Introduction
Atopic dermatitis (AD – ICD-10: L20; also called atopic eczema) is a chronic dermatosis characterized by inflammation of the skin, impaired epidermal barrier function and hypersensitivity to food and environmental allergens. The first signs and symptoms of AD usually occur in early childhood, but the disease may develop later in life [1, 2]. The most common clinical symptoms include inflammation, dry skin and itching [3]. Atopic dermatitis is characterized by a chronic and recurrent course. It is also associated with the presence of erythematous and follicular skin lesions accompanied by lichenification (Thickening of the skin with clearly marked furrows and increased skin markings.), with secondary excoriations and erosions. Itching is very often a dominant and basic symptom of the disease. The typical location of the skin lesions depends of the patient’s age [1, 2]. In children under the age of 2 years, lesions usually appear on the cheeks, scalp or forehead. In 3–11-year-olds, lesions are typically seen at sites of joint flexure (wrists, elbows and knee joints). Dorsal hand surfaces may also be affected in older children and adults. Atopic dermatitis may lead to the development of other allergic diseases, such as asthma or allergic rhinitis [4].
The etiopathogenesis of atopic dermatitis is multifactorial, including environmental and genetic factors [2, 4]. According to the results of numerous studies, the so-called “western lifestyle” associated with migration of the population or increasing urbanization promotes exposure to increasingly numerous new factors, including allergens, which may lead to the development and exacerbation of AD. It has not been clearly confirmed which environmental factors actually contribute to the development of the disease and which may act preventatively. Relevant publications provide various, mutually exclusive results, for example concerning diet, e.g. the duration of breastfeeding or timing of the introduction of solid food to the child’s diet. Another controversial issue is the place of residence (urban or rural) and exposure to domestic animals and livestock. On the one hand, it is considered that exposure to animals in early childhood reduces the risk of developing AD, but on the other hand, animals are a source of endotoxins, and exposure to microorganisms contributes to development of the disease. Factors influencing development of AD include air pollution (NO2, CO), tobacco smoke, impaired function of the protective epidermal barrier, frequent and complex antibiotic therapy, exposure to allergens, obesity and lack of physical activity [3–5].
The incidence of AD has been increasing steadily and it is estimated that presently it afflicts over 30% of the population. The onset of symptoms is in the first year of life in approximately 50% of AD cases and before5 years of age in almost 90% [1, 2]. Epidemiological data indicate differences in AD prevalence between countries. According to the International Study of Asthma and Allergies in Childhood – Phase One(ISAAC), among children aged 6–7 and 13–14 years, AD was the most prevalent in Australia, the United Kingdom and Scandinavia (20%) and the least prevalent in China (2%) [6, 7]. The European Community Respiratory Health Survey II (ECRHS II),conducted among 20–44-year-olds in 25 European centres and Portland in the United States, estimated the lowest prevalence rate in Switzerland (2.2%) and the highest rate in Estonia (17.6%) [8]. At present, the worldwide prevalence of AD is estimated at 15–20% among children and 1–3% in adults [9, 10]. Although AD is considered a childhood disease, in many cases it persists during the entire lifetime. Moreover, AD is increasingly often diagnosed among people over 60 years of age [11]. In Poland, according to the results of the ECAP (Epidemiology of Allergic Diseases in Poland) study, the mean prevalence of AD in the study population (N = 4783) was 6.5% (8.7% in 6–7-year-olds, 9.0% in 13–14-year-olds and 3.6% in adults) [12, 13]. The same study found that AD was diagnosed more often among women (OR = 1.52; 95% CI:1.30–1.77), people living in big cities (OR = 2.23; 95% CI:1.61–3.09), people with atopic mothers (OR = 2.07; 95% CI: 1.72–2.48), people with atopic fathers (OR = 2.00; 95% CI: 1.61–2.49), and people with higher education (OR = 1.61; 95% CI: 1.11–2.32) and higher socio-economic status (OR = 1.35; 95% CI: 1.04–1.74) [13]. The prevalence of AD based on data from the international literature is presented in Table 1 [12–21].
