Introduction
Hidradenitis suppurativa (HS) is primarily a chronic, inflammatory skin disease that affects the folliculopilosebaceous unit [1]. HS is diagnosed by clinical examination and could be missed as other inflammatory skin conditions such as furunculosis or boils [2]. Furthermore, clinical examination and staging without imaging tests could be challenging. Skin ultrasound, magnetic resonance imaging (MRI), positron emission tomography, computed tomography and long-wave medical infrared thermography (MIT) have been valuable diagnostic tools in recent years. MIT is a contactless imaging method; use of this method is rapidly expanding in HS diagnostics due to its ease of access, its ability to be performed in surgical interventions, its dynamic real-time temperature measurements, and painless nature compared to other non-invasive diagnostic methods [3].
The main component of MIT is the thermography camera, which captures the intensity of long infrared rays, transforms them into electrical signals and compares them with the radiance dependence of the temperature in the memory of the camera [4]. These modern thermography devices are small, compact, with high sensitivity (0.02°C) and high resolution to provide sharper images. Over recent years, a few producers of infrared thermography appliance have started to make low-cost cameras with faulty features. That way the camera is unable to provide a reliable analysis [5].
The skin temperature determined by this method is visualized on thermograms and the surface temperatures are shown in different colours [6, 7]. Despite the fact that all bodies radiate heat, the temperature emitted by inflamed areas is higher than normal. Active metabolic reactions and vasodilatation of blood vessels during inflammation result in a high amount of infrared radiation that can be captured by MIT [8]. Due to ongoing inflammatory processes and possible bacterial infections, the affected skin areas of HS are exposed to high temperatures [9]. That way HS is an appropriate disease to perform MIT.
This review presents a comprehensive overview of MIT application for diagnostics and inflammation evaluation for HS patients.
Material and methods
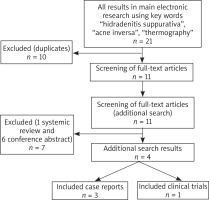
This protocol is under control with the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) statement [10]. During the period from 1 February to 17 April 2020, PubMed, Embase and Scopus databases were searched for studies published in the last 10 years employing the key words “hidradenitis suppurativa”, “acne inversa” and “thermography”. The following inclusion criteria were used in the search: clinical trials, case series or reports, articles in English. Exclusion criteria were systemic literature reviews, conference abstracts, articles in non-English. The primary search for “hidradenitis suppurativa”, “acne inversa” and “thermography” showed twenty-one articles. Seventeen articles were excluded because ten of them were duplicates, one article was a review and six articles were conference abstracts. In total, four articles fulfilled inclusion criteria and were reviewed for further data research. The detail search methodology is shown in Figure 1.
For clinical trials the risk of bias was assessed using the criteria generated by Downs and Black [11]. It is the feasibility of creating a checklist for the assessment of the methodological quality both of randomized and non-randomized studies of health care interventions. Reporting, external validity, bias, confounding and power were evaluated and the maximum achievable score was 27. The risk of bias of case reports was estimated using CARE guidelines and the maximum achievable score was 30 [12]. A clinical case published as Letter to the editor’s risk of bias was not evaluated due to the inconsistent structure of the column, but this article was included to increase the number of articles in the review.
Results
Among included studies, there were three case reports (one of them published as a letter to the editor) and one clinical trial. All 4 manuscripts emphasized MIT usability for HS patients. For the study by Zouboulis et al. [13] and by Downs and Black [11] the score was 8/27, accordingly to the reporting, external validity, bias, confounding and power. For the case reports, 15/30 [14] and 21/30 [15] points were calculated when evaluating CARE guidelines.
