Introduction
Exhaled breath condensate (EBC) is a liquefied vapour solution, obtained by the passing of warm humid breath through a cooled condenser device. Since it originates from air passaging through the respiratory system, EBC contains a mixture of non-volatile compounds, produced and released in that system [1]. It has been proven that the composition of EBC samples reflects the current pathophysiological status of the respiratory tract. Increased amounts of various biomarkers of oxidative stress or mediators of inflammation in EBC mainly correlate with intensity of inflammatory process [2]. Therefore, EBC assessment may be used as a valuable research and diagnostic tool, especially since the acquisition of EBCs requires minimal patient cooperation and may be used in younger children or patients in poor general condition [3].
Despite extensive studies on EBC usefulness in clinical research, the knowledge about this method is still incomplete. Sometimes various studies provide unexpected or astonishing results. Thus, in our previous study we have found that EBC samples collected from asthmatic children contained significant quantities of some regulators of angiogenesis [4]. In order to assess the biological activity of these factors, we conducted a series of in vitro experiments. EBC samples from asthmatic and healthy children were added to in vitro cultures of murine and human endothelial cells followed by the assessment of metabolic activity and viability of cultured cells [5].
Although EBC samples from patients with asthma contained detectable amounts of some regulators of angiogenesis, they did not stimulate proliferation or reveal any other significant influence on endothelial cells, used in our experimental model. Surprisingly, incubation of both murine and human endothelial cell cultures with breath condensates from healthy children led to cell death. The observed effect was associated with increased binding of annexin V, which is a typical feature of apoptosis, the programmed cell death. This effect was dose-dependent. Moreover, as found in further experiments with other cell types, including human lung fibroblasts (HLF) and human keratinocytes (HaCaT), it was tissue-nonspecific (i.e. not limited to endothelial cells only) [6].
Since the aforementioned observation might have significant clinical and therapeutic implications, it is of great importance to clarify a detailed mechanism of apoptosis triggered by EBC from healthy children. Moreover, we hypothesized that the determination of the pathway components could possibly allow further “retrograde” identification of the factor responsible for the cytotoxic effect of EBC.
Aim
The aim of the present study was to assess expression profiles of selected apoptosis pathway-related proteins in tested human and murine cell cultures exposed to samples of exhaled breath condensates from healthy children.
Material and methods
EBC samples collection and preparation
The study was performed with already deposited exhaled breath condensate samples from 10 healthy children, which were excessively collected for the recent project [7, 8], according to approval (KB/246/2012) of the Bioethics Committee at the Medical University of Warsaw. The clinical characteristics of the patients were already described in our previous publication [4]. The EBC samples were aliquoted and stored at –70°C to avoid repeated freezing-thawing cycles.
Cell cultures
The normal human lung fibroblasts (HLF), purchased from Cell Applications Inc. (San Diego, CA), passages 4th to 8th, and murine endothelial cell line C166 (ATCC® CRL-2581™) from American Type Culture Collection (ATCC, Manassas, VA), were used for in vitro studies. The cells were maintained in Fibroblast Growth Medium (FGM) or Endothelial Cell Growth Medium (ECGM), respectively, both from Cell Applications Inc. (San Diego, CA), in 25 cm2 culture flasks, under standard conditions of 5% CO2 in humidified air, at 37°C.
For the screening of intracellular factors, presumably involved in the cytotoxic effect of tested breath condensates, both HLF and C166 cells were exposed to EBC. Briefly, cells were trypsinized from subconfluent (approximately 80%) cultures, resuspended in the respective fresh medium and seeded onto 12-well flat-bottom culture plates at a density of 105 cells/well, in 1 ml.After 1 h, to allow a cell attachment to the plastic, 60 µl of EBC samples or double distilled water (DDW) used as a test control, were added to cultures followed by their further incubation for the next 4 h, at 37°C. At the end of incubation the cultures were inspected and photographed using phase contrast microscope (Zeiss Primovert, Carl Zeiss Microscopy GmbH, Gottingen, Germany).
