Introduction
Psoriasis is known as a chronic immune-mediated inflammatory disease, and its treatment is still a big challenge despite the development of therapeutic options [1–5]. These patients often desire complete skin clearance and improved long-term efficacy [6–8]. Selectively blocking the interleukin (IL)-23 or IL-17 pathways has become an important approach to treat psoriasis [9–11].
Previous findings confirmed that selective inhibition of IL-23 facilitated clinical response and molecular improvements in the skin [12–14]. Guselkumab, a fully human IgG1 lambda monoclonal antibody, can bind IL-23 and block binding and signalling through its receptor. Guselkumab was injected subcutaneously with 100 mg once every 8 weeks. Phase I and phase II revealed high levels of efficacy [9, 14]. Adalimumab, an anti-tumor necrosis factor α (TNF-α) therapy, has been widely used for the treatment of moderate-to-severe plaque psoriasis [15–18].
Recently, several studies have compared the efficacy of guselkumab with adalimumab for psoriasis, but their results show some inconsistency [9, 19, 20].
Aim
This meta-analysis of RCTs aims to assess the efficacy of guselkumab versus adalimumab for psoriasis.
Material and methods
This systematic review and meta-analysis were performed based on the guidance of the Preferred Reporting Items for Systematic Reviews and Meta-analysis statement and Cochrane Handbook for Systematic Reviews of Interventions [21, 22]. No ethical approval and patient consent were required because all analyses were based on previously published studies.
Literature search and selection criteria
We have systematically searched several databases including PubMed, Embase, Web of Science, EBSCO, and the Cochrane library for RCTs published from inception to March 2021 with the following keywords: “guselkumab”, and “adalimumab”, and “psoriasis”. The reference lists of retrieved studies and relevant reviews were also hand-searched and the above process was performed repeatedly in order to include additional eligible studies.
The inclusion criteria were presented as follows: (1) study design is RCT, (2) patients are diagnosed as psoriasis, and (3) intervention treatments are guselkumab versus adalimumab.
Data extraction and outcome measures
Some baseline information was extracted from the original studies, and they included the first author, number of patients, age, body mass index (BMI), duration of psoriasis and detailed methods in two groups. Data were extracted independently by two investigators, and discrepancies were resolved by consensus. We have contacted the corresponding author to obtain the data when necessary.
The primary outcomes were 100% or greater improvement from baseline in Psoriasis Area and Severity Index (PASI 100), PASI 90, PASI 75. Secondary outcomes included physician Global Assessment (PGA) 0/1, change in Dermatology Life Quality Index score (DLQI), and DLQI score 0/1.
Quality assessment in individual studies
The methodological quality of each RCT was assessed by the Jadad Scale which consists of three evaluation elements: randomization (0–2 points), blinding (0–2 points), dropouts and withdrawals (0–1 points) [23]. One point would be allocated to each element if they have been conducted and mentioned appropriately in the original article. The score of Jadad Scale varies from 0 to 5 points. An article with Jadad score ≤ 2 is considered to be of low quality. The study is thought to be of high quality if Jadad score ≥ 3 [24].
Statistical analysis
We assessed the odd ratio (OR) with 95% confidence interval (CI) for dichotomous outcomes (PASI 100, PASI 90, PASI 75, PGA 0/1, and DLQI score 0/1) and standard mean difference (SMD) with 95% CI for continuous outcomes (change in DLQI). Heterogeneity was evaluated using the I2 statistic, and I2 > 50% indicated significant heterogeneity [25]. The random-effects model was used for all meta-analyses. We searched for potential sources of heterogeneity for significant heterogeneity. Sensitivity analysis was performed to detect the influence of a single study on the overall estimate via omitting one study in turn or performing the subgroup analysis. Owing to the limited number (< 10) of included studies, publication bias was not assessed. Results were considered as statistically significant for p < 0.05. All statistical analyses were performed using Review Manager Version 5.3 (The Cochrane Collaboration, Software Update, Oxford, UK).
Results
Literature search, study characteristics and quality assessment
Figure 1 showed the detail flowchart of the search and selection results. 112 potentially relevant articles were identified initially. Finally, three RCTs were included in the meta-analysis [9, 19, 20].
