Introduction
Asthma is a disease associated with chronic inflammation of the airways, which can manifest in patients as wheezing, shortness of breath, coughing, or chest tightness [1]. In the vast majority of cases, asthma has a multifactorial and multigene basis, which means that we can observe different phenotypes of asthma. The most serious phenotype is severe asthma, while status asthmaticus is not a persistent phenotype – it is more of an exacerbation.
Asthma has a high prevalence rate, with an estimated 300 million people worldwide suffering from it. It is also an important factor in the development of chronic obstructive pulmonary disease [1]. According to the Global Burden of Disease study, women are more severely affected by asthma than men, particularly in adults; in addition, a faster increase in incidence is observed for women. Currently, it is estimated that about 2 million people in Poland suffer from asthma, which is about 4.9% of the population [1]. It is worth noting that the number of people with asthma in Poland remains unrecognized, as confirmed by a study conducted as part of the Epidemic of Allergic Diseases in Poland program, which revealed that among patients qualified for the program, only 30% of patients had previously diagnosed asthma [1].
Given the large number of asthma phenotypes, it is difficult to unequivocally identify direct causes of asthma. On the other hand, we can include the presence of allergic rhinitis as one of the most important factors in its development, especially in those with known bronchial hyperresponsiveness [1]. Other risk factors for asthma can also include, among others, low birth weight, parental smoking, or the use of antibiotics and paracetamol. It is also worth noting that people living in cities are more susceptible to developing asthma. Genetic factors are also important in the development of asthma, meaning that people with a family history of asthma are more likely to develop asthma [1]. In addition, asthma risk factors can vary between regions of the world, meaning that in highly developed countries, people who live in rural areas are less likely to develop asthma, compared to their peers who live in urban areas [1].
Treatment of asthma involves the use of inhaled steroids by patients to reduce inflammation in the respiratory system. In addition to these drugs, β2 mimetics are also used by patients to dilate the bronchi, thereby reducing the severity of symptoms [1]. These drugs provide good control of asthma symptoms in most patients, while in the treatment of severe IgE-mediated allergic asthma, in which the patients are treated with high doses of inhaled corticosteroids, which is associated with type 2 inflammation and elevated levels of interleukins (IL) 4 (IL-4), IL-5, and IL-13 is insufficient. In this case, treatment with monoclonal antibodies, characterised by targeting eosinophil-associated inflammatory pathways, is much better in the context of severe eosinophilic asthma [2]. Most of the new therapies used to treat severe asthma target eosinophil-associated inflammatory pathways, particularly interleukin-5, a major mediator of eosinophilic inflammation. IL-5, binding to the specific IL-5R receptor, regulates eosinophil proliferation, migration, maturation, and survival. Upon its activation, eosinophils degranulate, releasing cytotoxins that have antimicrobial effects, but they can also damage surrounding tissues and cells [2]. Therapy targeting IL-5 or IL-5R with monoclonal antibodies plays a crucial role in the pathogenesis of severe asthma by reducing the number of eosinophils, providing an effective alternative treatment for patients with severe asthma [3].
Pathophysiology of severe eosinophilic asthma and patient selection
Severe bronchial asthma is a disease characterised by chronic airway inflammation, bronchial hyperresponsiveness, and progressive bronchial wall remodeling, which makes airflow and symptom control difficult [3, 4]. Despite the use of high doses of inhaled corticosteroids and bronchodilators, patients with severe asthma experience frequent exacerbations and significant deterioration in quality of life. A key role in the pathogenesis of this form of the disease is played by type 2 inflammation associated with excessive activation of Th2 lymphocytes and elevated levels of cytokines such as IL-4, IL-5, and IL-13 [3–5]. It is worth mentioning that IL-13 directly modulates structural cells, including fibroblasts and epithelial cells, which contributes to mucus hypersecretion and extracellular matrix degradation. IL-4 enhances inflammatory cascades through IgE shifting, which promotes Th2 cells to prolong cytokine release, inducing chemokines that round up more eosinophils [6].
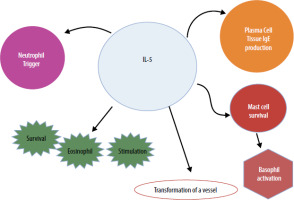
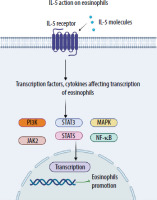
IL-5 is one of the main factors responsible for eosinophilic airway inflammation, which often occurs in severe asthma. IL-5 stimulates the differentiation, maturation, and survival of eosinophils in the bone marrow and promotes their migration into the bronchial mucosa [4]. An excess of eosinophils in the airways leads to epithelial damage, increased mucus production, and increased bronchial activity, which contributes to difficulty breathing and frequent exacerbations of the disease. In addition, IL-5 increases the resistance of eosinophils to apoptosis, which hinders their elimination and prolongs airway inflammation [4, 5]. IL-5 is produced by several cell types, including Th2 lymphocytes, group 2 innate lymphoid cells (ILC2), mast cells, and eosinophils. These cells release IL-5 in response to activation triggered by various environmental stimuli such as inhaled allergens, respiratory viruses, and air pollutants. IL-5 exerts pleiotropic effects on eosinophils, promoting their maturation, activation, survival, migration from the bloodstream, and recruitment to the airways. Additionally, IL-5 influences basophil activation and B cell antibody production, which results in a broader imbalance of the immune system in the progression of severe asthma [7]. Figure 1 shows the pleiotropic effect of IL-5. Figure 2 shows the molecular mechanism of IL-5 action on eosinophils.
