Introduction
Lupus erythematosus (SLE) is an autoimmune disease that causes complex multi-organ damage. It mainly affects women, more often at childbearing age [1]. People suffering from SLE have an almost 10-fold higher risk of developing cardiovascular diseases, and the risk of a heart attack in women increases up to fifty-fold [2]. Cardiovascular disease also remains the leading cause of death in people with SLE, accounting for over 1/3 of deaths in this population [3, 4]. The reasons for this phenomenon are not fully known. There are many hypotheses trying to explain them, and their common feature seems to be early and significantly accelerated development of atherosclerosis, compared to the general population [5]. This process is influenced by many risk factors, both classical and those related to SLE itself. The key role of disorders of immune regulation is indicated, including the synergistic action of genes involved in the initiation and development of atherosclerosis and genes involved in the autoimmune response [6]. Some authors indicate a correlation between the activity of the inflammatory process and the presence of specific antibodies with early atherosclerosis [7]. The early development of atherosclerosis is also influenced by classic risk factors, including the presence of hypertension, which occurs much more often than in the general population. Depending on the standards used, it may be present in up to 60% of patients with SLE [8]. In this situation, the negative influence of not only arterial hypertension itself, but also an incorrect diurnal pressure profile was demonstrated. People with an insufficient decrease in blood pressure (BP) at night compared to the hours of daily activity (non-dipper) have a significantly higher cardiovascular risk, also in the case of correct BP values in office measurements [9–11]. It seems that the association with early, as well as subclinical, atherosclerotic changes in vessels, not only of the coronary circulation, such as increased intima media thickness (IMT), presence of atherosclerotic plaques in carotid arteries, or increased stiffness of their walls, may be associated with the presence of nocturnal hypertension, including its particular form of isolated nocturnal hypertension [12–15]. There are many processes and mechanisms underlying the dysregulation of the blood pressure profile. One of the postulated processes is a disturbed autonomic balance with a predominance of the sympathetic nervous system [16, 17]. It is extremely important in autoimmune diseases, especially in SLE, where the presence of an autonomic imbalance with a predominance of sympathicotonia has already been demonstrated, mainly at night. This state of autonomic imbalance may contribute to the generation of disorders of arterial pressure regulation, and consequently to the formation of early atherosclerotic changes [18, 19]. Currently, the basic method of assessing the diurnal blood pressure profile is ambulatory recording with automatic devices – ambulatory blood pressure monitoring (ABPM). Moreover, the data obtained during the 24-hour measurements allow to assess not only the average values of pressure during the day and night, but also to determine its night-time dipping, systolic and diastolic load, pulse pressure, which enables the diagnosis of other forms of arterial hypertension, such as nocturnal hypertension, masked or white coat hypertension [20, 21]. Measurements obtained with ABPM also better correlate with possible organ damage, including left ventricular hypertrophy, especially in masked hypertension [22].
Aim
The aim of our study was to assess the frequency of abnormal circadian blood pressure profiles in patients with SLE without overt cardiovascular disease. Moreover, the impact of such a profile on the markers of increased cardiovascular risk and early symptoms of atherosclerosis in the form of increased IMT and the presence of atherosclerotic plaques in the carotid arteries was assessed.
