Introduction
Cancers of the external ear constitute about 5.5 % of all skin cancers [1]. The aggressiveness of these malignancies increases as they spread from the conchal bowl into the external auditory canal (EAC). The ratio of basal cell carcinoma (BCC) to squamous cell carcinoma (SCC) within pinna changes in the EAC is 2 : 1 in favour of more aggressive cancer types. SCC is the most common type of carcinoma of the EAC, and according to some authors, the rate amounts up to 70% [2]. BCC constitutes a less frequent type and appears in only 1/4 of malignancies in this location [3]. Similarly, the possibilities of their total extirpation decrease with the development of more aggressive cancers deep into the EAC. A separate issue is reconstruction of the auricular region done for aesthetic reasons, as well as maintenance of the structural firmness of the cavum conchae, which is important to the conduction of the sound waves to EAC [4].
There are only a few surgical techniques used to restore the central auricular part as the surgical techniques for defects of such location require greater surgical experience and are less common [5, 6]. Postauricular island flap (PIF), called also “revolving door’s technique” is intended for the auricular concha, and can be extended further to EAC [7, 8]. Most often PIF is done during a single stage operation [9, 10]. The method was popularized by Masson, and improved by Talmi, Rodendo or McInerney [11–15]. Refinements to the original technique have since been given; Ghassemi described the anterior pedicle retroauricular flap for reconstruction of full-thickness defects in different parts of the ear [16]. Cordova offered three techniques using the retroauricular island flap (RIF): the Superior Pedicle RIF, the Perforator RIF, and the Inferior Pedicle RIF [17]. Also, free flap, despite a short length of the posterior auricular artery, has been used, but with variable success [9].
Novel folded posterior island flap to reconstruct a subtotal full thickness conchal bowl was recommended by Roche, whereas Zhu demonstrated results after the use of a local flap from the preauricular region [4, 18]. Large full-thickness auricular defects require complex reconstruction. Restoration of the cartilage detail is not always necessary for patients undergoing an extensive auricular reconstruction following an ablative procedure for malignancy, notably where the conchal bowl is involved and the superior aspect of the auricle is preserved, especially for older patients with advanced malignancies and a risk of cancer recurrence [18]. Mohs micrographic surgery (MMS) is a common technique to facilitate the removal of tumours in this location [3, 19]. Some authors reported modifications of surgical approaches to improve visualization and access to these lesions [1].
Aim
The aim of our study was to present our clinical experience and postoperative results related to resections in cancer involvement of the auricular concha along with EAC with or without simultaneous radical mastoid surgery (using retroauricular approach) and with PIF reconstruction.
Material and methods
We performed an institution-based cohort study. The study comprised 37 patients (28 males, 9 females), aged 51–86 years, mean age 73 ±7.5 years, afflicted with carcinoma of the conchal bowl and EAC who underwent one stage operations – tumour excision and partial auricular reconstruction (using PIF) between March 2000 and January 2017. The following comorbidities: diabetes mellitus (12), cardiovascular diseases (19), and nicotinism (21) were confirmed in examined individuals. Preoperative biopsy revealed BCC infiltrative type in 12, BCC nodular type in 11, and SCC in 14 cases (G1 – 8, G2 – 6). Malignant tumour size ranged from 2 to 3.5 cm, the defect size after surgical excision was from 2.8 to 4–5 cm. Cancer extension into the opening of the EAC amounted up to 5–7 mm. The excision margin inside EAC was of special concern. All resected specimens were evaluated by histopathology.
Cancer infiltrations into EAC in all our patients were classified according to the modified Pittsburgh staging system [20]. The clinical classification of tumour advancement in the studied patients has been listed in Table 1. In all cases preoperative temporal bone computed tomography (CT) scans were performed. Qualification for radical mastoid surgery was decided on the basis of clinical, pathological, and radiological features of EAC bone infiltration. In case of negative margins in postoperative histopathological specimens, patients were not sent for any postsurgical radiotherapy, leaving this option open for possible recurrences. However, in the studied material there were no cases of local recurrence.
