INTRODUCTION
In recent years, new biologics have proven to be highly effective in the treatment of immune-mediated chronic inflammatory skin diseases and have thus brought significant progress in many clinical entities [1, 2]. This also applies to the management of psoriasis vulgaris, which can now be treated with highly selective antibodies, including those targeting interleukin 23 (IL-23). One such antibody, tildrakizumab, has been approved in 2018 [2]. By targeting specific pathways of inflammatory processes, most biologics have a more favourable side-effect profile than conventional immunosuppressants such as glucocorticoids or methotrexate. A possible side effect of all immunosuppressive or immunomodulating agents might be an increased risk of infection [1, 3]. In general, the potential risk of infection can be further reduced by making use of available vaccinations. During the COVID-19 pandemic, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the need for effective vaccines in patients undergoing immunosuppressive or immunomodulatory therapies for chronic inflammatory diseases has been raised increasingly by both physicians and patients. In this phase of the pandemic, strategies for booster vaccinations have been widely debated within this subgroup. As anti–IL-23 antibodies are used only in a few diseases, such as psoriasis, knowledge regarding the development of a sufficient protective antibody response to immunisation during therapy with these biologics remains limited. Consequently, more data are needed to determine whether vaccines against SARS-CoV-2 can be safely and effectively administered during this type of therapy [1, 3, 4].
As these biologics have a long biological half-life and are administered at intervals of 8 to 12 weeks, questions arise as to whether the time interval between therapeutic injections and SARS-CoV-2 vaccination is particularly critical for booster vaccinations. Currently, some physicians consider waiting at least one to two weeks after SARS-CoV-2 vaccination before resuming immunomodulatory therapy. Even a complete discontinuation of medication between two vaccinations has been discussed [3–5].
Because immunocompromised individuals were excluded from phase III pivotal trials for all vaccines approved by the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA), there are currently only a few studies involving this cohort, particularly in the context of psoriatic disease and the treatment with anti-IL23 antibodies [1, 4, 6]. Recent studies indicate that various immunosuppressive or immunomodulatory therapies, such as B-cell-depleting therapy, Janus kinase (JAK) inhibitors or tumour necrosis factor (TNF) blockade, can diminish the vaccination response [6–9]. However, other studies also suggest a favourable immune response in patients treated with targeted biologics compared with those receiving broader immunosuppressive therapies [10–16].
Studies by Bugatti et al. and Geisen et al. showed that after a single dose of the BNT162b2 mRNA COVID-19 vaccine (Comirnaty® by BioNTech and Pfizer), fewer non-responders were observed among patients treated with IL-17 or IL-23 inhibitors than among those treated with other immunosuppressive therapies. Discontinuation of the drug had no detectable effect on antibody formation in these cohorts [6, 9, 10]. Good seroconversion rates were also reported for small cohorts of other IL-23 inhibitors in a study by Mahil et al. and for IL-17 inhibitors in a study by Furer et al. after two doses of BNT162b2 [12, 13].
In summary, however, the diverse study results, together with the fact that mRNA vaccines represent a novel vaccine technology, contribute to increased uncertainty among patients [1, 6]. Nevertheless, the benefits of vaccination outweigh the risks, and vaccination is strongly recommended for psoriasis patients receiving immunosuppressive therapy [1, 3–5, 16].
Our study aimed to evaluate the response to SARS-CoV-2 vaccination in patients with psoriasis receiving IL-23 inhibitor therapy with tildrakizumab, considering the type of vaccine, vaccination dates, the time interval since the last biologic dose and patient age. The results of our study will help reduce patient uncertainty and facilitate discussions regarding frequency, for example yearly, booster vaccinations for psoriasis patients during tildrakizumab therapy. We therefore provide data demonstrating the immunogenicity of SARS-CoV-2 vaccines in patients on ongoing tildrakizumab therapy in a monocentric prospective observational study.
