Introduction
Psoriasis is a chronic inflammatory skin disease, whose pathogenesis stems from autoimmune activity of Th1, Th17 and Th22 lymphocytes and expression of proinflammatory cytokines secreted by these lymphocytes, i.e. tumor necrodsis factor-α (TNF-α), interleukin 23 (IL-23), IL-17 and IL-22 [1, 2]. Understanding immunological mechanisms underlying the disease has become crucial for the development of modern psoriasis treatments. Biologic agents used in the therapy of psoriasis can be divided into three groups based on the mechanism of action, i.e. TNF-α inhibitors, IL-12/IL-23 blockers and IL-17 inhibitors. Infliximab (IFX) is an IgG1 murine–human monoclonal antibody that binds with both the soluble subunit and transmembrane precursor of TNF-α [3]. IFX was approved by the U.S. Food and Drug Administration (FDA) in 2006 for the treatment of severe plaque psoriasis. The efficacy of IFX in the treatment of moderate to severe plaque psoriasis has been confirmed in numerous clinical trials [4]. In 2013 the European Medicines Agency registered two infliximab (Remicade) biosimilars, i.e. IFX (Remsima) and IFX (Inflectra). In the registration process, biosimilar drugs must demonstrate compatibility with the reference drug in terms of efficacy, safety and quality. The efficacy of IFX biosimilar drugs has been confirmed in numerous clinical trials [5]. The introduction of biosimilar drugs is associated with a significant reduction in treatment costs, which leads to more frequent use of this group of drugs in pharmacotherapy. Clinical data on the efficacy of biosimilar IFX for treatment of psoriasis are lacking, and there are even less real-life data regarding non-medical switching. In Poland, IFX treatment is available and reimbursed for psoriasis patients under the drug program “Treatment of moderate to severe form of plaque psoriasis” of the Ministry of Health. The provisions of the program allow for non-medical switching associated with the lowest drug price obtained in public bids conducted by the centres treating psoriasis patients.
Aim
The aim of the study was to evaluate the efficacy of 96 weeks of treatment with biosimilar IFX with non-medical switch option in patients with plaque psoriasis under the drug program “Treatment of moderate and severe plaque psoriasis” of the Ministry of Health in Poland.
Material and methods
The data were obtained from the Therapeutic Program Monitoring System (SMPT), which is an IT registry of the National Health Fund used to enter information on completed examinations for patients qualified for biological treatment under the drug program “Treatment of moderate and severe plaque psoriasis (B.47)”. Data on patients treated in 39 centres across Poland were entered into the registry. The severity of psoriasis before qualification was assessed using Psoriasis Area and Severity Index (PASI), body surface area (BSA) and Dermatology Life Quality Index (DLQI) scores. Patients over the age of 18 years who met criteria for disease severity defined as PASI > 10 points, BSA > 10%, and DLQI > 10 points were qualified for biological treatment with an IFX biosimilar. These patients had failed to improve after at least two systemic therapies prior to qualification, or had contraindications to systemic therapy, or developed adverse reactions after such therapy that made its continuation impossible. Lack of improvement after systemic therapies was evaluated after 3 months of PUVA, methotrexate ≥ 15 mg/week, cyclosporine ≥ 3 mg/kg/day, or after 2 months of systemic retinoids ≥ 0.5 mg/kg/day. Contraindications to treatment in the program were included in the characteristics of the medicinal products. Efficacy of treatment with biosimilar IFX was evaluated using PASI, BSA, and DLQI scoring performed at week 0, 14, 46, and 94. Failure to achieve a PASI score of 75 at week 14 was considered inadequate response to treatment. With a PASI score of 75–50, a minimum 5-point improvement in quality of life on the DLQI scale was required for continuation of therapy. The study group consisted of 96 patients qualified for IFX treatment in drug program B.47 from 2016 to 2018. A group of 91 patients was included in the clinical efficacy analyses. Their data in the registry were complete as they underwent the entire 94-week treatment cycle in the program completed by the end of 2020. Of the 91 patients treated, male patients accounted for 70.33% (64/91). The mean age of patients in the study group was 46.40 years and their mean body weight was 86.61 kg. The data are presented in Table 1.