FLIR thermal imaging systems were used for HS investigation in all published articles (Table 1). Three articles [13–15] were presented for MIT to measure inflammation during the surgery. Zouboulis et al. [13] find that MIT is an effective additive surgery tool in 18 patients by using infrared camera FLIR T650sc (FLIR Systems, Wilsonville, OR). Standardized photography and MIT were performed at a distance of 50 cm from the affected skin surface during the surgery. Polidori et al. [15] performed MIT (IR FLIR SC620 camera) during surgery for a 36-year-old man with HS when diagnosis of disease was delayed by 7 years. What is important that an alcohol-based antiseptic solution was applied on the inflammatory lesions before the imaging to make dissimilarity between healthy and inflamed skin more obvious. Moreover, possibilities to combine MIT with other imaging tools were reported by Derruau et al. [14]. Authors demonstrated the potential of MIT for pre-surgical management of severe HS along with MRI. In this case report, 1.5T MRI instrument and FLIR SC620 camera (FLIR Systems, Wilsonville, OR) were used. Firstly, the inflammatory lesions were detected by MRI in the pre-operative period and the real-time MIT was performed in the surgical room. The inflamed areas have shown an increase in temperature (34.3°C) and after the removal of lesions the temperature has become similar to the healthy skin sites. The images of both methods were to help the surgeon choose the best treatment option. In the last case report by Nazzaro et al. [6], the combination of MIT and skin ultrasonography was used to estimate the inflammation for an HS patient. A 26-year-old man with HS was clinically examined, colour Doppler and MIT were performed to evaluate the inflammation. The patient was scanned by MIT at 22 ±2°C 10 min after acclimatization.
Table 1
The summary of the studies included in this review (FLIR – forward-looking infrared camera, MIT – medical infrared thermography, MRI – magnetic resonance imaging)
| Author and year | Type of study | Number of patients | Age [years] | Hurley score | Infrared camera | Methods | Results | |
|---|---|---|---|---|---|---|---|---|
| Polidori et al. 2017 [15] | Case report | 1 | 36 | II | IR FLIR SC620 camera (FLIR Systems, Wilsonville, OR) | Alcohol-based antiseptic solution was applied on the inflammatory lesions MIT was performed in pre- and perioperative phases of the surgical procedure | MIT could be useful to know the true extent of the disease during surgery After inflammatory lesions are excised, MIT can be easily repeated in the surgery room to establish remains of HS | |
| Derruau et al. 2018 [14] | Case report | 1 | 28 | III | IR FLIR SC620 camera, FLIR Systems, Wilsonville, OR) | 1.5T MRI instrument was used to perform MRI before surgery Skin excision with an electrical scalpel MIT was repeated during surgery | MRI findings: two chronic abscesses in the right groin area and perianal region; subcutaneous fistula in the left perianal region MIT findings: inflammatory lesions were noticed by an increase in temperature (34.3°C) MIT allows to distinguish healthy areas of the skin from inflamed areas during surgery | |
| Zouboulis et al. 2019 [13] | Clinical trial | 18 | Median age 38.75 (95% CI: 28.5–51) | I – 5.5% II – 38.9% III- 55.5% | IR FLIR T650sc (FLIR Systems, Wilsonville, OR) | Standardized photography of the affected areas of the skin was done Patients were scanned by MIT at a distance of 50 cm from the skin surface MIT was repeated during surgery | MIT findings: axillary area: healthy skin temperature – 33.0°C, inflamed skin – 35.0–36.6°C. Groin: 33.0°C for healthy skin, 35.4–36.9°C for the central part of inflamed skin MIT is a valuable tool to be used in surgery and determine margins of inflammatory lesions | |
| Nazzaro et al. 2020 [6] | Case report | 1 | 26 | I | AGA thermovision 782, FLIR system | Clinical examination and skin ultrasonography (Hitachi Arietta) were performed MIT was done at 22 ±2°C, 10 min’ acclimatization | Clinical examination: two inflammatory nodules in the right and left axillary regions Skin ultrasonography: hypoechogenic areas (fistulas) on the right and left axilla MIT results between left and right axillary regions: 35.6°C and 34.3°C respectively |
Zouboulis et al. [13] in the MIT examination revealed 1°C temperature difference between healthy and inflamed skin areas of a symmetric body region. Moreover, this study has shown an increased temperature over 35°C of generally inflamed skin while the average temperature of healthy skin was 33°C. Additionally, the margins of the inflammatory lesions identified by MIT were used in the surgical treatment. Also, real-time measuring was reused during the surgery to evaluate depth of inflammation and to control the excision area. In a similar way Polidori et al. [15] in their case report used MIT to detect the margins of the inflammatory area and they were excised 1 cm apart from the side of activity. The temperature of the inflammatory lesion in the left axilla area was 34.8°C while healthy skin temperature was about 30.0°C. After finishing the surgery, real-time measuring with MIT was repeated and showed non-excised remains of inflamed lesions which was removed successfully.