Table 1
Clinical classification of malignancy advancement in the studied group
| Histological type of the external ear cancer | Tumour size | Total N (%) | |
|---|---|---|---|
| T1 | T2 | ||
| BCC nodular | 5 | 6 | 11 (29.73) |
| BCC infiltrative | 5 | 7 | 12 (32.43) |
| SCC G1 | 3 | 5 | 8 (21.62) |
| SCC G2 | 2 | 4 | 6 (16,22) |
| Total | 15 | 22 | 37 (100) |
All patients were operated under general endotracheal anaesthesia. Malignancy severity required removal of full thickness ear concha in all cases. In 5 patients with BCC nodular type, partial EAC resection (removal of skin and lesion with the margin of 3 to 4 mm of healthy tissue) was performed. Due to increased aggressiveness of the infiltrating form of BCC and SCC, in all patients with this diagnosis, the above procedure was extended by radical mastoid surgery with removal of the posterior EAC wall. Margins of the lesions were estimated and excised with microscopic control (microscope Zeiss Vario) under magnification 6–10× during the operation. Closure of the defect was achieved by a PIF of the size adjusted to the defect’s size. The operation scheme is shown in Figure 1.
Figure 1
Scheme of the mastoid radical surgery with postauricular island flap reconstruction due to cancer of the auricular conchal bowl and external auditory canal
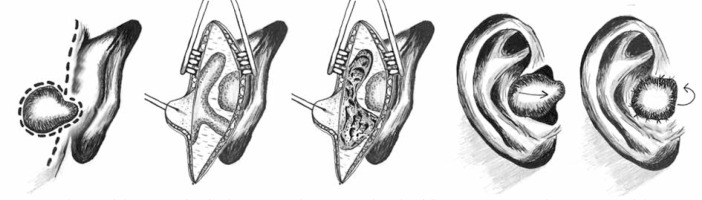
The operation was performed using the retroauricular approach in order to obtain adequate estimation of the lesions and good surgical access to the EAC. Postauricular incision was performed 0.5 cm behind the auricle to remove the cortex layer of mastoid bone, creating a triangular cavity along: bony plate of middle fossa dura, sigmoid sinus, and posterior wall of EAC. After creating a cavity with drilling, the following anatomical landmarks were identified: tegmen of tympanic cavity, sino-dural angle and posterior wall of EAC. The next step was to remove the posterior bony wall of EAC with a diamond blur to the level of the facial nerve canal. Next, the facial nerve was localized with the help of the following anatomical landmarks: cochleiform process, stapedial muscle tendon, oval window niche, lateral semicircular canal, pyramidal eminence, head of malleus, and incudo-mallear joint. Then, the anterior part of epitympanic recess was opened to give access to the head of the malleus, facial nerve recess and tegmen tympani. The following landmarks may assure that epitympanic recess is fully opened: visualization of the tympanic opening of the Eustachian tube and canal of the tensor tympanic muscle. During the radical mastoid surgery, the tympanic membrane and ossicular chain were assessed for neoplastic infiltration. The skin of the posterior wall of EAC was removed completely because of possible neoplastic infiltration. Thus, a wide opening into the postoperative cavity was achieved, which is much easier to control during the follow-up treatment. In order to ensure a wide opening of the postoperative cavity, a Merocel ear dressing was inserted for 7–10 days.
PIF (done in the next stage) was extended from the conchal navicular fossa up to the opening of the postoperative cavity. After removal of concha and EAC cancers, partial reconstruction of the ear using PIF was performed from the anterior conchal approach. Postauricular Island Flap (outlined behind the defect) involved the postauricular skin, the subcutaneous, and the muscle layers. Then, it was incised around the periphery and attached to its base, whereas margins of the flap were undercut and mobilized. With the ear pushed back, the flap was pulled through the defect. In order to reconstruct EAC, the anterior margin of PIF was extended to the posterior surface of the auricle, comprising in this area of skin and subcutaneous tissue only. Additional PIF elongation was inserted into the EAC and sutured to the margin of healthy skin inside the canal. This allowed to cover the wall of the EAC in half with healthy skin, which accelerated healing and reduced the risk of canal atresia. In all cases of mastoid radical surgery, the postoperative cavity was left for secondary healing. In 30 cases, where the flap coverage of the defect was not complete, it was left to heal by secondary intention. Antibiotic (aerosol – Neomycin) was applied to the postoperative wound. The sutures were removed after 7–10 days. All patients were followed up in the Out-Patient Clinic for a month after surgery, then after 3, 6 and 12 months, and once a year in the following years.