MATERIAL AND METHODS
Patient recruitment and study design
All patients were recruited between 30 August 2021 and 22 December 2021 at the Centre for Inflammatory Skin Diseases, University Medical Centre Schleswig-Holstein, Campus Kiel. Inclusion criteria for this monocentric prospective cross-sectional study were: age over 18 years, treatment of diagnosed moderate-to-severe psoriasis with tildrakizumab in accordance with the marketing authorisation, and completion of an anti–SARS-CoV-2 vaccination schedule according to the regulations in place at the time. A complete vaccination schedule was defined as two doses of an mRNA or vector vaccine from AstraZeneca, one dose of an mRNA vaccine combined with one dose of a vector vaccine from AstraZeneca, or one dose of the Johnson & Johnson vaccine.
Exclusion criteria included previous SARS-CoV-2 infection and receipt of a third booster vaccination. Eligible patients were asked by their responsible physicians to provide a blood serum sample and information at a single time point, which occurred between 13 and 286 days after vaccination. All subjects signed a privacy statement and provided written informed consent. The study was approved by the ethics committee of Kiel University (D409/21).
Patients completed a one-page questionnaire regarding their vaccination status, including the dates of all prior SARS-CoV-2 vaccinations and the vaccine product used. In addition, a file with relevant pseudonymised patient data was created. Data not obtained from the questionnaire were retrieved from the electronic patient chart when necessary.
Vaccinations were not part of the study and were performed in official vaccination centres according to the up-to-date recommendations of the Ständige Impfkommission (STIKO, the German standing committee on vaccination) of the Robert Koch Institute valid at that time.
Sampling and storage of serum samples
Serum samples were collected by study personnel. After collection, the samples were allowed to stand at room temperature for 30 to 60 minutes and were then centrifuged at 2000 × g for 10 minutes. Two to three aliquots per patient were prepared and stored at –20°C.
Detection of antibody and protein levels
Enzyme-linked immunosorbent assays (ELISAs) were performed to detect IgG antibodies (EUROIMMUN QuantiVac®) and IgA antibodies (Aeskulisa Aeskulab®) against the S1 protein, inhibition levels of neutralising antibodies (Invitrogen, Thermo Fisher Scientific®), and IgG antibodies against the nucleocapsid protein (to identify possible undetected SARS-CoV-2 infections). All measurements were performed according to the manufacturers’ protocols.
Statistical analysis
Differences between multiple groups were assessed using the Kruskal-Wallis test, pairwise comparisons by the Mann-Whitney test. Statistical analyses were performed using SPSS® and GraphPad Prism®. To analyse the impact of time elapsed since the full vaccination on the SARS-CoV-2-specific antibody levels, the 63 subjects were divided into three groups according to the days elapsed between the last vaccination and the sampling (0–90 days after the basic vaccination, 91–150 days and 151–286 days). The grouping was based on the time intervals between basic immunisation and booster vaccination currently recommended by STIKO.
For further statistical analyses, SAS® was used as the statistical software. IgG, IgA and neutralising antibodies were analysed in separate statistical models, the responses were log-transformed prior to analysis. A linear mixed model was used to consider the effect of various factors on the response. The variables included: time from full vaccination to sampling for titre assessment; time from the last dose of tildrakizumab prior to full vaccination to the date of full vaccination; time from the last tildrakizumab dose prior to any vaccine administration to the first vaccine dose; time from the last tildrakizumab dose prior to sampling for titre assessment; age; vaccination scheme; and body mass index (BMI) category. Vaccination scheme and BMI category were included as categorical variables, and the Kenward–Roger method was used to calculate the degrees of freedom. P-values < 0.05 were considered statistically significant.
RESULTS
Demographics
A total of 63 subjects receiving ongoing therapy with tildrakizumab were included in our non-randomised study. All patients had completed a SARS-CoV-2 vaccination schedule consisting of either one dose (COVID-19 Vaccine Janssen®) or two doses (Comirnaty® by Bio NTech/Pfizer, Spikevax® by Moderna, Vaxzevria® by AstraZeneca). The distribution of vaccination schedules was as follows: 2 subjects received two doses of Spikevax®, 3 received two doses of Vaxzevria®, 4 received a single dose of COVID-19 Vaccine Janssen®, 8 received a mixed schedule consisting of one dose of Vaxzevria® followed by one dose of Comirnaty®, and 1 subject received one dose of Vaxzevria® followed by one dose of Spikevax®. All remaining patients received two doses of Comirnaty®. The interval between the first and second doses ranged from 13 to 90 days, depending on the recommendations issued by STIKO.