Table 1
The prevalence rates of AD according to available studies
| Reference, year | Year of the study | Country | Study group | Prevalence | 95% CI | Age of the subjects |
|---|---|---|---|---|---|---|
| Samoliński et al. 2014 Sybilski et al. 2015 [12, 13] | 2006–2009 | Poland | 4,783 | 8.7% 9% 3.6% | No data | 6–7-year-olds 13–14-year-olds 20–44-year-olds |
| Pekkarinen et al. 2007 [14] | 2003 | Finland Russia | 344 children 344 mothers 427 children 284 mothers* | 91/344 (26.5%)** 64/344 (18.6%) 17/427 (4.0%) 9/284 (3.2%) | No data | Schoolchildren, aged 6–16 years, and their mothers |
| Priftanji et al. 2001 [15] | No data | Albania UK | 1,114 1,346 | 15% 17.8% | 0.8 (0.6–1.1) 95% CI | 9–11-year-olds |
| Björkstén et al. 1998 [16] | No data | Scandinavia and Eastern Europe | 79,000 | 1.5–35.7% 1.3–48.8% | No data | 6–7-year-olds 13–14-year-olds |
| Engebretsen et al. 2017 [17] | 1996–2002 | Denmark | 52,950 | 15.0% | No data | Birth cohort: 6 and 18 months |
| Shaw et al. 2011 [18] | 2003 | USA | 102,353 | 10.7% | No data | ≤ 17 years |
| Radtke et al. 2017 [19] | 2009 | Germany | 1,349,671 | 3.67% | No data | ≥ 18 years |
| Bergmann et al. 2016 [20] | 2008–2011 2003–2006 | Germany | 7,988 17,641 | 3.5% 13.2% | No data | 18–79-year-olds 0–17-year-olds |
| Barbarot et al. 2018 [21] | 2016 | United States, Canada, France, Germany, Italy, Spain, United Kingdom, Japan | 90,658 | 2.1–4.9% | No data | 18–65-year-olds |
Aim
The purpose of this study is to analyse the recorded prevalence of AD (diagnoses of AD reported to the Polish National Health Fund (NHF)) and to estimate direct costs of medical care for AD in the public health system based on information from the NHF database.
Material and methods
The study used a method of desk research, namely, secondary statistical analysis of data reported to the National Health Fund in 2008–2017. This method has been successfully used previously in the analysis of NHF data for epidemiological purposes [22–25]. The National Health Fund is a public payer which has a single-payer status for all healthcare services financed from public funds in Poland.
The NHF databases contain the data of all patients in Poland who received healthcare services related to diagnosed AD, financed from public funds. For the purposes of the analysis, AD was defined by the relevant ICD-10 diagnosis codes: L20 – atopic dermatitis, L20.8 – other atopic dermatitis, and L20.9 – atopic dermatitis, unspecified. The healthcare services related to ICD-10 diagnosis code L20.0 (L20.0 – Besnier prurigo (prurigo gestationis of Besnier) is a unique rare disease of pregnant women (afflicting about 2% of pregnant women) with a different pathomechanism and course (it occurs only during pregnancy) were not included in the present analysis.
The NHF databases were accessed for information about each treated patient with the ICD-10 L.20 codes (with specific subcodes) given as the principal diagnosis or a comorbidity in healthcare benefit settlement reports. A person with AD was defined as a patient with at least one healthcare service for AD recorded in the NHF database in a given calendar year. The data used in the analysis were anonymized in compliance with the Polish Act on the Protection of Personal Data.
The analysis of the costs was based on the expenditures reported to the NHF by healthcare service providers. The total cost of healthcare services related to AD and incurred by the public payer was calculated for each patient who had received such benefits (the total amount of costs of healthcare services related to this diagnosis). The total cost included the cost of the medical procedures rendered, but not patient-incurred cost of prescription drugs.
The prevalence rates in 2017 were calculated using the NHF data and the population estimates obtained from the Central Statistical Office of Poland (GUS) (as at 31.12.2017) (Ludnoœæ. Stan istrukturaludnoœciorazruchnaturalny w przekrojuterytorialnym. Stan w dniu 31 XII 2017 r. https://stat.gov.pl/obszary-tematyczne/ludnosc/ludnosc/ludnosc-stan-i-struktura-oraz-ruch-naturalny-w-przekroju-terytorialnym-w-2017-r-stan-w-dniu-31-xii,6,23.html [20.06.2018]). For the purposes of this study, the AD prevalence rate was defined as the ratio of patients who received healthcare services for AD in outpatient or hospital settings to the total size of the population in a given region.
No ethics committee approval was required as the study was non-invasive and used data collected by a public institution.
Exchange rates were defined on the basis of data from the National Bank of Poland (NBP) (http://www.nbp.pl/home.aspx?f=/kursy/arch_a.html). Purchasing Power Parities (PPP) were based on data from the Organisation for Economic Co-operation and Development (OECD) (http://stats.oecd.org/Index.aspx?datasetcode=SNA_TABLE4).