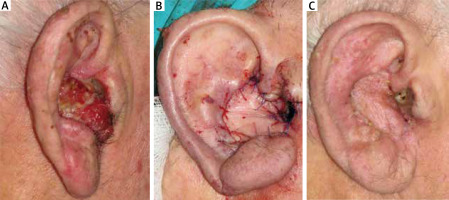
We analysed postoperative results, which were additionally evaluated by a plastic surgeon on the basis of physical examination, as well as by the patients (estimation based on standards adopted in our Department). All patients were subjected to endoscopic assessment of the EAC, as well as the examination of hearing within 3 months after the operation (Figure 2).
Figure 2
A–C – Patient (aged 75) with squamous cell carcinoma of the auricular conchal bowl and external auditory canal, before and after radical mastoid surgery and postauricular island flap reconstruction
The study was approved by the Bioethical Committee of the Medical University of Lodz (RNN/376/19/KE). Written informed consent documents were obtained from each participant.
Results
All patients with infiltrating BCC and SCC underwent mastoidectomy with removal of the posterior wall of the EAC (26 cases). It allowed easy insight into the postoperative cavity and good control for early detection of possible relapse. No infiltration of the tympanic cavity structures was found in any case, thus avoiding the infusion/removal of its contents while maintaining good hearing. Neck lymphadenectomy was abandoned due to the absence of features of lymphadenopathy on clinical and CT examination.
Histopathologic evaluation confirmed the results of preoperative biopsy (Table 1). Histopathologic evaluation of margins showed that excision was complete in all cases; the width of margin was evaluated. The minimal excision margin ranged from 3 to 4 mm (an average: 3.6 ±0.2 mm). No recurrence of cancer at control follow-up in operated patients was confirmed.
Postoperative outcomes in our group have been shown in Table 2. Abnormal wound healing, infection, flap’s shrinkage, depression in the contour of the flap, or non-aesthetic appearance of the donor site, were not demonstrated in the studied group. Postoperative venous congestion resolved within a few consecutive days. Pinning of the operated ear towards the scalp (in large and prominent ears) had the tendency to improve within a few months. Neither doctors, nor patients expressed any need of corrective operations. A proper shape (contour) of the reconstructed ear was obtained in all except for individuals with large prominent ears after surgery, but they did not undergo more corrections. Initially the constricted EAC (from 20% to 40% of its lumen before surgery) in six individuals of eleven (without radical mastoid surgery) was widened. We wanted to avoid recurrent atresia, so delayed full thickness skin grafts (from the contralateral retroauricular region) were used.
Table 2
Results of ear reconstruction in the examined group
Endoscopic assessment of the EAC did not reveal any significant narrowing resulting in impairment of conductive hearing in cases with a preserved posterior part of EAC. Also, in patients after radical mastoid surgery there were no cases of conductive hearing loss as the tympanic cavity content with the ossicular chain was left intact. In five consecutive years after surgery, we did not find any functional abnormalities requiring further EAC operations.
Evaluation by a plastic surgeon and by patients has been shown in Table 3 – based on standards adopted in our Department.