The baseline characteristics of three included RCTs were shown in Table 1. These studies were published between 2015 and 2017, and the total sample size was 1658. Among the included RCTs, guselkumab was administered at a dose of 100 mg weeks 0 and 4, then every 8 week or 200 mg at weeks 0 and 4 and every 12 weeks thereafter. Adalimumab was taken at the dose of 80 mg week 0, then 40 mg week 1, and every 2 weeks. Among three included RCTs, all three studies reported each outcomes [9, 19, 20]. Jadad scores of the three included studies varied from 4 to 5, and all three studies have high quality based on the quality assessment.
Table 1
Characteristics of included studies
Primary outcomes: PASI 100, PASI 90 and PASI 75
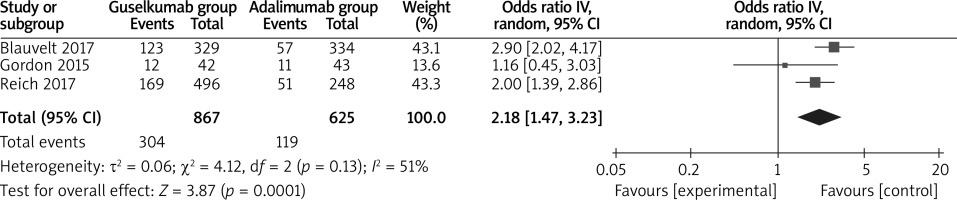
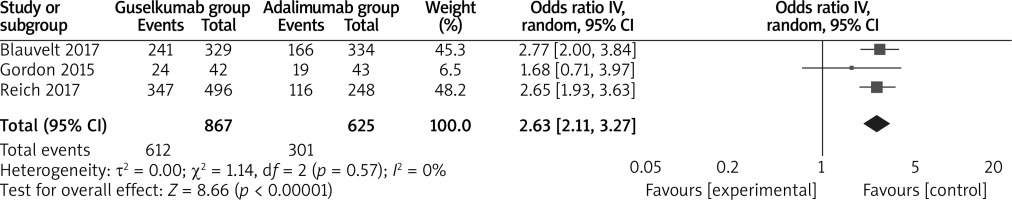
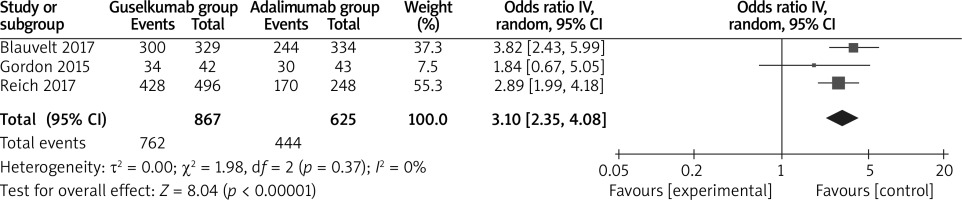
The random-effects model was used for the analysis of primary outcomes. The results found that compared to adalimumab for psoriasis, guselkumab was associated with improved PASI 100 (OR = 2.18; 95% CI: 1.47 to 3.23; p = 0.0001) with significant heterogeneity among the studies (I2 = 51%, heterogeneity p = 0.0001, Figure 2), PASI 90 (OR = 2.63; 95% CI: 2.11 to 3.27; p < 0.00001) with no heterogeneity among the studies (I2 = 0%, heterogeneity p = 0.57, Figure 3) and PASI 75 (OR = 3.10; 95% CI: 2.35 to 4.08; p < 0.00001) with no heterogeneity among the studies (I2 = 0%, heterogeneity p = 0.37, Figure 4).
Sensitivity analysis
There was significant heterogeneity for PASI 100. However, some heterogeneity was still seen when performing sensitivity analysis by omitting one study in each turn.