Results
Epidemiology
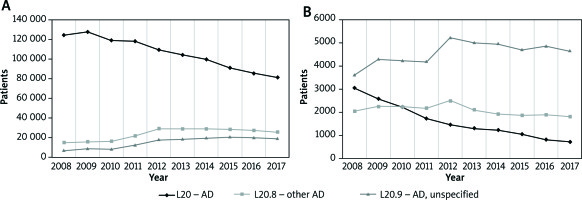
In 2017, there were 259.4 thousand outpatient consultations for 120.4 thousand patients with a diagnosis of AD (an average of 2.4 consultations per person). By comparison, in 2008, there were 315.3 thousand consultations for 143.2 thousand patients (an average of 2.2 consultations per person). In 2017, there were altogether 7.7 thousand hospitalizations for AD in 7.0 thousand patients (an average of 1.1 hospitalizations per person). In 2008, there were 11.6 thousand hospitalizations for 9.1 thousand patients (an average of 1.3 hospitalizations per person). The detailed data concerning the number of patients treated in outpatient settings and hospital by diagnosis are presented in Figures 1 A and B.
Figure 1
A – The number of patients treated in specialist outpatient care due to conditions with the ICD-10 diagnosis codes L20, L20.8 and L20.9 in 2008–2017. B – The number of patients hospitalized for conditions with the ICD-10 diagnosis codes L20, L20.8 and L20.9 in 2008–2017
Among patients receiving healthcare services for AD in 2017, women constituted 55.2%. In 2008–2017, this percentage varied between 54.1% and 55.3%. In 2017, 66.4% of patients with AD were people residing in urban areas.
In 2017, the prevalence rate of AD in the Polish population was 32.5 per 10,000 inhabitants (34.7/10,000 for women and 30.1/10,000 for men). The highest prevalence rates were found in Podlaskie Voivodeship (38.8/10,000) and Kujawsko-Pomorskie Voivodeship (38.8/10,000) and the lowest in Lubuskie Voivodeship (26.9/10,000) and Mazowieckie Voivodeship (27.2/10,000).
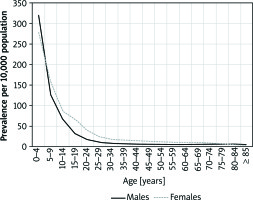
The highest prevalence was observed in the youngest age groups, in both girls and boys (300.4/10,000 in children up to 4 years of age and 141.3/10,000 in 5–9-year-olds). The prevalence rate decreased with age and AD was the least prevalent in patients over 85 years of age (3.6/10,000). The details are presented in Figure 2.
Direct costs
In 2017, the NHF spent PLN 26.5 million (EUR 6.2 million) on health care services for AD. This represented a 6.6% decrease in expenditure in comparison with 2016 (the 2016 figure represented a 14.3% increase compared to the previous year). In 2008–2017, NHF expenditure on AD treatment varied between PLN 19.9 million (in 2008) and PLN 28.4 million (in 2016). After conversion into EUR (accounting for the annual exchange rate), the highest expenditure was in 2016 (EUR 6.5 million) and the lowest in 2011 (EUR 5.4 million). The detailed data are presented in Table 2.
Table 2
The NHF expenditure on AD treatment in hospital and specialist outpatient care in 2008–2017
| Parameter | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 |
|---|---|---|---|---|---|---|---|---|---|---|
| Hospital | 9,358,450 | 10,877,493 | 10,140,054 | 9,806,248 | 11,885,012 | 11,295,922 | 10,949,232 | 10,648,217 | 13,969,996 | 12,504,860 |
| SOC* | 10,522,216 | 12,568,349 | 11,839,575 | 12,352,818 | 13,132,495 | 13,014,210 | 13,148,887 | 14,220,229 | 14,442,243 | 14,041,880 |
| Total | 19,880,666 | 23,445,842 | 21,979,629 | 22,159,066 | 25,017,507 | 24,310,132 | 24,098,119 | 24,868,446 | 28,412,239 | 26,546,740 |
| Total (USD) | 8,251,978 | 7,523,857 | 7,288,400 | 7,477,582 | 7,681,150 | 7,691,133 | 7,637,830 | 6,596,230 | 7,205,559 | 7,027,223 |
| Total (EUR) | 5,653,377 | 5,418,123 | 5,502,335 | 5,378,675 | 5,977,899 | 5,791,574 | 5,757,937 | 5,943,843 | 6,512,834 | 6,235,142 |
| Total (PPP GDP**) | 10,791,555 | 12,543,860 | 12,189,228 | 12,300,861 | 13,928,267 | 13,796,544 | 13,636,776 | 13,961,985 | 15,894,159 | 14,896,809 |
In 2017, NHF expenditure varied considerably between voivodeships, partially due to differences in the number of inhabitants. The highest amount was allocated for this purpose in the Mazowieckie Voivodship (PLN 2.9 million), and the lowest in Lubuskie, Opolskie and Œwiêtokrzyskie Voivodeships (between PLN 0.5 and 0.6 million). In total, the costs of outpatient care for AD accounted for 52.9% of the total cost. This percentage differed considerably between individual voivodeships, from 69.7% in Œwiêtokrzyskie Voivodeship to 21.4% in Warmiñsko-Mazurskie Voivodeship. The highest expenditure per patient was recorded in Warmiñsko-Mazurskie Voivodeship (PLN 394/person) and the lowest in Œwiêtokrzyskie Voivodeship (PLN 155/person). Regarding the entire Polish population, mean annual NHF expenditure was PLN 0.69 per person. The lowest amount was recorded in Œwiêtokrzyskie Voivodeship (PLN 0.47 per person) and the highest in Warmiñsko-Mazurskie Voivodeship (PLN 1.25 per person). The detailed data are presented in Table 3.
