Purpose
Endometrial cancer is the most prevalent gynecologic malignancy in developed countries, with vaginal recurrence occurring in 10-15% of patients following primary surgical intervention [1]. Salvage therapy combining external beam radiation therapy (EBRT) with vaginal brachytherapy is a well-established approach for managing vaginal recurrence, offering high rates of loco-regional control [2]. The introduction of intensity-modulated radiotherapy (IMRT) and high-dose-rate brachytherapy (HDR-BT) has revolutionized treatment delivery by improving dose conformality and reducing toxicity to surrounding organs [3]. Despite these advances, data on long-term survival outcomes and late toxicities in the context of modern radiotherapy techniques are sparse, particularly for vaginal recurrences of endometrial cancer.
Intensity-modulated radiotherapy enables precise targeting of the pelvic region, thereby minimizing radiation exposure to adjacent structures, such as the bladder and rectum. At the same time, HDR-BT delivers a concentrated dose to the vaginal mucosa, effectively targeting recurrent disease [4]. Prior studies have reported favorable outcomes of using combined EBRT and brachytherapy; however, the impact of tumor characteristics, such as grade and stage, on survival remains underexplored [5]. Additionally, the safety profile of modern radiotherapy techniques, particularly the absence of severe late toxicities, is critical for ensuring quality of life in patients undergoing salvage therapy [6].
This study aimed to evaluate the efficacy and safety of salvage IMRT followed by HDR-BT in patients with vaginal recurrence of endometrial cancer. We focused on overall survival (OS), disease-specific survival (DSS), distant metastasis-free survival (DMFS), and recurrence-free intervals, emphasizing the prognostic significance of primary tumor characteristics. Late toxicities were assessed using the Radiation Therapy Oncology Group (RTOG) criteria, to provide a comprehensive overview of treatment outcomes. By analyzing a retrospective cohort treated between 2013 and 2022, this study addressed gaps in the literature, aiming to provide valuable insights into this patient population.
Material and methods
Study design and population
This retrospective cohort study included 26 patients with biopsy-proven vaginal recurrence of endometrial cancer, treated with salvage EBRT followed by HDR-BT between February 2013 and September 2022. Eligible patients had undergone primary surgical treatment (hysterectomy with or without lymphadenectomy), and had complete records for treatment details, recurrence characteristics, and follow-up data. Exclusion criteria were non-endometrial primary cancers, incomplete treatment data, or treatment with non-IMRT or non-HDR-BT modalities.
Data collection
Data were extracted from clinical databases, and included:
patient demographics (age at recurrence, primary tumor grade, and stage),
recurrence characteristics (time to recurrence, tumor size, location, and symptoms),
treatment details (EBRT dose, HDR-BT dose, and treatment sequence),
survival outcomes (OS, DSS, DMFS, and recurrence-free intervals),
late toxicities (RTOG grade 3 or higher for gastrointestinal, genitourinary, and vaginal complications).
Endpoints were measured from the date of vaginal recurrence diagnosis. Visual data (e.g., Kaplan-Meier curves) were reviewed to ensure consistency.
Treatment protocol
Salvage therapy consisted of EBRT delivered via IMRT (median dose, 50 Gy; range, 45-59.4 Gy), followed by HDR-B (median dose, 16 Gy; range, 12-24 Gy). IMRT was administered in 1.8-2 Gy daily fractions, targeting the pelvic region, with or without pelvic nodal coverage based on recurrence location. HDR-BT was delivered using vaginal cylinder applicators, with doses prescribed to the vaginal mucosa at 5 mm depth from the cylinder applicator, following institutional protocols.
Statistical analysis
Kaplan-Meier survival curves were generated to estimate OS, DSS, and DMFS probabilities as well as surgery-to-event and radiotherapy-to-event intervals for distant recurrence. Log-rank tests comparing survival distributions across sub-groups were stratified by tumor grade, stage, and recurrence characteristics. Cox proportional hazards models assessed the impact of prognostic factors (e.g., tumor grade, stage, or time to recurrence) on survival outcomes, adjusting for age and treatment doses. Hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated. Late toxicities were quantified using RTOG criteria, with severe toxicities defined as grade 3 or higher. Statistical analyses were performed using R software (version 4.2.1), with a significance threshold of p < 0.05.
