due to the horseshoe-shaped design that facilitated adjustment to various dimensions. In addition, when the catheters were inserted into the holes of the applicator, adequate pressure was evidenced for their stability. The catheters remained firmly in place, even in tampering, confirming the functionality of the design.
Tomographic simulation
The results show an adequate fit between the applicator and the fingers of the hand phantoms. Figure 4A and B illustrates the correct fixation of the applicator with guides on the middle finger of each phantom, while Figure 4C shows the replacement of the guides by catheters in the applicator.
In the tomographic simulation study, adequate contrast was observed between the phantom, silicone applicator, and catheter’s guidewires, which allowed them to be clearly differentiated in reconstructed images. This result was due to differences in electron density of each component. In particular, the silicone used has a relative electron density of 1.176, which makes it easy to distinguish. High uptake of the catheter guide did not generate artifacts in the environment. Cutting thickness of 1.25 mm was optimal, since the study area was small. Another important aspect to consider was the approach of the ends of the longitudinal part of the applicator, so that their internal surfaces could establish direct contact with the surface of the finger.
In the first two model studies, the approach of the applicator to the phantom was moderate, resulting in the formation of small air zones at the interface. In contrast, in subsequent studies, a larger approach was applied, which was maintained by adhesive tape, achieving an optimal adaptation between the inner surface of the applicator and that of the phantom’s finger. Figure 4E, F, and G illustrates the sagittal, axial, and coronal views, respectively, where uniform adhesion of the applicator on the surface of the phantom is evidenced. Figure 4D presents the three-dimensional model reconstructed from the tomographic simulation. It was observed that, due to the uniformity of the internal radius of the applicator and asymmetrical morphology of the finger (which is not perfectly circular, and exhibits a progressive reduction in distal diameter), the applicator tended to cover a larger surface area in the distal phalanx compared with the base.
Treatment planning
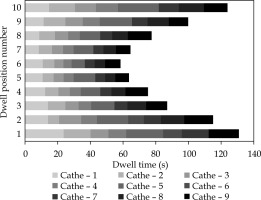
The symmetry of the applicator and equidistant arrangement of the catheters assured a favorable spatial distribution of the dose in the target volume. In addition, the modulation of stop times permitted for longer times to be assigned to both the catheters located at the ends of the assembly (the first and last) as well as to the initial and final positions within each catheter (Figure 5). This configuration provided greater control over dose conformation in peripheral areas, thus improving dose coverage around the treatment area. Additionally, the need to activate an additional stop beyond the last visible cut of the treatment volume in all catheters was identified. This decision helped to ensure complete coverage of the distal areas of PTV, avoiding regions with potential underdosing, which could compromise the treatment objective.
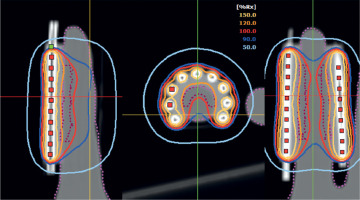
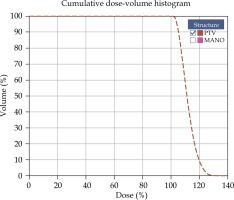
In terms of dose distribution, it was observed that most of the 120% isodose was located on the surface of the applicator, which was consistent with the dose gradient characteristic of surface configurations, where the dose on the skin tended to exceed the prescription [3, 21]. The maximum recorded surface dose reached 129% of the prescribed dose, a value within the ABS recommended threshold (ideally < 150%) and in line with ACROP (typical prescription depth 3-5 mm and approximately 130-135% surface for standard applicators), provided that critical structures were not involved and high-dose region was clinically tolerable [3, 15, 21]. On the other hand, the 100% isodose completely covered the volume defined as PTV, thus ensuring adequate coverage of the therapeutic target [15], as demonstrated in Figure 6.
Higher dose curves, such as 150% isodose were enclosed within the applicator, which can be verified by the dose-volume histogram shown in Figure 7, with no volume values observed for that dose. However, in the case of air between the surface of the applicator and the surface of the phantom, the dose on the surface of the finger should be increased, since the 100% curve tends to move further away from the applicator. For this reason, it is essential to ensure good adhesion between the applicator and the phantom’s surface.
Conversely, the size of the lesion is an important factor in treatment planning, as it directly influences the choice of applicator design and the distribution of stopping times and positions. In this study, the treated areas had an approximate extension of 7.5 cm2, which provided adequate coverage with the configuration used. However, in larger lesions, a detailed evaluation of morphology and anatomical limits of the target area is required, since these conditions could require a re-distribution of catheters and a different modulation of stop times. These adjustments are important to ensure adequate dose delivery, avoiding areas of both underdosing and excessive concentrations, which may increase the risk of toxicity.