Table 1
Basic patient characteristics
| Characteristic | IFX (n = 91) (min.–max. ± SD) |
|---|---|
| Age [years] | 46.40 (22–72 ±13.47) |
| Gender (M/F) | 64/27 |
| Mean weight [kg] | 86.61 (50–169 ±20.14) |
| Mean weight M [kg]/Mean weight F [kg] | 92.55 (65–169 ±18.77)/72.54 (50–113 ±16.04)* |
| Mean PASI | 16.53 (10.20–40.8 ±6.17) |
| Mean BSA | 40.1 (11–94 ±18.37) |
| Mean DLQI | 21.12 (11–30 ±5.4) |
Four out of 96 patients who were not included in the 94-week efficacy evaluation discontinued treatment at week 14 due to adverse drug reactions (3/4) and nonresponse to treatment, i.e. failure to achieve a PASI score of 50 (1/4). The 3 patients with adverse events by week 14 were diagnosed with sepsis due to urinary tract infection, acute anaphylactic reaction during infusion and death due to gastrointestinal bleeding. It should be noted that only adverse reactions resulting in termination of IFX therapy or the need for a change of the drug are entered into the SMPT registry. Therapy-related adverse events that do not result in termination of a patient’s participation in the program are not recorded in the registry and reported by the centres. One out of 5 patients who were not included in the analysis responded to IFX treatment and discontinued therapy at week 38 without explanation. Therefore, IFX treatment was not continued by 5.2% of those originally eligible. Secondary loss of response resulting in treatment discontinuation was defined as an increase in the disease severity below PASI75 or PASI75–50 and an improvement in DLQI of > 5 points from baseline visit value. IFX was administered at a dose of 5 mg/kg body weight by intravenous infusion at weeks 0, 2, 6, and every 8 weeks thereafter. Bioethics committee approval was obtained for the analysis.
Results
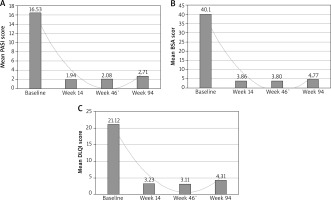
The mean PASI score among 91 psoriasis patients treated with IFX was 16.53 (Figure 1 A). At the time of qualification, the mean PASI score in the male group was significantly higher than in the female group (p = 0.043). The mean BSA score was 40.1 and the mean DLQI score was 21.12 (Figures 1 B, C). The differences in mean BSA and DLQI scores between the male and female groups at the time of qualification were not statistically significant. The mean body weight in the female and male groups was 72.54 kg (50–113 ±16.04) (min.–max. ± SD) and 92.55 kg (65–169 ±18.77) (min.–max ± SD), respectively. Body weight of male patients was statistically significantly higher compared to body weight of female patients (p = 0.000004). The PASI, BSA and DLQI values at the evaluated weeks are presented in Table 2.
Table 2
Mean PASI (*p = 0.043, **p = 0.036), BSA (*p = 0.016) and DLQI (*p = 0.016, **p = 0.0004) scores in female, male, and total groups
At week 14 of therapy, the mean PASI score in the treated group was 1.94 and the mean BSA score was 3.86 (Figure 1). There were no significant differences between male and female patients. The mean DLQI score at week 14 of the therapy was 3.23. The mean DLQI score was statistically significantly higher in the female group (5.44) than in the male group (2.30) (p = 0.016).
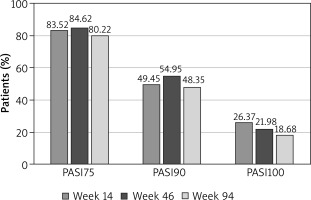
The improvement rate assessed as the mean change in PASI, DLQI, and BSA scores at week 14 of therapy was 89.92%, 93.75% and 90.91%, respectively. By week 14 of therapy, 83.52% (76/91) of patients achieved PASI75, 49.45% (45/91) achieved PASI ≥ 90 and 26.37% (24/91) achieved PASI100 (Figure 2).
At week 46 of IFX treatment, the mean PASI score was 2.08 (Figure 1 A). It was significantly higher in the female group (2.45) than in the male group (1.92) (p = 0.036). The mean BSA score in the study group was 3.80 (Figure 1 B). The mean BSA score was significantly higher in female patients (4.48) than in male patients (3.52) (p = 0.0165). The mean DLQI score in the study group was 3.11 (Figure 1 C). The mean DLQI score in the female group (5.15) was significantly higher than in the male group (2.25) (p = 0.0004).
The mean change in PASI, DLQI and BSA scores at week 46, compared to the beginning of therapy, was 91.67%, 96%, and 91.67%, respectively. At week 46 of therapy, 84.62% (77/91) of patients achieved PASI75, 54.95% (50/91) achieved PASI ≥ 90, and 21.98% (20/91) achieved PASI100 (Figure 2).
At week 94 of IFX therapy, the mean PASI score was 2.71, the mean BSA score was 4.77, and the mean DLQI score was 4.31 (Figure 1). The mean PASI (p = 0.063), BSA (p = 0.057) and DLQI (p = 0.057) scores at week 94 showed no significant differences between the male and female groups.
The mean change in PASI, DLQI and BSA scores at week 94 compared to the beginning of therapy was 89.39%, 90.91%, and 92.31%, respectively. At week 94 of therapy, 80.22% (73/91) of patients achieved PASI75, 48.35% (44/91) achieved PASI ≥ 90, and 18.68% (17/91) achieved PASI100 (Figure 2).