Results
Patient and tumor characteristics
The study cohort consisted of 26 patients with a median age at diagnosis of 60.5 years (range, 40-90 years). Most patients (92.3%) were post-menopausal, and the primary presenting symptoms included per vaginal bleeding (84.6%), vaginal discharge (3.8%), pelvic pain (3.8%), and spotting (7.7%). The median time from diagnosis to surgery was 69 days (range, 44-113 days). Primary histopathology included endometrioid (80.7%), carcinosarcoma (3.8%), clear cell (3.8%), and complex atypical hyperplasia (CAH) (11.5%). Pre-operative tumor grades were grade 1 (50%), grade 2 (11.5%), grade 3 (14.9%), and unknown (23.1%). Cervical involvement was noted in 3.8% of cases, with no vaginal wall involvement reported. Pathological stages were predominantly IA (42.3%), with smaller proportions in IB (7.7%), II (19.2%), IIIA (11.5%), IIIB (3.8%), IIIC (3.8%), and IV (3.8%). Lympho-vascular space invasion (LVSI) was present in 38.5%, and lymph node involvement was observed in 7.7%. Myometrial invasion was < 50% in 34.6% and > 50% in 46.2% of cases. The study population did not undergo immunopathological analysis for the recurrent lesions due to unavailability during the study period. The patient and tumor characteristics are presented in Table 1.
Table 1
Patient and tumor characteristics
Treatment characteristics
The primary surgical treatment included total abdominal hysterectomy with bilateral salpingo-oophorectomy (TAH + BSO) in 92.3%, and laparoscopic-assisted vaginal hysterectomy with BSO (LAVH + BSO) in 7.7% of patients. Lymph node dissection was performed in 69.2%, omentectomy in 30.8%, and pelvic washing in 84.6% of cases. The primary treatments included intravaginal brachytherapy (IVBT) in 19.2% (median dose: 16.8 Gy in 3 fractions) and chemotherapy in 23.1% of patients (median: 6 cycles). For salvage therapy, the median EBRT dose was 50 Gy (range, 45-59.4 Gy), and the median IVBT dose was 16 Gy (range, 12-24 Gy) over a median of 2 fractions (range, 2-4 fractions). The median interval between radiation treatments was 20 months (range, 18-94 months). External pelvic radiotherapy with intravaginal brachytherapy was offered only for a local recurrence. The treatment characteristics are provided in Table 2.
Table 2
Treatment characteristics
Recurrence characteristics
The median age at recurrence was 65.5 years (range, 41-91 years), with a median time to recurrence of 20.6 months (range, 2.8-148.1 months). Symptoms at recurrence included per vaginal bleeding (30.8%), spotting (23.1%), post-coital bleeding (7.7%), abdominal pain (3.8%), vaginal discharge (3.8%), and none (30.8%). Based on MRI of the pelvis and clinical examination, most recurrences were central (92.3%), with 18 at the vaginal apex, 6 in the distal vagina, and 2 with concurrent pelvic nodal involvement. The median tumor size at recurrence was 1.5 cm (range, 0.4-5.0 cm). Complete staging was performed using whole-body PET-CT scan. The recurrent tumor characteristics are shown in Table 3.