Secondary outcomes
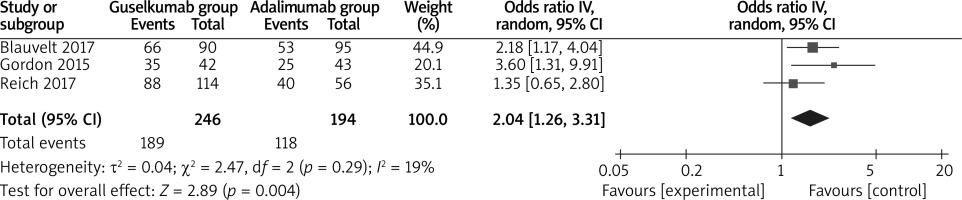
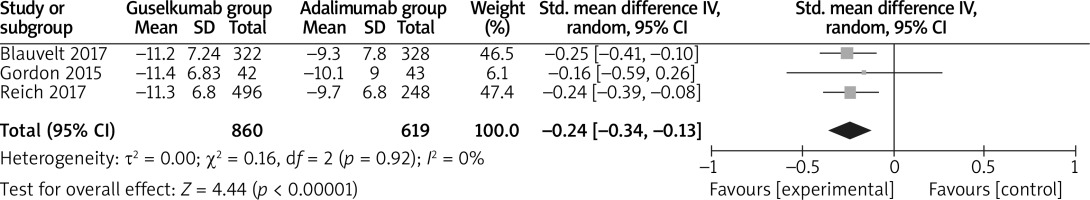
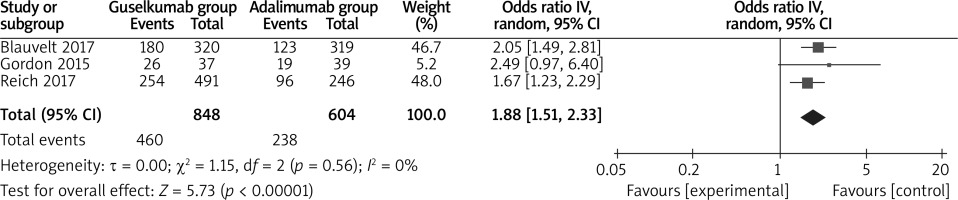
In comparison with adalimumab for psoriasis, guselkumab resulted in the obvious increase in substantially improved PGA 0/1 (OR = 2.04; 95% CI: 1.26 to 3.31; p = 0.004; Figure 5), decreased DLQI (SMD = –0.24; 95% CI: –0.34 to –0.13; p < 0.00001; Figure 6) and higher DLQI score 0/1 (OR = 1.88; 95% CI: 1.51 to 2.33; p < 0.00001; Figure 7).
Discussion
Guselkumab was documented to be highly effective in treating a broad moderate-to-severe psoriasis population [20]. VOYAGE 2 revealed that guselkumab was superior to placebo at the week-16 coprimary endpoints (IGA 0/1 and PASI 90), and was also superior to adalimumab in terms of IGA 0/1, PASI 75/PASI 90 [19]. Guselkumab had the potential in treating difficult regional psoriasis, including scalp, nails, and hands/feet, which was evidenced by improvements in the patient-reported outcomes evaluated in VOYAGE 1 [20].
Our meta-analysis confirmed that guselkumab was able to produce better treatment efficacy than adalimumab for psoriasis, which was supported by the improvement in PASI 100, PASI 90, PASI 75, PGA 0/1 and DLQI score 0/1. In addition, significant improvements in health-related quality of life (Medical Outcomes Study 36-Item Short Form) were reported in VOYAGE 2 after guselkumab treatment, which was superior to both placebo and adalimumab [19].
Guselkumab was effective in treating adalimumab nonresponders (not achieving PASI 90). After 20 weeks of guselkumab treatment, two thirds of the 112 adalimumab nonresponders who switched to guselkumab were reported to achieve PASI 90 [19]. The mechanisms mediating the broader efficacy of guselkumab is elusive in psoriasis. TNF-α and IL-17A 29 are important effector cytokines to primarily act on keratinocytes [26, 27]. IL-23 is the overarching master cytokine for psoriasis through activating T cells and inducing the production of IL-17 and IL-22 which are implicated in psoriasis pathogenesis [28, 29]. Therefore, guselkumab could provide higher efficacy and durable responses via targeting the IL-23 pathway [20].
The safety profile of guselkumab was confirmed in VOYAGE 1 and VOYAGE 2 [19, 20]. Several limitations exist here. Firstly, our analysis was based on only three RCTs, and more RCTs with a larger sample size should be conducted to explore this issue. Next, different doses and methods of guselkumab were included, which may account for the significant heterogeneity. Finally, ideal methods for guselkumab remain elusive.