PASI100 at week 94 of therapy was maintained by 9 (9.89%) patients who achieved PASI100 at week 14. Five patients (5.49%) with a PASI score of 100 at week 14 of therapy, achieved PASI ≥ 90 at week 94. Six (6.59%) patients with a PASI score of 100 at week 14 of therapy, achieved PASI75 at week 94. However, 4 (4.4%) patients who achieved PASI100 at week 14 of therapy did not achieve PASI75 at week 94. The above differences are statistically significant (p = 0.04).
PASI100 at week 94 of therapy was achieved by 4 (4.4%) patients who achieved PASI ≥ 90 at week 14. Ten (10.99%) patients who achieved PASI ≥ 90 at week 14 of therapy maintained that score at week 94. Five (5.49%) patients who achieved PASI ≥ 90 at week 14 of therapy achieved PASI75 at week 94. However, 2 (2.2%) patients who achieved PASI ≥ 90 at week 14 of therapy did not achieve PASI75 at week 96. The above differences are statistically significant (p = 0.04).
PASI100 at week 94 of therapy was achieved by 2 (2.2%) patients who achieved PASI75 at week 14. PASI ≥ 90 at week 94 was achieved by 10 (10.99%) patients who achieved PASI75 at week 14. Twelve (13.19%) patients who achieved PASI75 at week 14 of therapy maintained that score at week 94, whereas 7 (7.69%) patients who achieved PASI75 at week 14 of therapy did not maintain that improvement at week 94. The above differences are statistically significant (p = 0.04).
PASI100 at week 94 of therapy was achieved by 2 (2.2%) patients who did not achieve PASI75 at week 14. PASI ≥ 90 at week 94 was achieved by 2 (2.2%) patients who did not achieve PASI 75 at week 14. PASI75 at week 94 of therapy was achieved by 6 (6.59%) patients who did not achieve PASI75 at week 14. Five (5.49%) patients achieved PASI75 neither at week 14 nor at week 94. The above differences are statistically significant (p = 0.04).
Discussion
This study was conducted in order to evaluate the efficacy of 94 weeks of therapy with IFX biosimilars in the first cycle of treatment under the “Treatment of moderate to severe plaque psoriasis (B.47)” drug program of the Ministry of Health in Poland. The program enabling IFX reimbursement was launched in mid-2015. Initially, IFX Remsima (Celltrion Healthcare Hungary Kft.) was the only biosimilar drug which could be used in the program. The IFX biosimilar, Flixabi (Samsung Bioepis NL B.V.), has been available since 2018 and Zessly (Sandoz GmbH) since 2019. The use of particular drugs depended on the results of public tenders conducted by the centres providing the therapy. The lowest price offered was the main drug selection criterion. There was the possibility of multiple non-medical switching during the course of treatment due to the price of the drug approved by the Ministry of Health in Poland. The possibility to evaluate the clinical efficacy of this solution in clinical practice enhances the value of the obtained data. Difficulties in the detailed assessment arise due to the fact that tenders for the IFX drug were conducted separately at each of the 39 centres, and the names of individual drugs are not entered into the SMPT registry. At week 14 of therapy, 83.52% (76/91) of patients achieved PASI75, 49.45% (45/91) achieved PASI ≥ 90 and 26.37% (24/91) achieved PASI100. At week 46 of therapy, 84.62% (77/91) of patients achieved PASI75, 54.95% (50/91) achieved PASI ≥ 90 and 21.98% (20/91) achieved PASI100. At week 94 of therapy, 80.22% (73/91) of patients achieved PASI75, 48.35% (44/91) achieved PASI ≥ 90 and 18.68% (17/91) achieved PASI100. In the EXPRESS trial, at week 10, PASI ≥ 75 was achieved by 80% and PASI90 by 57% of patients receiving IFX. PASI100 was achieved by 26% of patients. The study included moderate to severe plaque psoriasis patients with PASI ≥ 12 and BSA > 10%. At week 24, PASI ≥ 75 and PASI90 were achieved by 82% and 58% of patients receiving IFX, respectively. At week 50, 61% of patients achieved PASI75 and 45% achieved PASI90 [6]. In the EXPRESSII study evaluating the efficacy of IFX in continuous and intermittent therapy, PASI75 was achieved at week 10 in 70% and 76% of patients treated with IFX at doses of 3 mg/kg and 5 mg/kg, respectively. Treatment response was maintained more frequently during continuous therapy. In the group receiving 3 mg/kg, PASI75 at week 50 was achieved in 44% of patients in the continuous therapy subgroup versus 25% in the intermittent therapy subgroup. Furthermore, in the group receiving 5 mg/kg, PASI75 at week 50 was achieved in 55% of patients in the continuous therapy subgroup versus 38% in the intermittent therapy subgroup. At week 10, both groups showed significant improvement in DLQI (p < 0.001) [7]. Higher efficacy of continuous therapy was also demonstrated in the RESTORE study. PASI75 at week 52 of therapy was achieved in 80% of patients in the continuous therapy subgroup versus 47% in the intermittent therapy subgroup [8]. In the REALITY long-term efficacy and safety