IL-5 binds to the α subunit of the IL-5 receptor (IL-5Rα), inducing its interaction with βc subunits, which leads to the activation of a complex network of intracellular signalling, including JAK1/2-STAT1/3/5 modules, MAP kinases, p38 and ERK, and the transcription factor NF-κB. The subsequent stimulation of specific target genes leads to maturation, survival, and activation of eosinophils [7]. Selection of patients that are eligible for an anti-IL-5/IL-5R therapy is based on several factors, such as type 2 high asthma with frequent exacerbations, despite maximal doses of drugs used in inhaled therapies. In addition, key biomarkers include blood eosinophil count, which should be above the upper limit of normal for the population and for those who take high doses of ICS, BEC count ≥ 150/μl. Elevated fractional exhaled nitric oxide (FeNO ≥ 25 ppb) and clinical evidence of T2 inflammation are also crucial in the assessment of patients who qualify for the biological therapy [8].
IL-5 as a therapeutic target
Due to the key role of IL-5 in the pathogenesis of severe eosinophilic asthma, novel biological therapies targeting its action have been developed [5]. The most important drugs include mepolizumab and reslizumab, which are monoclonal antibodies that block IL-5, and benralizumab, which acts on the IL-5 receptor (IL-5R), leading to a reduction in the number of eosinophils [7–9]. These therapies are effective in reducing inflammation, reducing the frequency of exacerbations, and improving disease control in patients for whom standard treatment is inadequate. Blocking IL-5 represents a breakthrough in the treatment of severe eosinophilic asthma, enabling a better quality of life and reducing the use of oral corticosteroids, which can cause numerous side effects [10]. Due to the important role of IL-5 in the pathogenesis of eosinophilic asthma, biological therapies targeting this cytokine have been developed [11].
Mepolizumab is a humanised monoclonal antibody of the IgG1κ class that binds specifically to free IL-5, preventing its interaction with the receptor on the surface of eosinophils. The drug is administered by subcutaneous injections every 4 weeks [11, 12]. The efficacy of mepolizumab has been confirmed in clinical trials, which showed that the use of the drug primarily reduces the number of eosinophils in the blood and respiratory tract [12–14]. The reduction in the number of eosinophils in the blood and airways caused by taking mepolizumab also makes it possible to reduce the oral corticosteroids taken by patients who take high doses of corticosteroids in severe asthma and are forced to take them chronically [12, 13]. Reducing the number of eosinophils in the blood results in a 50–70% reduction in the frequency of asthma exacerbations [13, 14]. Reducing the frequency of exacerbations and decreasing the doses of chronic corticosteroids taken by patients makes disease control easier and more affordable for patients, further increasing and improving patients’ quality of life [12–15].
Reslizumab is a monoclonal antibody of the IgG4 class that neutralises IL-5, thus preventing its interaction with the receptor on the surface of eosinophils. Reslizumab, compared to mepolizumab, differs in its method of administration, namely, it is used in an intravenous form, which is repeated every 4 weeks [16, 17]. Reslizumab has shown efficacy in reducing the number of eosinophils, which translates into improved lung and whole respiratory function in patients with severe eosinophilic asthma. What is more, with the use of the drug in patients, an improvement in lung functional parameters is observed, more precisely, an improvement in first-second expiratory volume is observed [18, 19]. The benefits that result from the use of reslizumab in patients include a reduction in the frequency of disease exacerbations, which also translates into improved asthma control by patients [19]. Additionally, the use of the drug results in patients experiencing a reduction in asthma symptoms, such as shortness of breath and wheezing [18].
Benralizumab, on the other hand, is an IgG1-class monoclonal antibody that does not directly block IL-5 but binds to the α subunit of the IL-5 receptor on the surface of eosinophils and basophils [20]. Due to this mechanism of action, the drug enables rapid and effective clearance of eosinophils through an antibody-dependent cell-mediated cytotoxicity (ADCC) mechanism that is activated by NK cells [21]. Benralizumab is administered similarly to mepolizumab through subcutaneous injections, initially with the first 3 doses every 4 weeks and subsequent doses administered every 8 weeks. Clinical trials using benralizumab have shown that it eliminates eosinophils almost completely from both the circulatory system and tissues. This results in a reduction in the incidence of asthma exacerbations by up to 70%. Enabling a reduction in doses of oral corticosteroids or even their complete discontinuation in those patients who use them chronically, the reduction in the frequency of exacerbations resulting from the elimination of eosinophils makes it possible to improve asthma control as well as lung function, which in turn translates into an improved quality of life for patients suffering from severe eosinophilic asthma [22]. For a comparison of the effectiveness of anti-IL-5 therapies in the treatment of severe eosinophilic asthma, see Table 1.