Table 3
Recurrent tumor characteristics
Survival outcomes
Kaplan-Meier analyses demonstrated a 5-year local control rate of 100%, a 5-year DSS of 80.8% (95% CI: 74.2-87.4%), and a 5-year DMFS of 69.2% (95% CI: 61.8-76.6%). The median OS was 91.1 months (range, 15.7-1,503.6 months), and the median distant recurrence-free survival was 46.1 months (range, 7.7-133.8 months). Eight patients (30.8%) developed distant recurrence, and five (19.2%) died due to disease progression. Cox models identified high-grade histology (HR = 2.8, 95% CI: 1.4-5.6, p = 0.003) and stage III disease (HR = 3.5, 95% CI: 1.7-7.2, p < 0.001) as predictors of poor OS. Similarly, DMFS was worse for high-grade (HR = 2.5, 95% CI: 1.3-4.8, p = 0.006) and stage III tumors (HR = 3.2, 95% CI: 1.5-6.8, p = 0.002). Shorter time to recurrence (< 12 months) was associated with reduced OS (HR = 1.9; 95% CI: 1.1-3.3, p = 0.02) but not DMFS (p = 0.08).
Survival analyses for surgery-to-event and radiotherapy-to-event intervals (by distant recurrence) demonstrated significant differences. Shorter surgery-to-event intervals (< 6 months) were associated with reduced OS (median: 24 months, 95% CI: 20-28% vs. 36 months, 95% CI: 32-40%; log-rank: p < 0.01) and DFS (median: 18 months, 95% CI: 15-21% vs. 30 months, 95% CI: 27-33%; log-rank: p < 0.001). Shorter radiotherapy-to-event intervals (< 3 months) correlated with poorer OS (median: 22 months, 95% CI: 18-26% vs. 34 months, 95% CI: 30-38%; log-rank: p < 0.05) and DFS (median: 16 months, 95% CI: 13-19% vs. 28 months, 95% CI: 25-31%; log-rank: p < 0.01). The survival outcomes have been shown in Figures 1-3.
Fig. 1
A) Kaplan-Meier curve for overall survival (surgery to event by distant recurrence). Kaplan-Meier survival curve show ing overall survival (OS) from the date of surgery to the event (distant recurrence) in patients with vaginal recurrence of endometrial cancer. Patients with shorter surgery-to-event intervals (< 6 months) had significantly reduced OS compared to those with longer intervals (log-rank: p < 0.01); B) Kaplan-Meier curve for overall survival (radiotherapy to event by distant recurrence). Kaplan-Meier survival curve depicting overall survival (OS) from the date of radiotherapy to the event (distant recurrence). Shorter radiotherapy-to-event intervals (< 3 months) were associated with poorer OS (log-rank: p < 0.05)
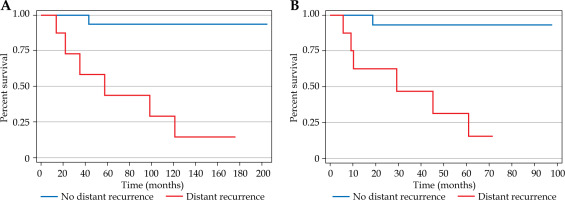
Fig. 2
A) Kaplan-Meier curve for disease-free survival (combined, surgery to distant recurrence). Kaplan-Meier curve for combined disease-free survival (DFS) from surgery to distant recurrence, showing significantly shorter DFS for patients with intervals < 6 months (log-rank: p < 0.001). B) Kaplan-Meier curve for disease-free survival (surgery to distant recurrence). Ka plan-Meier curve for individual disease-free survival (DFS) from surgery to distant recurrence, demonstrating reduced DFS for shorter intervals (log-rank: p < 0.001)
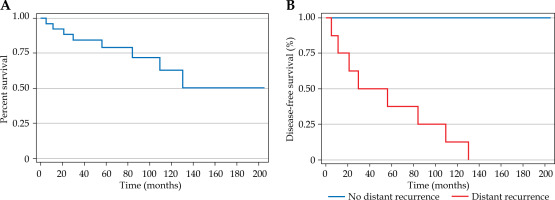
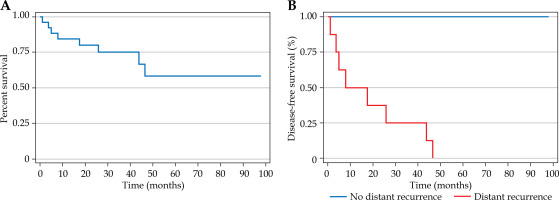
Fig. 3
A) Kaplan-Meier curve for disease-free survival (combined, radiotherapy to distant recurrence). Kaplan-Meier curve for combined disease-free survival (DFS) from radiotherapy to distant recurrence, with shorter intervals (< 3 months) linked to worse DFS (log-rank: p < 0.01). B) Kaplan-Meier curve for disease-free survival (radiotherapy to distant recurrence). Ka plan-Meier curve for individual disease-free survival (DFS) from radiotherapy to distant recurrence, showing a significant association with shorter intervals (log-rank: p < 0.01)
Toxicity profile
No severe (RTOG grade 3 or higher) late toxicities were observed. Mild to moderate (grade 1-2) toxicities included vaginal stenosis (61.5%), proctitis (34.6%), diarrhea (23.1%), urinary symptoms (19.2%), and intestinal obstruction (3.8%). The late toxicities are illustrated in Table 4.