Afterwards, the cells were carefully washed with cold phosphate buffered saline (PBS) and subjected to further assessment in protein microarrays.
Protein microarrays
The analysis was performed using Proteome Profiler™ Mouse Apoptosis Array Kit for C166 cells and Proteome Profiler™ Human Apoptosis Array Kit for lung fibroblasts, according to detailed protocols provided by the manufacturer (both kits from R&D Systems Inc., Minneapolis, MN). In brief, cells were lysed directly on the culture plates using Lysis buffer, provided in a kit, which was supplemented with a cocktail of proteinase inhibitors (Complete Mini, Roche Diagnostics, Mannheim, Germany). Cell lysates were vortexed and homogenized by their passing several times through a tip, and applied onto respective nitrocellulose membranes spotted with capture antibodies. After overnight incubation at 4°C, the membranes were washed and incubated with streptavidin-horseradish peroxidase conjugate. Then, stabilized luminol in a hydrogen peroxide solution was applied and membranes were scanned in the chemiluminescence detection system (FluorChem E system, ProteinSimple, San Jose, CA).
The signal density for each spot was measured using ImageJ software 1.51p (National Institutes of Health scientific image-analysis program) [9, 10]. The relative expression of assessed proteins was shown as a percent of reference signal density, calculated as a ratio of mean signal density of the tested signal to mean density of reference spots. Each test was repeated twice.
Results
The murine endothelial cells (C166), when treated with EBC samples from healthy children, rapidly revealed significant morphological changes. Within 4-hour incubation these changes involved all cells in a culture and included formation of vacuoles, cell shrinkage and detachment from a culture plate (Figure 1 A).
Figure 1
A – The culture of murine endothelial cell line (C166) in the phase contrast microscope after 4-hour incubation with double distilled water (DDW), used as a test control (left picture) or with a representative EBC sample (right picture). B – The membranes of Mouse Apoptosis Array exposed to C166 cell lysates incubated for 4 h with DDW (left picture) or a representative EBC sample (right picture). Each pair of spots in white frames represents a respective target molecule. The numbers at the frames correspond to respective molecules on graph C: 1 – SMAC/Diablo, 2 – Hsp70, 3 – Hsp27. The brightest spots correspond to reference markers (dashed line frames). C – Relative expressions of selected apoptosis pathway-related proteins with the highest signal density, calculated as the percent of reference spots
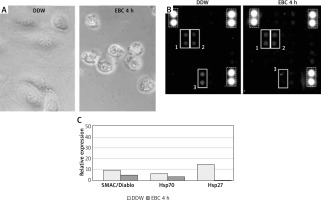
The protein microarray analysis of DDW-treated control lysates of C166 cells revealed low quantities of pro-apoptotic protein SMAC (second mitochondria-derived activator of caspases), also known as Diablo (direct IAP binding protein with low pI). Furthermore, small amounts of two members of the heat shock protein (Hsp) family – Hsp70 and Hsp27 were detected. The homogenates from C166 cells incubated with EBC from healthy individuals, when compared to DDW-treated controls, contained slightly reduced amounts of SMAC/Diablo and Hsp70, whereas the amount of Hsp27 was significantly lower, below the detection level of this method (Figures 1 B, C).
The incubation of human lung fibroblasts with exhaled breath condensates from healthy children resulted in morphological changes, similar to those observed in C166 cells. In contrast to the fast effect of EBC on murine endothelial cells, in HLF cultures these changes emerged much more slowly. Thus, although they also concerned all cells in a culture, in the majority of cells even after 4-hour incubation they were less advanced (Figure 2 A).