Discussion
In a review article, Poltorak et al. [13] highlighted the advantages of combining 3D printing with brachytherapy, which contrasts with traditional methods. Before the 3D printing recognition, the construction of finger applicators was done manually. For example, Wadhwa et al. [22] fabricated a custom applicator using alginate, wax, and other materials, allowing the insertion of three catheters around the treatment area. Similarly, Somanchi et al. [1] described a procedure based on plaster bandages and alginate, while Tagliaferri et al. [23] used a home-made surface mold to irradiate a finger, obtaining good results with four-year follow-up. These methods required a prior fabrication of a finger’s replica for the elaboration of applicator. In contrast, the present study eliminates the need for such replication. Conversely, Gupta et al. [24] used a thermoplastic sheath coated with a 5 mm wax bolus incorporating five catheters, which involved an additional process to ensure equidistant placement and fixation to the bolus. Our design integrates both the catheter insertion holes and silicone bolus into a single structure, simplifying the clinical procedure. In addition, this applicator allows the placement of up to nine catheters, increasing stopping points, and improving the modulation of dose distribution in treatment area compared with other studies.
Guinot et al. [3] recommended the use of superficial flaps, H.A.M., and catheter flap set in lesions with moderate irregularities. Although they are flexible devices, their essentially flat geometry can limit adaptation in marked curvatures and generate surface misalignments. On the cost side, while some commercial systems may be more expensive, clinical practice and published reviews described low-cost local manufacturing alternatives (e.g., dental silicones, thermoplastics, acrylics, and desktop 3D printing), which permit functional and reproducible molds and applicators [25]. In this framework, the applicator presented in the current study is characterized by its low cost, favoring its implementation in clinical practice. From a geometric point of view, the applicator’s cylindrical design approves the natural coupling to the morphology of the finger, stabilizes the source-surface distance, and facilitates parallel trajectories of the catheters. In relation to the protection of adjacent tissue, we observed that in the distal portion, the catheters are located further away from contiguous structures, while at the base, they tend to approximate; therefore, separators and external shielding (e.g., Pb sheets) are recommended to maximize partition and reduce dose to neighboring fingers [26].
The silicone applicator is designed to treat the skin along all phalanges of the finger; however, it has limitations in specific areas. For example, the region inferior to the hyponychium (corresponding to the most distal and palmar surface of the finger) demonstrates a complex anatomical arrangement that hinders proper insertion of catheters, thus preventing effective treatment with the current design. In addition, the applicator is optimized to treat lesions with a maximum longitudinal extension of 4 cm and a radial reach of 2.5 cm, therefore, injuries that exceed these limits will require a re-configuration of the original design to ensure effective coverage.
In the study, the tests were performed using a handheld phantom. In contrast, a real patient’s hand exhibits greater flexibility and adaptability, making it easier to attach the applicator to the anatomy of the finger, and consequently, optimizing treatment. It is essential to ensure close and continuous contact between the applicator and the surface of the treatment area to avoid areas of air, especially on uneven surfaces, as recommended by the GEC-ESTRO/ACROP guidelines [3] and that of the GEB-SEOR/SEFM [27]. The presence of air can effectively increase the distance between the catheters and the most distal region of the target volume, and given high-dose gradient, favor superficial overdoses; thus, careful positioning and verification of full contact are critical. In relation to catheters, it is essential to position them at the top of the distal part of each applicator’s hole for reproducible reference.
In relation to dosimetric data in silicone molds, Salamat et al. [28] validated a flexible applicator using EBT3 radiochromic films, Monte Carlo simulations (MCNP), and TPS, evidencing a dosimetric agreement ≤ 0.73% at 0.5 cm depth. However, surface overestimates of up to 1.3% were observed by TPS. Similarly, Jaberi et al. [29] reported discrepancies of up to 7.7% between TPS and MOSFETs at 5 mm depth, with differences being even greater on the surface. These findings support the recommendation to prescribe the dose at greater depth, avoiding immediate surface as a dosimetric reference plane. Despite these technical limitations, clinical evidence supports the efficacy of silicone molds in superficial brachytherapy. A series of 273 skin tumors treated with custom molds, including silicone, achieved 95.2% of local control, moderate toxicity, and a recurrence rate of < 5% after a median follow-up of 25 months [30]. In addition, a retrospective review of 65 lesions in 59 patients treated with custom molds (silicone or 3D-printed) showed a complete response of 96.8%, local control of 84.9%, and no grade 3 to 5 toxicity [31]. Moreover, Membrive et al. [32] reported a local control of 94% at 10 years with personalized HDR molds, with limited toxicity. These findings support the clinical use of silicone molds in superficial brachytherapy.
Conclusions
The development of the present silicone applicator proved to be a viable, efficient, and reproducible alternative for the treatment of superficial skin neoplasms on the fingers. Its cylindrical design, together with the incorporation of up to nine catheters, provided an equidistant and symmetrical distribution of the stopping positions of radioactive sources, optimizing the coverage of the target volume and guaranteeing adequate dosimetric modulation. The tests carried out on phantoms showed a good adaptability of the applicator to different morphologies of the fingers, confirming its functionality, structural stability, and capacity to minimize air spaces between the device and the tissue, an essential condition to avoid irregularities in the distribution of the dose.
This study confirms that the manufacturing of personalized devices using 3D printing not only facilitates technical implementation in clinical settings, but also enhances dosimetric control and treatment shaping in hard-to-reach areas. In addition, it represents a solution to the limitations of conventional flat applicators, providing a significant advance in therapeutic personalization in brachytherapy.