Of the included patients on tildrakizumab therapy, 71.4% were male, and the mean age was 52.3 years (±16.3; range 22–91). All patients were receiving maintenance therapy with tildrakizumab for moderate-to-severe plaque psoriasis at the time of their first SARS-CoV-2 vaccination, which occurred between 15 January 2021 and 19 October 2021.
Psoriasis severity at the time of sampling during ongoing tildrakizumab therapy was assessed using the Psoriasis Area and Severity Index (PASI) and body surface area (BSA). The mean PASI score of all patients was 1.89 (±1.9; range 0.0–7.8), and the mean BSA score was 1.52% (±1.7; range 0.0–4.5%).
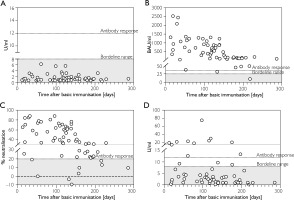
None of the patients had experienced a SARS-CoV-2 infection prior to full baseline immunisation between 17 February 2021 and 12 November 2021. Prior infection was assessed by detecting IgG antibodies against the nucleocapsid protein using ELISA. No patient had values above the manufacturer’s stated detection borderline range of 8-12 U/ml. The mean value was 1.5092 U/ml (±1.2; range 0.3220-6.2325), with a median of 1.0927 U/ml (Figure 1 A). Table 1 provides more detailed baseline patients' characteristics.
Table 1
Demographics and clinical characteristics of the included patients
Figure 1
Distribution of SARS-CoV-2 specific antibody levels detected in patients plotted against the time between the last SARS-CoV-2 vaccination and sample collection in days. A – Serum anti-SARS-CoV-2 nucleocapsid antibodies; B – Serum anti-SARS-CoV-2 IgG antibodies; C – Inhibitory activity of anti-SARS-CoV-2 neutralising antibodies, D – Serum anti-SARS-CoV-2 IgA antibodies. Each symbol represents a single study participant
SARS-CoV-2 vaccines show immunogenicity in psoriasis patients with ongoing tildrakizumab therapy
For the IgG ELISA against the S1 protein of SARS-CoV-2, the cut-off value for detecting sufficient antibody development is between 25.6 and 35.2 BAU/ml according to the manufacturer’s instructions. In our cohort, 61 (96.8%) subjects showed values above 35.2 BAU/ml (and thus immunogenicity after completion of basic immunisation), 1 (1.6%) subject showed an IgG concentration in the borderline range, and only 1 (1.6%) subject did not show sufficient antibody development (Figure 1 B). Subjects showed a mean anti-SARS-CoV-2 IgG antibody level of 594.0550 BAU/ml (±575.3740; 10.2302–2520.7966) with a median of 453.2389 BAU/ml. For neutralising antibodies against SARS-CoV-2, 54 (85.7%) subjects also achieved values above the ELISA cut-off of 20% inhibitory activity, while 9 (14.3%) had values below this range (Figure 1 C). On average, inhibitory activity was 44.1931% (±24.8609; –9.3465–88.3781) with a median of 38.3526%. In addition, we observed a strong two-way correlation, significant at the 0.01 level, between the IgG antibody levels achieved and the concurrent inhibitory activity of neutralising antibodies against SARS-CoV-2 (p < 0.001; correlation coefficient 0.944). SARS-CoV-2 IgA antibodies were detected above the cut-off level of 8–12 U/ml in 11 (17.5%) of the subjects and 3 (4.8%) showed results in the cut-off range. In 49 (77.8%) subjects in our cohort, levels were below 8 U/ml (Figure 1 D). On average, subjects showed a mean IgA antibody level of 6.6940 U/ml (±11.3634; 0.2358–75.3776) with a median of 2.3986 U/ml.