Figure 2
A – The culture of human lung fibroblasts (HLF) in the phase contrast microscope after 4-hour incubation with double distilled water (DDW), used as a test control (upper picture) or with a representative EBC sample (bottom picture). B – The membranes of Human Apoptosis Array exposed to HLF lysates incubated for 4 h with DDW (left picture) or a representative EBC sample (right picture). Each pair of spots in white frames represents a respective target molecule. The numbers at the frames correspond to respective molecules on graph C: 1 – CD95/Fas, 2 – pro-caspase-3, 3 – cleaved caspase-3, 4 – SMAC/Diablo, 5 – Hsp27, 6 – Hsp70. The brightest spots correspond to reference markers (dashed line frames). C – Relative expressions of selected apoptosis pathway-related proteins with the highest signal density, calculated as the percent of reference spots
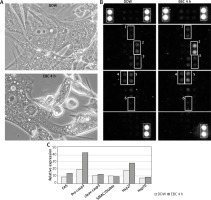
The proteome microarray assessment of DDW-treated control lysates from HLF has shown that, similarly to C166, they contained small quantities of SMAC/Diablo, Hsp27 and Hsp70. However, apart from the aforementioned proteins, fibroblasts also contained small quantities of pro-apoptotic receptor CD95/Fas as well as detectable amounts of pro-caspase-3 and its active form.
The microarray membranes, incubated with lysates from EBC-treated fibroblasts, when compared to those with control lysates, displayed increased amounts of pro-caspase-3 and Hsp27. The remaining signals were similar to those on control membranes (Figures 2 B, C).
Discussion
Our previous study has shown that exhaled breath condensates from healthy children may induce apoptosis in both human and murine endothelial cells [5]. Moreover, we have found that this phenomenon was cell type-unspecific [6]. Although several factors were suggested as possible candidates responsible for this effect, none of them has been confirmed so far. In this study we aimed to analyse presumable molecular changes associated with triggering of apoptotic pathways, using two in vitro models – murine C166 endothelial cell line and normal human lung fibroblasts. Since the apoptosis induced by EBC seems to be a dynamic process, for the proteomic assessment we used 4-hour incubation. Based on data from our pilot studies, that time appeared sufficient to observe typical morphological changes, but short enough to avoid a complete cell damage.
Apoptosis, or programmed cell death, is a multi-step process, which is precisely controlled by numerous positive and negative regulators. Depending on the trigger and different upstream mechanisms, usually two main death pathways, the intrinsic and extrinsic ones, are distinguished. The intrinsic pathway is activated by intracellular signals generated by cellular stress and depends on the release of proteins from the intermembrane space of mitochondria. The activation of the extrinsic pathway results from binding of extracellular ligands to their specific death receptors, followed by formation of the death-inducing signalling complex. Despite a separate initiatory phase, both pathways are linked and molecules of one pathway can influence the other [11]. Both of them induce cell death by activating the cascade of caspases – the family of cysteinyl-aspartate proteases, responsible for degradation of various cellular components in a controlled manner.
Although challenging, careful assessment of molecules involved in regulation of apoptosis, induced by EBC from healthy children, might enable retrograde identification of the trigger molecule. However, due to a large number of target proteins to be analysed and also the limited amounts of EBC samples, such studies are difficult, time-consuming and very expensive. Therefore, we decided to use the most cost-effective tool for fast and broad screening – the protein microarrays designed for apoptosis research. Below we shortly discuss the main findings of our study.
We have found that all tested lysates contained detectable amounts of SMAC/Diablo protein. This small molecule is located in the mitochondrial intermembrane space, but it enters the cytosol, when the cell undergoes apoptosis. SMAC/Diablo promotes apoptosis through the intrinsic pathway by binding the inhibitor of apoptosis proteins (IAP), thus allowing the activation of caspases [12, 13]. However, we suppose that the involvement of this molecule in our system seems to be marginal, especially since in both murine and human cells there were no visible differences in its levels between DDW-treated control cells and cells incubated with EBC samples. Any significant changes in amounts of other intrinsic pathway-associated molecules, like cytochrome C, IAP or Bcl/Bax in our experimental system were not observed.