Material and methods
Sixty-seven people under the care of the Dermatology Clinic of the Medical University of Lodz were pre-qualified for the study. These patients had previously been diagnosed with SLE in accordance with the applicable criteria of the 1982 American College of Rheumatology as modified in 1997 and SLICC of 2012 [23, 24]. Due to the aim of the study, the criterion qualifying for participation in the study was the correct blood pressure values obtained in the office twice during subsequent visits, and carried out in accordance with the applicable ESH/ESC guidelines [25, 26]. Participants had a negative history of any overt cardiovascular disease, including ischemic heart disease, heart failure, cerebral circulation disorders, and peripheral arteriosclerosis. The exclusion criteria were also prior treatment of hypertension, diabetes, renal failure, and autoimmune diseases other than SLE. It was not allowed to take any medications, apart from steroids and immunosuppressants used in the treatment of SLE. All pre-qualified participants of the study had detailed medical history taken, clinical examination conducted, and medical documentation analysed. They also had basic laboratory tests, resting ECGs and echocardiography performed. The participants then had in-office blood pressure measurements taken in accordance with the current ESH/ESC guidelines. These tests were carried out using an automatic Omron M2 device (OMRON Healthcare Japan) operating on the oscillometric principle, twice, with an interval of one week, at rest between 8:00 and 10:00. Subjects with measurements below 140/90 mm Hg twice were eligible for the relevant study. Due to such narrow qualification criteria only 32 women were finally included in the study. The control group consisted of 30 healthy women, matched appropriately for age and anthropometric data. The participants of the study had a 24-hour automatic blood pressure recording performed using the HolCARD CR-07 system by Aspel Zabierzow Polska and the CR07 recorder performing measurements using the oscillometric method. During the day, measurements were taken every 20 min, and every 30 min during the night rest. The obtained results were additionally evaluated by a cardiologist, experienced in the diagnosis and treatment of arterial hypertension. Records of sufficient quality and quantity of measurements were obtained from all study participants. In addition, after the registration was completed, the patients had an ultrasound examination of the carotid arteries using the HP Hewlett PACKARD SONOS 1000 ultrasound machine. IMT measurements were carried out at 3 points of the common carotid artery, i.e. about 1 cm proximal to the bulb, the middle course and approx. 2 cm from the origin artery. The measurements obtained were averaged. The presence of atherosclerotic plaques was assessed on the entire available course of the common, internal and external carotid arteries.
Statistical analysis
Statistical calculations were made on the basis of the Statistica 13.1 PL package – by StatSoft Inc. The normal distribution of the studied populations was tested using the Shapiro-Wilk W test. To verify the significance of differences of results between treatment groups the t-test was used (for variables having a distribution close to the normal), and in other cases, non-parametric tests were used ANOVA Kruskal-Wallis, and Wilcoxon and U Mann-Whitney test.
The χ2 test was used for the quality features. Spearman’s R coefficient was used to test possible correlations between the selected variables. P-values < 0.05 were taken as indicating the existence of statistical significance. The obtained results were presented depending on the normality of the distribution in the form of the mean and standard deviation as well as the median of min/max.
The study was performed in agreement with the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the Bioethics Committee of Medical University of Lodz (approval no. RNN/591/09/KB). Written informed consent was obtained from all subjects.
Results
Both of the compared groups, i.e. patients with SLE and the control group, did not differ statistically significantly in terms of anthropometric features, including the age of 52.06 ±10.55 vs. 53.60 ±10.58 years; p = 0.57; weight 68.89 ±13.58 vs. 68.12 ±10.74 kg; p = 0.81, height 160.47 ±6.20 vs. 162.87 ±6.18 cm; p = 0.12 and body mass index (BMI) 26.75 ±5.89 vs. 25.73 ±4.37 kg/m2; p = 0.44. In the group of women with SLE, 56.25% were in the postmenopausal period and this percentage did not differ significantly from the control group – 57.00% (p > 0.05). The disease duration from diagnosis was on average 11.09 ±7.28 years. Echocardiography revealed no typically described lesions, including valve lesions of the Libman-Sacks type, in any of the examined women. There were no significant differences in the size of the atria and ventricles, the function of the left and right ventricles, including the ejection fraction. Left ventricular diastolic dysfunction was only more frequent in the SLE group – 48.9% vs. 13.0% (p < 0.05). The vast majority of patients underwent immunosuppressive treatment at the time of study entry. Steroids were taken by 21/32 (65.6%) women, in the mean daily dose of 6.7 mg as prednisone, 4 patients took chloroquine 250 mg/day, and 2 patients – azathioprine 50 mg/day. In 15 (46.9%) subjects the presence of atherosclerotic plaques in the carotid arteries was found. Women with atherosclerotic plaques were statistically significantly older 59.13 ±5.68 vs. 45.82 ±9.96 years; p < 0.001. What is more, they had a statistically significantly bigger IMT in relation to people without atherosclerotic plaques and the control group 0.833 ±0.216 vs. 0.606 ±0.121 mm; p < 0.001 and 0.833 ±0.216 vs. 0.660 ±0.16 mm; p < 0.01. A statistically significant positive correlation was also found between IMT and the age of patients R = 0.482; p < 0.05, nocturnal systolic blood pressure – R = 0.394, night systolic pressure load R = 0.350, night-time drop in BP – R = 0.401 and the presence of atherosclerotic plaques R = 0.543, all at p < 0.05 (Figure 1). Also, the presence of atherosclerotic plaques was positively correlated with age and numerous parameters obtained from ABPM (Table 1).