Table 3
Postoperative satisfaction by the plastic surgeon and by the patient
| Scale of evaluation | Postoperative evaluation by | |||||
|---|---|---|---|---|---|---|
| Patient | Plastic surgeon | |||||
| Males (n = 28) | Females (n = 9) | Total (n = 37) n (%) | Males (n = 28) | Females (n = 9) | Total (n = 37) n (%) | |
| Completely satisfied – real contour/shape of the reconstructed auricular concha | 11 | 3 | 14 (37.84) | 10 | 3 | 13 (35.13) |
| Very satisfied – almost real contour/shape of the reconstructed auricular concha | 0 | 0 | 0 | 1 | 0 | 1 |
| Moderately satisfied – almost real contour/shape of the reconstructed auricular concha and pinning of the operated ear or prominent earlobe | 15* | 3* | 18 (48.65) | 15* | 3* | 18 (48.65) |
| 11# | 3# | 14 (37.84) | 11# | 3# | 14 (37.84) | |
| 3∞ | 3∞ | 6 (16.22) | 3∞ | 3∞ | 6 (16.22) | |
| Slightly satisfied – unreal contour/shape of the reconstructed auricular concha, and pinning of the operated ear or prominent earlobe are apparent or EAC constriction | 0 | 0 | 0 | 0 | 0 | 0 |
| Not satisfied – additional surgical revision is needed, unreal conchal shape, pinning of the operated ear, prominent ear lobe, EAC constriction | 0 | 0 | 0 | 0 | 0 | 0 |
Discussion
Cancers of the ear involving EAC can originate from its tissues or result from auricle lesion extension. To date the most widely used EAC carcinoma staging system is the Pittsburgh system (radioclinical) [2, 20]. All patients in our study were evidenced/diagnosed with tumours spreading from the auricular concha into EAC, and lesions were estimated on the basis of the aforementioned classification. As other authors note, the most common type of cancer involving EAC is SCC, and less frequent are: BCC, melanoma, or Merkel cell carcinoma [21]. These tumours must be operated at an early stage as it is possible to avoid mutilating treatment, inevitable in more advanced stages. Complete resection without positive surgical margins is essential for the treatment of patients with T1 and T2 ear cancers. Prophylactic superficial parotidectomy or neck dissection is not mandatory for T1 and T2 stages as long as the extent of disease is precisely assessed preoperatively. Resection surgery, sometimes with adjuvant radiotherapy, is suggested as a standard procedure in cancers of this location. However, similarly to other authors, we claim that postoperative radiotherapy should be performed only for patients with positive surgical margins [22, 23].
Cancers of our group involving ear concha together with EAC were removed with histopathologically free margins and full thickness concha under microscopic control. Recurrences were not seen within next follow-up. We agree with other authors, who indicate primary aggressive treatment of the conchal bowl carcinoma [2, 19, 21]. Only the removal of a whole thickness tumour involving the auricular concha reduces the risk of incomplete cancer excision to the greatest extent. Mohs surgery allows for minimal handling of excised tissues, but some authors report examples of recurrences following cancer removal using this technique, which consequently required further resections [24–26].
Patients from our study underwent excision of the whole thickness of the ear concha, and in cases of aggressive forms (SCC and infiltrative form of BCC) invading deep into EAC also radical mastoid surgery was performed. Access to EAC back wall in order to assess its infiltration from the mastoid process allows for better evaluation of the tumour extent, thus showing the advantage of this method over Mohs technique. Good insight into the surgical field and additional removal of the posterior EAC wall with intact eardrum and tympanic cavity contents allows to achieve surgical margins free of neoplastic infiltration without impairing hearing and maintaining good conditions for further reconstruction with PIF. Cancers in this location spreading deep into the EAC require very precise extirpation after accurate visualization, thus as wide as possible access to the operating field. We recommend aggressive and multidisciplinary treatment including a laryngologist and a plastic surgeon, similarly to other authors [3, 19, 27]. We implemented the retroauricular approach in cases with extension of the tumours from the auricle to the EAC. The results of our study confirmed its effectiveness as the way facilitating access to EAC and estimation of the tumour extent, making it possible to perform a radical operation and remove cancer completely, despite a very limited amount of tissue available in this location. In our group of patients, no recurrences were confirmed in long-term follow-up (at least 5 years). In patients afflicted with carcinoma of the auricular concha together with EAC involvement, it is advisable to combine the above-mentioned method with the use of a retroauricular island flap to restore the cavum concha and even partially EAC. On the other hand, the use of partial auriculectomy to create proper access to the tumour, advocated by other authors, carries a risk of insufficient blood supply (from the inferior pedicle) for the ear to survive [1].
