Drug hypersensitivity reactions are dose-independent, unpredictable, and immune-mediated type B adverse drug reactions (ADR) in patients sensitized to a certain drug [1]. The first signs appear within minutes to hours of drug intake, ranging from low-grade to possibly life-threatening anaphylactic symptoms; they can also appear several hours to days later in the form of exanthema [2]. An allergic reaction should be substantiated within 4 weeks to 6 months after the reaction, aiming to identify the trigger and assess the risk for subsequent reactions [3].
Clinical classification, based on the morphology and timing of the reaction, is necessary for correct diagnosis. In the case of distinct symptoms of hypersensitivity and unambiguous findings of validated skin (in vivo) and/or laboratory tests (in vitro), the assignment to a trigger may be considered sufficient [4]. However, skin and laboratory tests lack high sensitivity and frequently show false negative or non-significant results [5].
The European Academy of Allergology and Clinical Immunology defines a drug provocation test (DPT) as a controlled administration of a drug in order to exclude or establish the diagnosis of a drug hypersensitivity reaction. If clinical symptoms are observed during the DPT, hypersensitivity to a certain drug can be identified with a high sensitivity [6]. However, the DPT as a gold-standard-procedure is debatable due to the risk of hypersensitivity reactions [7]. For this reason, the diagnostic value of the DPT has to be evaluated individually in each patient by assessing the possible risks and benefits. If substances of high clinical value (e.g. antibiotics, analgesics) are presumed to induce hypersensitivity reactions, the beneficial aspects of the DPT are considered to outweigh the potential risks. DPT can reduce the risk of misdiagnosis, which may lead to severe reactions on repeated exposure or unfounded treatment restrictions [8].
DPT is safe for the assessment of hypersensitivity reactions if emergency backup facilities are guaranteed and experienced clinicians are present to treat potential anaphylactic reactions during the testing procedure [9].
Because immediate treatment of life-threatening anaphylactic reactions is crucial, we retrospectively investigated a DPT series to evaluate the risk-benefit ratio of DPT in an inpatient setting.
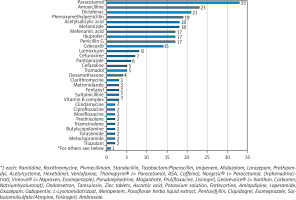
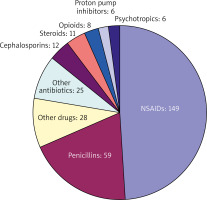
Standard procedure in the diagnosis of drug hypersensitivity reactions are as follows: I) evaluation of the patient history, II) in vivo and/or in vitro testing, and – in case a diagnosis cannot be established – III) drug provocation testing (DPT) [10]. At our department, 101 patients were hospitalized and exposed to drugs over a period of 6 months. During the DPT 69 different substances were tested with a total of 304 administrations allocated to all 101 patients, leading to an average of 3.01 substances administered per person (Figure 1). The drugs investigated were non-steroidal anti-inflammatory drugs (NSAIDs) 149 (49%), antibiotics 96 (31.6 %) – of which 59 were penicillins (19.4%), steroids 11 (3.3%), proton pump inhibitors and psychotropics each 6 (2%), and other drugs 28 (9.2%) (Figure 2). DPT was stopped as soon as symptoms of an anaphylactic reaction were reported. Drug provocation protocols followed a standardized procedure of administering selected drugs intravenously and/or orally, with increasing concentrations of the substance tested. Seventeen out of the 101 patients undergoing a DPT showed symptoms of a hypersensitivity reaction (itching/exanthema, conjunctivitis, rhinitis, globus sensation, asthma, angioedema, or dyspnoea). The incidence of hypersensitivity reactions during our observation period was lower than reported in a study conducted by a Danish allergy clinic, which included 1659 patients and 1913 testing procedures, in which 211 (11%) patients with positive DPTs were reported [11]. However, this may be due to the large number of positive DPTs following administration of antibiotics – 198 (93.8%) – which constituted the majority of substances tested (92.8%). In total 69 different substances were administered orally or intravenously at our department, most of them being NSAIDs and β-lactam antibiotics. Most hypersensitivity reactions occurred after exposure to NSAIDs, 9 (52.9%), manifesting as type I allergic reactions, β-lactam antibiotics, mainly penicillins – 5 (29.4%), macrolide antibiotics – 2 (11.8%), and H2-blockers – 1 (5.9%). All hypersensitivity reactions were treated according to the guidelines for acute management of anaphylactic reactions [12]. No cases of anaphylactic reactions grade III or higher were observed in this study, which was probably due to the immediate treatment of type 1 hypersensitivity reactions.
Figure 2
Each administration of a test substance was allocated to a substance class. The predominant substances are non-steroidal anti-inflammatory drugs and antibiotics
We observed that more than half of the hypersensitivity reactions occurred after exposure to NSAIDs. Nine out of 149 administrations (6%) of NSAIDs induced a hypersensitivity reaction. Most adverse reactions in patients tested for NSAID hypersensitivity occurred after administration of the non-selective cyclooxygenase (COX)-inhibitor lornoxicam. Four out of 8 patients exposed to lornoxicam showed symptoms of a type 1 hypersensitivity reaction. All patients showing symptoms of drug hypersensitivity reactions to lornoxicam had a presumed COX-1-inhibitor intolerance. Therefore, exposure to lornoxicam has to be critically reviewed for these patients. A study investigating oral provocation of COX-2 inhibitor therapy in NSAID-sensitive patients emphasises monitored testing procedures because about 75% of the patients showed cross-reactivity [13]. This is reflected in current recommendations for diagnostic work up and patient management in suspected NSAID hypersensitivity, which state that cross reactivity in COX-inhibitors is usually present, rendering standardized and monitored conditions necessary [14].
Skin tests for β-lactam antibiotics are relatively reliable, although false negative results are observed [5]. In our retrospective analysis, β-lactam antibiotics were the second most common cause of hypersensitivity reactions. 8.5% of patients undergoing DPT for β-lactam antibiotics developed symptoms of a type 1 hypersensitivity reaction. Although DPT bears a high risk of developing an anaphylactic reaction in this patient group, it is considered the best diagnostic tool when investigating a possible hypersensitivity against β-lactam antibiotics [15].
Diagnostic uncertainty in the work up of drug hypersensitivity reactions necessitates provocation testing. Our results indicate that there is a 5.6% chance of developing a hypersensitivity reaction among patients undergoing a DPT, which is lower than previously published. Most hypersensitivity reactions occur during administration of NSAIDs and antibiotics. In accordance with the literature, cross reactivity with COX-2 inhibitors in NSAID-sensitive patients is likely to occur, rendering proper medical attention mandatory in these patients. The DPT can only be regarded safe if performed under thoroughly monitored circumstances by experienced clinicians in an inpatient setting offering emergency backup facilities.