The time-period since completion of basic immunisation has a significant impact on antibody concentration
Our analyses showed a reduction in IgG antibody concentrations, as well as a clear decrease in the inhibitory activity of neutralising antibodies and in IgA antibody levels, with increasing time intervals from the last SARS-CoV-2 vaccination. When comparing SARS-CoV-2-specific antibody levels between the three defined groups, there was a significant difference in IgG antibody concentrations between the 0–90 days group and the 151–286 days group (p < 0.0001), and between the 91–150 days group and the 151–286 days group (p = 0.0002). No significant difference was observed between the 0–90 days and 91–150 days groups (p = 0.4829) (Figure 2 A).
Figure 2
Distribution of SARS-CoV-2 specific antibody concentrations in the groups 0–90, 91–150 and 151–286 days after the last SARS-CoV-2 vaccination in serum. A – Anti-SARS-CoV-2 IgG antibody concentration in serum; B – Inhibitory activity of anti-SARS--CoV-2 neutralising antibodies in serum; C – Anti-SARS-CoV-2 IgA antibody concentration in serum. Each symbol represents a single study participant. The bars represent the median values. Significance values were determined using Kruskal-Wallis and Mann-Whitney test. Significance levels values are indicated as follows: *p < 0.05, ***p < 0.001, ****p < 0.0001
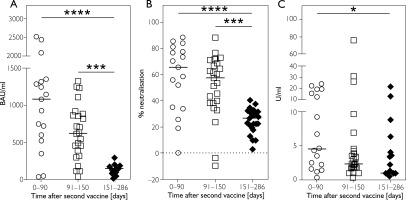
The same pattern was observed for the inhibitory activity of neutralising antibodies. A significant reduction was found between the 0–90 days (p < 0.0001) and 91–150 days (p = 0.0002) groups compared with the 151–286 days group, while no significant difference was observed between the first two groups (p > 0.999) (Figure 2 B).
IgA antibody levels also showed a marked decrease between the 0–90 days group and the 151–286 days group (p = 0.0316), but no significant differences were observed between the 151–286 days and 91–150 days groups (p = 0.9296), nor between the 0–90 days and 91–150 days groups (p = 0.2686) (Figure 2 C).
Detailed demographic and clinical characteristics of the patients in each group are provided in Table 2.
Table 2
Demographic and clinical characteristics in the individual groups
The time interval of tildrakizumab dosing to SARS-CoV-2 vaccination has no influence on immunogenicity
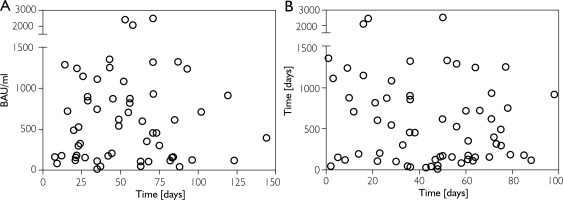
A linear mixed model in SAS was used to determine whether tildrakizumab dosing plays a role in the formation of IgG, IgA or neutralising antibodies in response to SARS-CoV-2 vaccination. None of the tildrakizumab-related variables: time from the last dose of tildrakizumab to full vaccination; time from the last tildrakizumab dose prior to the first vaccine dose; or time from the last tildrakizumab dose prior to sampling for titre assessment, showed a significant association with antibody response (p > 0.05 for each variable for IgG, IgA and neutralising antibodies). Scatter plots of serum anti–SARS-CoV-2 IgG antibody concentrations (BAU/ml) plotted against the time between the last tildrakizumab dose prior to full vaccination and the date of full vaccination are shown in Figures 3 A and 3 B. Thus, our data set does not indicate that the time interval between tildrakizumab administration and SARS-CoV-2 vaccination influences vaccine immunogenicity.