Table 1
Comparison of efficacy of anti-IL-5 therapies in the treatment of severe eosinophilic asthma. Source: Own study
Mepolizumab reduced the rate of clinically significant exacerbations by 44% (up to 68%) in addition to placebo; the ACQ-5 score improved by 0.27–0.59 points and the St George’s Respiratory Questionnaire total score by 5.0–11.6 points [23]. The introduction of antibody therapy reduced the annualised exacerbation rate significantly, by –3.79 (95% confidence interval [CI]: –4.53, –3.04), –3.17 (95% CI: –3.74, −2.59), and –6.72 (95% CI: –8.47, –4.97) with benralizumab, mepolizumab, and reslizumab, respectively [24]. Reduction of exacerbations was the most noticeable in dupilumab (RR = 0.32; 95% CI), comparing to the other drugs: mepolizumab (RR = 0.37; 95% CI) and benralizumab (RR = 0.49; 95% CI), and improvement of the spirometric parameter FEV1 was most prevalent in treatment with dupilumab [25].
Immunogenicity and safety profile
As with the use of other biologic drugs, the use of mepolizumab, reslizumab, or benralizumab in the treatment of eosinophilic asthma is associated with some challenges that may occur during the treatment of patients [26].
Anaphylaxis is extremely rare. Mepolizumab and benralizumab are approved for home use without need for epinephrine supervision [27]. All three drugs are used at fixed doses as per the drug label, without routine individualisation based on patient characteristics. At the beginning of therapy, monitoring of the patient’s condition is required during the first doses of drugs [28]. Moreover, data show no significant increase in infection risk or cancer incidence in patients treated with anti-IL-5/IL-5R therapies [29]. Anti-IL-5/IL-5R antibodies exhibit low immunogenicity, with rare anti-drug antibodies that have no proven impact on efficacy [30]. Mepolizumab is approved for children above 6 years of age, while benralizumab is approved for children 12 years old or above. Data during pregnancy are limited, so therapy is individualised. Both antibodies are also effective in eosinophilic granulomatosis with polyangiitis, hypereosinophilic syndrome, and chronic rhinosinusitis with nasal polyps [31].
Also, through regular monitoring of patients, it is possible to determine the correct effect of drugs on patients or the overall response to patients. This is important because some patients may not experience full improvement, which may be due to differences in response to therapy, and regular monitoring is able to determine whether a patient is responding to the treatment used [32]. Because treatments that rely on biologic drugs are expensive, the financial aspect can be a barrier to treatment availability for people with severe eosinophilic asthma. This is especially true in countries that have limited financial resources in the health care system. The price of treatment can be a particular problem for patients who require ongoing treatment during long-term therapy [33, 34].
Although mepolizumab, reslizumab, and benralizumab offer a modern approach to treating severe eosinophilic asthma, they also come with some challenges. Proper control and monitoring of patients are important to ensure that therapy is effective.
Conclusions
IL-5 plays a key role in the development of severe eosinophilic asthma. Its overactivation leads to an increase in the number of eosinophils, resulting in increased inflammation, damage to the bronchial epithelium, and worsening of asthma symptoms. Therapies that are based on monoclonal antibodies – –mepolizumab, reslizumab or benralizumab – effectively reduce the number of eosinophils and reduce the frequency of asthma exacerbations [35, 36].
Blocking IL-5 and its receptor allows for better disease control in patients in whom standard corticosteroid treatment is insufficient. The use of biological drugs that block IL-5 leads to a reduction in the number of eosinophils or their almost complete elimination. This results in a reduced need for oral corticosteroids, as well as improved quality of life for patients [37, 38]. There is a significant reduction in membrane thickness, airway smooth muscle layer thickness, and the number of eosinophils residing in tissue (p < 0.001). The scale of reduction in airway smooth muscle layer thickness positively correlates with the submucosal eosinophil reduction [39].
On the other hand, the use of such drugs in patients with severe eosinophilic asthma is also associated with the occurrence of allergic reactions or hypersensitivity to them. There is also an increased risk of upper respiratory tract infections due to the effect of the drugs on the immune system, which is weakened during therapy [40]. It should be noted that the variability of patients’ responses to treatment means that regular monitoring of patients is required, due to the possibility of infections or allergic reactions [41, 42].
In conclusion, IL-5 blockade is a breakthrough approach in the treatment of severe eosinophilic asthma, which limits the quality of patients’ lives across the world.