study, at week 14 of the first phase of therapy, PASI75 was achieved in 60.8% and PASI90 was achieved in 35.1% of 521 patients. At week 50, PASI75 and PASI90 were achieved by 56.8% and 39.4% of patients, respectively. The mean change in PASI score at week 14 was – 75.2%. The values found are lower than in our study. The REALITY study demonstrated that adequate response was significantly more frequent in non-biologically treated patients. In the REALITY trial, 42% of eligible patients had undergone prior biologic treatment. In Poland, the availability of biologic therapies was low and it can be assumed that this percentage is much lower. In the extended treatment phase of the REALITY study, at week 98 of therapy the mean change in PASI was –84.2% from baseline and is comparable to the results of our study. PASI75 at week 98 was achieved by 66.3% and PASI90 by 48.5% of patients enrolled in the study [9]. The study found that PASI75 was maintained at week 50 in 64.7% of patients who achieved it at week 14 of therapy. Moreover, PASI75 was maintained at week 98 in 71.6% of patients who achieved it at week 50. The efficacy of IFX therapy has also been evaluated in uncontrolled open-label studies. Schopf et al. [10] demonstrated a mean improvement in PASI of 88% at week 14 while Poulalhon et al. [11] showed that PASI75 was achieved by 68% of enrolled patients during this period. It should be noted that few patients participated in these studies and that patients with forms of psoriasis other than plaque psoriasis were also enrolled. The EXPRESS studies showed improved quality of life in patients treated with IFX [6]. However, at week 50 of therapy, the improvement in patients’ quality of life was less than at week 10. In the evaluated group of patients, the improvement as assessed by mean change in DLQI was 93.75% at week 14 of therapy, increased to 96% at week 46 and decreased to 90.91% at week 96. Feldman et al. demonstrated a significant improvement in quality of life in patients during IFX induction therapy. At week 10, patients in the 3 mg/kg and 5 mg/kg IFX treatment groups had median percentage improvements in DLQI scores of 84.0% and 91%, respectively, compared with 0% in the placebo group (p < 0.001). Furthermore, there was a significant correlation between improvement in psoriasis severity as assessed by PASI and percentage change in DLQI scores [12]. Literature data indicate that patients with psoriasis taking original IFX can switch to a biosimilar drug without significant change in clinical response or additional adverse events [13]. The NOR-SWITCH study showed that switching from the original IFX to the biosimilar CT-P13 was not associated with a worse response compared with the original drug [14]. Overall, these biosimilar drugs have been shown to have equivalent efficacy, tolerability, and immunogenicity profiles compared to their original substances in patients with rheumatic diseases. However, it should be noted that data from studies involving patients with psoriasis are limited. Thus, additional studies are needed to assess drug interchangeability [15]. In this aspect, the results of our analysis provide information on the use of biosimilar drugs in clinical practice. Based on data from the PSObiosimilars registry, it was shown that PASI75 was achieved after 6 months by 80% of patients with psoriasis who had not undergone prior biologic therapy [16]. The data are comparable to those obtained from our registry. According to data from the registry DERMBIO, 73.3% of patients reported the same or better effect after switching from original IFX to a biosimilar. The rate of congruence between the self-reported and measured PASI change (same/better or worse) was 66.7%. Quality of life as measured by the DLQI remained the same or improved in 69.0% of patients in this group [17]. Gisondi et al. showed that switching from CT-P13 to SB2 was not associated with a significant change in mean PASI. However, the switch resulted in discontinuation of treatment in 10% of patients mainly due to loss of response (70%) or drug-related side effects. Loss of response, assessed as relapse of the disease (PASI > 10) was apparent at week 12. It should be noted that progressive loss of response is common during the course of biologic therapies [18]. According to our registry data, PASI100 was maintained at week 94 of therapy by 37.5% (9/24) of patients who achieved PASI100 at week 14. However, 16.7% (4/24) of patients in this group did not achieve PASI75 at week 94. Thus, the registry data confirm the loss of response during IFX treatment. It is noted that non-medical switching is currently becoming a common practice to increase access to therapies and reduce budget burdens. Clinical practice provides data that are difficult to obtain in clinical trials. Limitations of an observational study include its open design, data limit, and lack of a control group.