Figure 3
A – Scatter plot of serum anti-SARS-CoV-2 IgG antibody concentration in BAU/ml plotted against the time between the last tildrakizumab dosing prior to full vaccination and date of full vaccination in days. B – Scatter plot of serum anti-SARS-CoV-2 IgG antibody concentration in BAU/ml plotted against the time between the last tildrakizumab dosing prior to first vaccination and the date of the first vaccination dose in days
The type of vaccination has a significant impact on antibody concentration
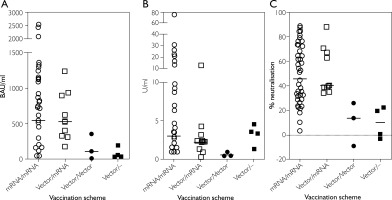
Our statistical model suggested that the vaccine type impacts the immune response with higher antibody-titres observed in patients who had received a vaccination regimen involving at least one dose of an mRNA vaccine. This was particularly significant for IgG antibody serum concentrations (p < 0.0001) and neutralising antibodies (p < 0.0001) but not for IgA antibody concentrations (p = 0.1548). Visual inspection of the scatter plots, however, also suggest a numeric trend for IgA antibody titres (Figures 4 A–C).
Figure 4
Scatter plot of SARS-CoV-2 specific antibody levels in serum plotted against the different vaccination regimens. A – Anti-SARSCoV-2 IgG antibody concentration in BAU/ml; B – Anti-SARS-CoV-2 IgA antibody concentration in U/ml; C – Inhibitory activity of anti-SARS-CoV-2 neutralising antibodies in %-inhibition. Each symbol represents a single study participant
DISCUSSION
We showed that patients on ongoing therapy with tildrakizumab developed anti-SARS-CoV-2 specific antibodies and that the timing of tildrakizumab administration before SARS-CoV-2 vaccination did not affect immunogenicity.
The only subject who did not achieve an anti-SARS-CoV-2 IgG antibody concentration above the manufacturer’s stated threshold was a 54-year-old male patient who had received two doses of Vaxzevria®, was not taking any additional immunosuppressive medications, had no other autoimmune diseases; the collection of his sample was performed 218 days after the last SARS-CoV-2 vaccination. One subject whose anti-SARS-CoV-2 IgG antibody concentration was in the borderline range was a 61-year-old female patient who had received a single dose of the COVID-19 Vaccine Janssen®, was also not taking any additional immunosuppressive drugs, had no other autoimmune diseases; her sample was collected 133 days after the last SARS-CoV-2 vaccination. In the case of both subjects, there was an interval of more than 14 days between the administration of tildrakizumab and the SARS-CoV-2 vaccination. Both subjects also had low inhibitory values, which is consistent with our analyses of the correlation between anti-SARS-CoV-2 IgG antibody concentrations and the inhibitory activity of neutralising anti-SARS-CoV-2 antibodies, which has also been demonstrated in other studies [17–20].
The seven remaining subjects with low inhibitory activity of neutralising anti-SARS-CoV-2 antibodies also showed low IgG concentrations (mean: 84.6739 BAU/ml; 37.1770–156.5400 BAU/ml, median: 50.7514828 BAU/ml). The time interval between the last SARS-CoV-2 vaccination and sample collection was 37, 52, 140, 158, 160, 216, and 286 days. The two subjects who had inadequate inhibitory concentrations 37 and 52 days after the last SARS-CoV-2 vaccination had both been vaccinated once with COVID-19 Vaccine Janssen®, were 49 years old and male, and both had no additional immunosuppressive therapy or other known autoimmune diseases. The subject who was below the manufacturer’s cut-off at 140 days had received two doses of Vaxzevria®, all others received two doses of ComirNaty®. None of these subjects had any other known autoimmune disease or immunosuppressive therapy besides tildrakizumab.
These observations are consistent with our findings that both anti-SARS-CoV-2 IgG antibody concentration and inhibitory activity of neutralising anti-SARS-CoV-2 antibodies decrease significantly with increasing time since the last anti-SARS-CoV-2 vaccination. This was also observed in other studies in healthy and other immunocompromised patients three, four and six months after the second anti-SARS-CoV-2 vaccination [9, 20–23]. Since the number of vaccine-induced, short-lived T and B cells physiologically decreases over time and only some of these cells differentiate into specialised, long-lived immune memory cells, a decrease in antibody levels with increasing time since vaccination is expected [24, 25]. Therefore, early consideration should be given to a third vaccination dose against SARS-CoV-2, which has shown a significant rebound in anti-SARS-CoV-2 antibody levels in other studies [20, 23, 26–28] as well as the need for repetitive booster vaccinations.