Table 1
Correlation between the selected parameters of ABPM and age, IMT and the presence of atherosclerotic plaques in the carotid artery. Spearman rank test R value
| Blood pressure monitoring [mm Hg] | Age [years] | IMT [mm] | Plaque |
|---|---|---|---|
| 24-h SBP | 0.288 | 0.194 | 0.499* |
| 24-h DBP | 0.030 | 0.061 | 0.231 |
| 24-h MAP | 0.182 | 0.118 | 0.404* |
| Systolic load | 0.318 | 0.200 | 0.485* |
| Diastolic load | 0.083 | –0.039 | 0.271 |
| Pulse pressure | 0.316 | 0.229 | 0.444* |
| Daytime SBP | –0.048 | –0.048 | 0.112 |
| Daytime DBP | –0.163 | –0.123 | -0.058 |
| Daytime MAP | –0.139 | –0.083 | -0.003 |
| Systolic load | 0.093 | –0.030 | 0.193 |
| Diastolic load | –0.173 | –0.259 | -0.058 |
| Pulse pressure | 0.176 | 0.066 | 0.248 |
| Nigh-time SBP | 0.415* | 0.394* | 0.678* |
| Nigh-time DBP | 0.246 | 0.280 | 0.587* |
| Nigh-time MAP | 0.319 | 0.343 | 0.648* |
| Systolic load | 0.434* | 0.350* | 0.656* |
| Diastolic load | 0.322 | 0.269 | 0.546* |
| Pulse pressure | 0.431* | 0.303 | 0.580* |
Figure 1
Correlations between IMT and age, night blood pressure dipping, systolic night load and nocturnal mean arterial pressure (MAP)
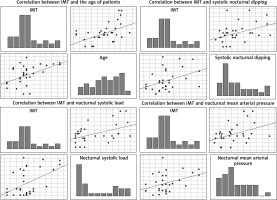
Table 2 presents a comparison of the results obtained in the ambulatory blood pressure measurement between women with SLE and the control group. There were no statistically significant differences in the 24-hour mean systolic and diastolic blood pressure. Such differences were not found both at night and during the hours of daily activity. There were also no significant differences for the mean arterial pressure and the value of pulse pressure.
Table 2
Differences between the studied groups
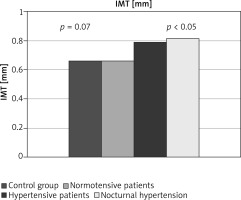
Half of the women with SLE had an abnormal 24-hour blood pressure profile. Four of them had a non-dipper profile, 8 people had the invert type, and the remaining 4 women had an excessive night pressure drop – hyper-dipper profile. In line with the assumptions of the study, none of the participants had previously been diagnosed with arterial hypertension based on office measurements. In contrast, based on ABPM, it was found that 14 (43.75%) patients at baseline normotensive women can, however, recognize hypertension, according to the ESC/ESH guidelines [25, 26] – masked hypertension. The vast majority of this group (12 people – 37.5%) was characterized by the presence of nocturnal arterial hypertension. Of these, 3 people had isolated nocturnal hypertension. Compared to the remaining normotensive group of women, women with SLE and HA had atherosclerotic plaques significantly more often (11/14) 78.57% vs. (4/18) 22.22%; p < 0.01. Women with nocturnal arterial hypertension (83.33%) had an even higher percentage of atherosclerotic plaques. They were also older 57.07 ±8.80 vs. 48.17 ±10.35 years; p < 0.05. IMT was also higher in the hypertensive group, but still not statistically significant 0.79 ±0.24 vs. 0.66 ±0.16 mm; p = 0.07. Such significance appeared in the group of women with nocturnal hypertension 0.81 ±0.25 vs. 0.66 ±0.16 mm; p < 0.05. Exactly the same difference could be found in comparison with the control group – IMT value 0.66 ±0.16 mm. In any of these cases, no statistically significant difference was found with the duration of the disease (Figure 2).