Reconstruction of the external ear includes restoring its orientation, size, and shape to match the contralateral ear as closely as possible and maintain the EAC and the postauricular sulcus. The use of postauricular flaps allow cohesion of the flap skin colour, give less contour defect, minimal donor site morbidity, and inconspicuous scar formation. This method surpasses effectiveness of skin grafts, which are prone to delayed wound healing, pigmentation, centripetal contraction and finally to the deformation of the conchal cavity [15, 28]. Depending on the size of the auricular defect and its location, a retroauricular island flap is also very effective in combination with a preauricular translocation flap for final outcomes of the reconstruction, according to Zhu’s observations. As a single preauricular flap, due to the range, it is more appropriate for the repair of defects in the lower part of the conchal cavity, for defects in the upper part, it is not long sufficient [4].
Another novel technique offered by Roche for large defects is an expanded, postauricular folded flap to restore a full thickness defect of the conchal bowl, with only one donor site. The portion of the ear after resection maintained its structural support without evidence of a lop ear and without restoring the cartilaginous architecture. The technique has advantages in persons with large defects because it is vascularized basing on the contrast and robust posterior auricular artery, provides colour matching the skin, with the restoration of the postauricular sulcus, and has a hidden donor site [18]. The use of cartilage grafts together with flaps improves the contour of the reconstructed ear, but many authors noted that combining both these techniques prolong surgery, increasing donor and reception site morbidity [10].
We did not use cartilage grafts with a proper final conchal shape after reconstruction. We used and still recommend a radical but not burdensome operation, with a relatively low early complication rate in spite of the senile patient’s age and ratio of noted cardiovascular diseases (50%), diabetes mellitus (33.3%), or nicotinism (38.9%). PIF can be used to restore auricular concha in the range even exceeding 6 cm [19]. The defects of our patients were less extensive, and transient congestion but not flap necrosis was seen after surgery, similarly to other reports [10, 11]. Krespi claimed that a consequence of this technique is posterior setting of the pinna, resulting in elimination of the postauricular sulcus [9]. Also, Leitenberger offered a postauricular pull-through subcutaneous pedicle flap with staged cartilage grafting to repair a full thickness lesion with a range of 4.8 × 3.2 cm defect, and also evidenced noticeable ear pinning after pull-through flaps [18]. Similarly to the results of other studies, postoperative auricle pinning showed a tendency to improve within a few consecutive months, and additional surgical revisions were not required in our patients. Postoperative pinning is more visible in persons with large ears, and also is related to the limitation of flap’s mobility, which enhances the asymmetry of both ears [7, 10]. A larger PIF size had an influence on the final prominence of the ear lobe, which other authors did not pay attention to, and we did note several such cases. Stenosis of the EAC, following cancer resections, occurs after extensive cartilage removal. In our individuals with postoperative auditory canal constrictions, we used delayed skin grafting with no functional impairment or need of next surgery, which confirms the observations of others [19].
Postoperative results in our patients are compatible with preserving of the proper shape of the reconstructed ear independently from extensive tumour extirpation, additionally without any evidence of recurrent cancer. Although, it should be stated that this reconstruction method was limited to our patients with T1 and T2 tumours of the conchal bowl with about maximum 1 cm infiltration into EAC.
Conclusions
Retroauricular approach in cancer of the auricular concha and EAC allowed for accurate evaluation of the tumour extent and proper surgical access, which facilitated complete removal of the tumour.
Use of radical mastoid operation with conchal bowl resection and PIF reconstruction in patients with SCC or infiltrating BCC of auricular concha and osseous EAC resulted in cancer extirpation and good aesthetic outcomes, despite minor functional consequences. In these cancers mastoidectomy offered a wider access to determine the radicality of oncological resection.