The next interesting molecule, found in all cell lysates, is a member of the heat shock protein family, Hsp70. This molecule protects cells from environmental stress and is one of the most important anti-apoptotic proteins. Hsp70 prevents apoptosis on several levels [14], e.g. it blocks mitochondrial translocation and activation of Bax, thereby preventing permeabilization of the mitochondrial membrane and release of other pro-apoptotic factors [15]. It has been proven that impaired function of Hsp70 is related to several diseases, including various neurological disorders, cancer, and virus infections [16]. Furthermore, in some observations increased Hsp70 levels in induced sputum and plasma correlated with asthma severity [17]. However, in our experimental model we did not observe any significant differences in Hsp70 amounts between tested lysates.
Another regulatory molecule, detected in all lysates assessed, is Hsp27. It is present in both cytoplasm and nucleus and, similarly to Hsp70, provides significant resistance to heat shock and oxidative stress [18]. Hsp27 inhibits both intrinsic and extrinsic apoptotic pathways by binding cytochrome C and preventing caspase-8-dependent activation of Bid, a pro-apoptotic member of Bcl-2 family [19]. On the other hand, it was suggested that Hsp27 may have both anti- and pro-apoptotic functions that depend on general cell condition [20]. Impaired expression of Hsp27 is associated with several diseases, including myopathy and multiple sclerosis [21]. According to Salinthone et al., overexpression of Hsp27 may inhibit serum-induced proliferation of airway smooth muscle myocytes and, therefore, it may play an important role in asthma-associated remodelling. It has been found that Hsp27 provided protection against H2O2-induced cytotoxicity by the increase in cellular glutathione levels, thus preventing necrotic, but not apoptotic cell death [22]. The aforementioned study may possibly provide some support to explain our data. In lysates of C166 murine endothelial cells, after their 4-hour exposure to EBC, we have observed a significant decrease in Hsp27, which was accompanied by rapid cell death. In contrast to that, lysates from EBC-treated human fibroblasts contained significantly increased amounts of Hsp27, as compared to control cells. Therefore, it may be hypothesized that although increased levels of Hsp27 were not sufficient to fully protect HLF from death, they could be responsible for delayed induction of apoptosis.
Noteworthy, in all HLF and only in some C166 lysates, we have found small quantities of death receptor CD95/Fas. Its activation by a specific ligand (FasL) results in induction of apoptosis through specific adapter molecule-mediated recruitment and activation of a cascade of caspases [23]. However, it seems unlikely that CD95/Fas receptor would play a key role in our experimental system, since the previous screening of EBC samples did not confirm any significant amounts of FasL, necessary for its activation [5].
The last molecule, detected in large amounts in HLF lysates, is pro-caspase-3, the key effector molecule involved in the execution of programmed cell death and activated by both intrinsic and extrinsic pathways. It is produced as an inactive proenzyme which undergoes proteolytic processing (cleavage) to produce two subunits, which further dimerize to form the active enzyme (cleaved caspase-3) [24]. In our experimental system we have observed a significant increase in pro-caspase-3 in EBC-treated cells, however, there was only a little increase in the active enzyme, i.e. cleaved caspase-3. Possibly, since proteolytic processing of pro-caspase-3 requires at least 3–4 h from the exposure to the apoptosis inducer, to observe a significant increase in cleaved caspase-3 it would certainly require longer incubation [25–27]. On the other hand, as found in C166 cells, the effective cell death may occur even at low quantities of cleaved caspase-3. Obviously, it should be considered that metabolism and reaction patterns of murine endothelial cells and human lung fibroblasts may be similar, but not identical. Hence, a similar effect of incubation with EBC from healthy children, i.e. apoptotic cell death, might be achieved in different ways.
The implementation of protein microarray technology to broad screening of classical apoptosis pathways allowed identification of two molecules, Hsp27 and caspase-3, involved in mechanisms of cellular response to exhaled breath condensates. Although our research has not defined so far the detailed pathway of apoptosis activation in the selected experimental system, the number of possible candidates for further studies was significantly reduced. However, the factor responsible for induction of the cytotoxic effect of EBC from healthy children is still unknown. Therefore, to clarify this phenomenon, further studies are required.