Table 3
The NHF expenditure on AD treatment in hospital and specialist outpatient care in 2017 by voivodeship
Discussion
In the period of interest (2008-2017), the number of patients treated for AD (according to ICD-10 diagnosis codes: L20, L20.8 and L20.9) in public health care in Poland ranged from 131.5 to 162.2 thousand people annually. The incidence of AD has been steadily increasing in Poland and worldwide as documented by the numerous epidemiological studies included [3–21]. The observed increase in AD prevalence may be associated with improved diagnostic accuracy. The diagnosis of AD in clinical practice should be based on internationally recognized diagnostic criteria, which include the clinical characteristics of skin lesions, historical data and selected laboratory parameters [26]. Atopic disease might sometimes be overdiagnosed given that there are many other skin diseases with similar clinical features. This is especially true for children and for clinical cases in which lesions are relatively small [26].
The rates calculated in this study seem to be underestimated when compared to the prevalence estimates reported in the studies quoted. According to our estimates based on NHF data, recorded prevalence of AD accounts for 0.32% (32/10,000) in the Polish population. In the present study, the estimated prevalence rates were as follows: 1.41% (141/10,000) in 5–9-year-olds, 0.75% (75/10,000) in 10–14-year-olds and 0.15% (15/10,000) among young adults (20–44 years of age). The discrepancy between our results and data from earlier studies may be due, among others, to the specific research methodology and scope of the present analysis. The present study is based on official data (NHF) collected for the whole country. However, it should be noted that access to specialists (including allergists and dermatologists) is limited in Poland. Some patients may use private healthcare or self-treat and, as a result, the calculated prevalence rates may be underestimated.
According to the present analysis, AD prevalence was slightly higher among women than men (34.7 vs. 30.1/10,000; OR = 1.15). The results are consistent with estimates from the ECAP study [12], in which women were more often diagnosed with AD (OR = 1.52; 95% CI: 1.30–1.77). Research from other European countries confirms this relationship. Among teenagers, AD was more common in girls in countries such as Germany [27], Spain [28] and Italy [29]. In adults, a similar correlation was also observed in the study of Harrop et al. (covering 25 European countries and Portland, USA) [8].
According to our estimates, the highest prevalence was observed in the youngest age groups and similar results have been presented in other studies [12, 13, 18, 30]. The present analysis demonstrated substantial variations in the prevalence of AD between voivodeships. In men, the prevalence rate per 10,000 population varied between 24.7 and 36.2, and in women between 28.4 and 42.4, depending on the geographical region. The epidemiological data from the literature demonstrated considerable differences in the prevalence of AD both between countries and between geographical regions in a given country [8, 14, 15].
The present analysis demonstrated an increase in the costs incurred by the public payer for treatment of AD (although fluctuation is present). The estimates of direct costs of AD available in the literature vary. It is impossible to compare them with the present study due to methodological differences (mostly questionnaire-based surveys rather than publications based on official data).
The analysis was based on the data available from the NHF. The NHF database was established for accounting purposes rather than to support research (such as epidemiological studies). The NHF register does not include information on private healthcare services. As a result, the calculated prevalence rates may be underestimated (a person with AD was defined as any patient who in a given year received a healthcare service related to the diagnosis, either as the principal disorder or comorbidity). On the other hand, not every patient with an initial suspicion of AD was ultimately diagnosed with this disease. This may result in overestimating the prevalence rates.
The analysis is based on official data covering the entire country (the entire Polish population). The present study analyses the information for 2008–2017, i.e. 10 reporting periods. In each reporting period, data were collected in the same manner, thus ensuring a high level of standardisation and comparability of the results.
Conclusions
The estimated prevalence rates of AD in Poland based on NHF data are significantly lower than those reported in previous epidemiological studies conducted in Poland and worldwide. This may indicate that the prevalence of AD in the Polish population is underestimated or there are no adequate disease control measures in patients with a confirmed diagnosis. Some patients are treated in primary or private health care facilities. Further studies are required to confirm this hypothesis. The direct costs incurred by the NHF in relation to AD increased steadily in the years 2008–2017 while the number of provided services decreased.