When analysing together people with an abnormal night decrease in BP, i.e. non-dipper and invers, compared to women with normal and excessive night decrease blood pressure, statistically significant differences were found for the age of 57.5 (min. 30.0/max. 71.0) vs. 48.5 (min. 30.0/max. 65.0) years; p < 0.05 and IMT 0.75 (min. 0.5/max. 1.2) vs. 0.65 (min. 0.4/max. 1.2) mm; p < 0.05. Also, people with an overnight increase in BP (invers) were statistically significantly older than those with a normal night BP dipping – dipper (59.63 ±6.97 vs. 51.81 ±8.93 years; p < 0.05) and were characterized by a higher IMT (0.86 ±0.21 vs. 0.66 ±0.20 mm; p < 0.05). The pressure profile, however, was not related to the disease duration.
Discussion
Systemic lupus erythematosus according to the American Heart Association is an independent risk factor for the development of cardiovascular diseases [27]. The mechanism by which it accelerates the atherosclerotic lesions remains largely unknown. The various theories existing so far have not been fully confirmed. Recent reports indicate an important role of disturbances in blood pressure regulation. The incorrect diurnal pressure profile was confirmed by Sabio et al. [12]. They showed that women with SLE, compared to the control group, have a disturbed circadian blood pressure profile, mainly in the form of the non-dipper profile and the presence of nocturnal hypertension, especially isolated nocturnal hypertension. On the other hand, the abnormal nocturnal blood pressure profile significantly correlated with abnormal carotid-femoral pulse wave velocity (PWV) values, which is an index of arterial stiffness, an early risk factor for CVD. Interestingly, despite the relatively young age of the study participants – the median of 38 years, as many as 39% received antihypertensive treatment, of which 48% required at least 2 antihypertensive drugs. These results are consistent with those obtained by the authors. The group of women studied by us was normotensive in office measurements, but using ABPM in 43.75% of cases it was possible to diagnose arterial hypertension, and in 37.5% – nocturnal hypertension. A relatively large group of patients was characterized by a disturbed diurnal profile with an inverted night profile – invers affecting as much as 35% of the participants. However, in comparison to research by Sabio et al., we investigated by us an older population – an average of 52.06 ±10.55 years. Neither of the participants was treated for hypotension. The population of women with SLE selected in our study is specific. It is a population in which, according to standard diagnostics, it was not possible to diagnose arterial hypertension, and thus initiate early treatment limiting its negative consequences. This translated into a much more frequent presence of markers of early atherosclerotic changes in the form of atherosclerotic plaques in the carotid arteries in 78.57%. Moreover, the group most at risk included women with nocturnal hypertension, among whom atherosclerotic plaques were present in as much as 83.33%, and IMT was significantly higher compared to normotensive persons and the control group. Interestingly, the severity of these changes was positively correlated with the age of patients, and did not correlate with time from the moment of SLE diagnosis. The presence of atherosclerotic plaques and abnormal IMT indicates a significantly higher cardiovascular risk in this group. Moreover, masked hypertension itself, diagnosed by us in 43.75% of cases, according to Pierdomenico et al. [28], gives the same increase in cardiovascular risk as persistent overt hypertension, as opposed to, for example, white coat hypertension. In turn, De la Sierra et al. [29] emphasize that the highest cardiovascular risk is found in people with an abnormal non-dipper profile and the presence of nocturnal hypertension. Chang et al. [13] in the paediatric-onset SLE population found an early development of subclinical atherosclerotic lesions in the form of increased IMT and PWV acceleration, which were particularly correlated with abnormal nocturnal systolic blood pressure dipping and non-dipper profile. These results are consistent with the observations obtained from our studies confirming the association of higher IMT with the presence of an abnormal nocturnal blood pressure profile and the presence of additional atherosclerotic plaques in the carotid arteries. Similar conclusions can be drawn from the study by Canpolat et al. [30], which also found a significant relationship between the non-dipper profile and increased IMT. The increase in IMT, as in the