In Derruau et al. [14] case, MRI before the surgery revealed two chronic abscesses in the right groin area and perianal region and subcutaneous fistula in the left perianal region. During MIT, inflammatory lesions were noticed by an increase in temperature. The results revealed that the combination of MIT and MRI could be useful to detect HS-affected sites in the pre-surgical period and MIT is a suitable real-time method to check healthy areas of the skin from inflamed during surgery. In next case report by Nazzaro et al. [6], skin ultrasound and MIT were combined. Clinical examination confirmed two inflammatory nodules in the right and left axillary regions while skin ultrasonography revealed hypoechogenic areas (fistulas) on the right and left axilla. Moreover, colour Doppler showed higher peripheral vascularization on the left axilla compared with the right axilla. MIT showed the temperature difference between left and right axillary regions of 35.6°C and 34.3°C respectively. The authors suggest that a combination of MIT and skin ultrasound could be a valuable tool for inflammation evaluation in HS patients.
Discussion
MIT is a promising tool and its usage for HS is evolving. All reviewed articles have been published in the last 3 years. Nevertheless, statistics on this topic are lacking. Only one study (n-18) [13] has been described and several related clinical cases have been reported [6, 14, 15]. Due to that, a precise standardized methodology of MIT is still missing. Prior to the procedure, the patient should acclimatize for at least 10 min at room temperature [6]. Alcohol-based antiseptic could be used before MIT to retain the temperature, cool the skin and to make the inflammation of the skin more visible when comparing with healthy skin [15]. Also, MIT should followed the same standardized HS patient photo documentation procedure [13, 16].
The application of MIT occurs in several ways. As an imaging biomarker this method can be used to assess the inflammation in HS, treatment effectiveness, but standardized scales have still not been published [13]. Due to inflammation evaluation, authors suggest that MIT could be suitable during surgical procedures: to optimize surgery and remove remains of inflammatory lesions. This can be done through documentation before, during and after the surgery [13–15]. Excision margins should be located within 1 cm of the inflamed edges. After the surgery the wounds are more likely to heal in a secondary intention healing way. Because of this, MIT improves HS outcomes [15]. Also, the combination of MIT and ultrasonography could be used to amplify the sensitivity for inflammation estimation and help for staging HS [6]. It is clear that after ultrasound examination for HS patients, treatment tactics change for 82% of adults [17] and 93% of children [18]. Unfortunately, similar data for MIT have not been published.
This review has several limitations. Given the limited data published on MIT as an inflammatory marker for HS, we included all published data regardless of their design and methodology. Also, studies do not compare MIT with other imaging tests as ultrasonography or MRI. As a result, we were unable to conclude evidence-based advantages of MIT when comparing with other imaging methods.
It is important to mention that MIT could be used not only for HS patients, but it is also a beneficial technique for other dermatological diseases. For example, MIT is a reliable and effective way to identify activity of localized scleroderma without atrophy of the skin in paediatric and adult patients [19]. Furthermore, thermography is a good tool to exclude allergic and irritant contact dermatitis in patch testing [20]. Also, studies discussed the possibility of using MIT to spot melanoma at early stages [21]. In addition, thermography can be used as a method to provide real-time results in a variety of dermatological procedures. For example, skin cooling is assessed during cryotherapy or laser parameters can be adjusted during laser procedures using MIT [22]. Consequently, the applicability of MIT in dermatology will gradually increase in the future.
Conclusions
A combination of imaging tests such as MIT, MRI, and ultrasonography could facilitate the diagnosis and staging of HS. Moreover, collection of MIT data could be helpful to evaluate the severity, treatment effect and to improve follow up of HS patients. There is no alternative imaging tool that quickly, painlessly and objectively evaluate the inflammation area and depth during surgical procedures. Despite these facts, more purposeful research is needed to standardize the monitoring of HS patients.