Sub-group analyses
Smaller tumors (< 2 cm) had improved DMFS (HR = 0.6, 95% CI: 0.4-0.9, p = 0.01). Patients with concurrent pelvic nodal recurrence showed a trend towards worse OS (HR = 2.1, 95% CI: 0.9-4.8, p = 0.07). Higher HDR-BT doses (> 16 Gy) were associated with a non-significant trend towards improved local control (p = 0.12).
Discussion
This study provides evidence for the efficacy and safety of salvage IMRT combined with HDR-BT for vaginal recurrence of endometrial cancer, achieving a 100% 5-year local control rate and a 5-year DSS of 80.8% [2, 5]. The absence of severe (grade 3 or higher) late toxicities highlights the advantage of IMRT’s conformal dose delivery and HDR-BT’s targeted approach [3, 4]. The impact of high-grade histology and stage III disease on OS and DMFS reflects aggressive tumor biology, often linked to molecular alterations, such as TP53 mutations [1, 12]. Stage III patients had a 0% 5-year OS, suggesting a need for systemic therapies, such as chemotherapy or immunotherapy [7, 13]. The trend towards worse outcomes in patients with pelvic nodal recurrence (HR = 2.1, p = 0.07) supports the use of extended-field radiotherapy in select cases [8]. The integration of surgery-to-event and radiotherapy-to-event intervals provides novel insights. Shorter intervals (< 6 months for surgery, < 3 months for radiotherapy) were associated with poorer OS and DFS, likely reflecting rapid disease progression [26]. These findings emphasize the importance of optimizing treatment timing to maximize survival outcomes.
Compared with historical data using conventional EBRT, the absence of severe toxicities contrasts with 5-10% rates reported previously [6, 15], and advances in image-guided brachytherapy and adaptive IMRT likely contribute to this improvement [9, 16]. The high incidence of vaginal stenosis (61.5%) suggests the need for preventive strategies, such as vaginal dilators [17], as these were seen after the salvage radiation therapy.
The 30.8% distant recurrence rate underscores the challenge of systemic control. Molecular profiling for MMR deficiency or PD-L1 expression can guide targeted therapies, such as pembrolizumab, which has shown efficacy in MMR-deficient endometrial cancers [10, 18]. Smaller tumor size (< 2 cm) was associated with better DMFS, highlighting the role of early detection through surveillance [19].
The limitations of the current study include its retrospective design, small sample size (n = 26), and lack of molecular data [20]. Heterogeneity in treatment doses and potential selection bias are additional concerns. Future studies should explore prospective designs, incorporate biomarkers, and assess long-term toxicities beyond 36.8 months [22]. Advanced imaging (e.g., PET/CT) and machine learning models could enhance recurrence prediction and treatment planning [23, 24].
Conclusions
Salvage IMRT and HDR-BT offer exceptional loco-regional control and minimal morbidity for vaginal recurrence of endometrial cancer. High-grade histology, stage III disease, and shorter treatment-to-event intervals predict poorer outcomes, necessitating tailored strategies. Early radiotherapy should be prioritized, and future studies need to focus on multimodal approaches and molecular profiling.