When looking at the mean and median anti-SARS-CoV-2 IgG and neutralising antibodies in other studies of immunocompromised patients, which included healthy controls for comparison, immunocompromised patients showed lower mean and median values [5, 9, 20, 22, 29, 30]. This suggests that our patients also have lower mean and median values compared to healthy subjects. However, samples in our study were not collected at a uniform time point and thus often had a longer time period between the completion of the vaccination scheme and the sample collection. In addition, the measured humoral immune responses depend on the specific assays used including their inherent variability, which limits comparability. A reliable statement about the relationship between the values measured in our patients and those of healthy subjects would therefore only have been possible with a separate control group of healthy subjects. Given the high variability of our subjects, collecting a matched cohort of healthy controls would have been difficult, which is why we chose an observational study design. Nevertheless, we were able to demonstrate a humoral immune response to the vaccine, representing a broad spectrum of psoriasis patients undergoing tildrakizumab therapy and examining the humoral response in both the rising and falling phases over a long period of time.
Messenger-RNA vaccines are a promising alternative to conventional vaccines that will certainly accompany us increasingly in the future as an important new vaccine technology not only in the prevention of infectious diseases, but also as a source of hope in cancer treatment [31]. Concerns that the SARS-CoV-2 mRNA vaccines would not result in an antibody response were refuted, as shown by other studies [5, 10–12, 32]. On the contrary, there is evidence from our data that IgG and neutralising antibody formation is lower with dual vaccination with Vaxzevria® and single vaccination with COVID-19 Vaccine Janssen® compared to mixed vaccination and dual vaccination with an mRNA product. These results are consistent with those of other studies in healthy individuals comparing both homologous and heterologous vaccination regimens with Vaxzevria®, ComirNaty® and Spikevax® in terms of IgG antibody levels as well as neutralising titres [17, 33, 34]. Similar findings were reported for IgG levels in a study by Lim et al. in liver transplant patients [35]. Significantly lower IgG levels were also found in COVID-19 Vaccine Janssen® vaccinated cancer and maintenance dialysis patients compared to mRNA vaccinated patients with the same disease [21, 36]. However, given the small cohort of vector-based vaccinations and the heterogeneity in sampling times, these results should be viewed with caution.
When investigating the impact of tildrakizumab dosing prior to vaccination, our statistical model did not identify any of the variables related to tildrakizumab dosing as significant effect modifiers. A study by Bugatti et al. confirms that the timing of immunosuppressive therapy does not affect anti-SARS-CoV-2 antibody formation [10]. Nevertheless, these analyses depend on sampling time, and available data remain scarce and require further study.
We acknowledge that the cohort is small and that our results may be attributable to patient selection and limited demographic diversity. In addition, blood collection was not performed at a uniform time interval, meaning that the specific anti-SARS-CoV-2 antibody levels reflect a cross-section. Further research is needed regarding responses to new SARS-CoV-2 variants and to third or fourth booster vaccinations, as the ELISA methods we used were developed based on the α-variant, and our results refer only to antibody levels after basic immunisation [6, 10]. Questions also remain about the extent to which anti-SARS-CoV-2 IgG and neutralising antibody levels correlate with protection against infection or severe disease [26, 37, 38]. Additional studies are needed to investigate how SARS-CoV-2 infection prior or subsequent to vaccination affects immunogenicity, as such patients were excluded from our cohort [10, 30]. Furthermore, both humoral and cellular responses appear critical for vaccine-induced protection [3, 39]. Continued monitoring and adaptation of assays and vaccines to emerging variants will remain essential for the future management of psoriasis patients receiving tildrakizumab with regard to COVID-19 [3, 30, 32].