data obtained by us, was not related to the activity of the disease or its duration. On the other hand, in contrast to Canpolat et al., we have demonstrated the existence of a positive, although quite weak correlation between the IMT value and the night systolic pressure, the night systolic pressure load and the value of the night systolic pressure drop. Rather, these authors point to the dependence of IMT on diastolic pressure, which we have not shown in our studies. In turn, they indicate nocturnal systolic blood pressure as a more important risk factor for left ventricular hypertrophy than its values during the day. Similarly, Mercurio et al. [7] confirmed the correlation of systolic night pressure with artery stiffness and the early development of atherosclerotic lesions. It may be suspected that our results may be related to the higher age of our study population or to the nocturnal predominance of the sympathetic nervous system at night, but further research is still required. Chang et al. indicate as a possible cause of the accelerated development of atherosclerosis in people with an abnormal blood pressure profile, including those initially normotensive, as in our study population, with vascular endothelial dysfunction. It may result from high INFα expression, which disrupts the release of nitrogen oxygen in endothelial cells [31]. Another, but consistent with the above, explanation of this process is a disorder of autonomic regulation with sympathetic overdrive presented by Grassi et al. [17]. These authors point to a significant advantage of the sympathetic function in all forms of hypertension, including masked hypertension. We confirmed such disturbances of autonomic regulation in women with SLE in our earlier observations in patients with SLE [19]. We demonstrated the presence of an advantage in the sympathetic nervous system, especially at night. Other authors also obtained similar results [32]. Immune disorders and the presence of increased levels of inflammatory cytokines, as indicated by Pongratz et al. [33], may constitute a buckle linking autoimmune processes with autonomic imbalance that occurs in SLE patients.
Our results confirm a significant underestimation of the presence of arterial hypertension in patients with SLE and having normal blood pressure in office measurements. More than 40% of the women who started normotensive, were found to have hypertension, which had only just been diagnosed with ABPM. As a result, these women could receive treatment that minimized the adverse effects of hypertension. We also showed that most of them had nocturnal hypertension, which is a stronger risk factor for the development of atherosclerosis and cardiovascular complications than the daily or 24-hour values. In our studies, we also showed a significant relationship between an abnormal blood pressure profile and nocturnal hypertension with the accelerated development of atherosclerosis, which translated into a greater incidence of atherosclerotic plaques and higher IMT even in such a selected group, with no history of any cardiovascular diseases. So far, only a few studies address this issue, which is important for those involved in the care of patients with SLE. It therefore requires further investigation.
We are aware of the numerous limitations of our study. The first one is the small number of participants and the fact that only women were included. This was due to the specific, very narrow selection of the study group, which was to be initially without a history of any cardiovascular diseases, which was to exclude the influence of pre-existing complications and the drugs used on the assessment of risk factors. More than half of the examined women were postmenopausal, which we did not take into account in our considerations, mainly because the percentage of postmenopausal women in the control group was similar. Also, the method of measuring ambulatory blood pressure itself is burdened with a significant risk of disturbances, which, however, we managed to avoid by obtaining records suitable for interpretation. Therefore, it seems that recommending 24-hour blood pressure recording in patients with SLE, and perhaps more broadly with other autoimmune diseases, seems to be a cheap and effective method of actively searching for cardiovascular risk factors in the form of arterial hypertension, especially nocturnal hypertension, which may contribute to the earlier implementation of appropriate antihypertensive treatment. Thus, it may contribute to the reduction in cardiovascular morbidity and mortality in this group of patients. This requires further research.