Introduction
Peanut allergy is one of the most common food allergies in children [1]. The estimated prevalence of oral food challenge confirmed allergy to peanuts in Europe is 0.2% [2]. Based on recent studies, the prevalence of peanut allergy may be increasing [3]. Peanut allergy causes a significant health care and socioeconomic burden, as it is likely to persist throughout life [4]. The presentation of allergy to peanuts depends highly on the sensitization pattern of an allergic individual, and allergic reactions experienced by patients with a peanut allergy may range from mild to severe [1]. A mono-sensitization to birch pollen protein Bet v1 homolog cross-reactive peanut protein Ara h 8 is usually asymptomatic or manifests in milder reactions such as oral allergy syndrome [5]. In contrast, sensitization to seed storage proteins Ara h 1, 2, 3, and 6 can cause more severe allergic reactions in peanut-allergic patients [6]. Sensitization to Ara h 9 – a nonspecific lipid-transfer protein (LTP) – may lead to symptoms ranging from oral allergy syndrome to systemic reactions [7]. Ara h 15, a protein belonging to the oleosin family, is associated with a more severe allergy [8].
The exact prevalence of peanut allergy in Lithuania is currently unknown. The prevalence estimate of peanut sensitization in 7- to 10-year-old Lithuanian children was 2.65% [9]. Birch is a highly prevalent tree species in Lithuania [10]; therefore, a fraction of described sensitizations could have been related to Bet v 1 cross-reactivity. Currently, no in-depth studies describe the peanut sensitization molecular profiles in Lithuanian children. As the dynamics of peanut allergy change, sensitization profiles could help determine the need for interventions, such as early peanut introduction in high-risk children to prevent the development of peanut allergy [11].
Aim
We aimed to describe the age-related patterns of molecular peanut sensitization profiles among Lithuanian children with suspected allergic symptoms.
Material and methods
We performed a retrospective analysis of peanut sensitization profiles in 576 Lithuanian children up to the age of 18 with suspected allergic symptoms. No information on the patient’s clinical reactivity to peanuts was available. To analyse age-related food sensitization patterns, patient data were categorized according to age groups: 0–2, 3–6, 7–12, and 13–18 years. Specific immunoglobulin E (sIgE) levels to peanut molecular components: Ara h 1, Ara h 2, Ara h 3, Ara h 6, Ara h 8, Ara h 9, Ara h 15, and birch major allergen component Bet v 1 were measured using a multiplex allergy test (ALEX² Allergy Explorer). The sensitization was defined by detecting a sIgE level greater than 0.3 kUA/L. Analyses were performed using Microsoft Excel and SPSS for Windows (release 27.0.1; SPSS Inc., Chicago, IL, USA). Categorical data were reported as percentages showing the proportion of positive results. The Vilnius Regional Biomedical Research Ethics Committee provided ethical approval to perform this retrospective study.
Results
Ninety-one (15.8%) children were ≤ 2,241 (41.8%) 3–6, 159 (27.6%) 7–12 and 85 (14.8%) ≥ 13 years old. Around half of the children were male – 332 (57.6%). Sensitization to Bet v 1 was observed in 206 (35.8%) children. Sensitization to at least one peanut protein was observed in 148 (25.7%) children. Most sensitized children were mono-sensitized to one peanut allergen component – 86 (58.1%) and 62 (41.9%) were sensitized from two to six allergen components. Overall, 68 (11.8%) children were sensitized to Ara h 1, 34 (5.9%) to Ara h 2, 34 (5.9%) to Ara h 3, 36 (6.3%) to Ara h 6, 88 (15.3%) to Ara h 8, 23 (4.0%) to Ara h 9 and 4 (0.7%) to Ara h 15.
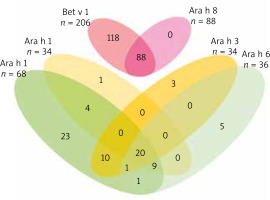
A considerable sensitization overlap was found with Bet v 1 and Ara h 8 components – 88 (42.7%) Bet v1 sensitized children were also sensitized to Ara h 8, and all (100%) children sensitized to Ara h 8 were sensitized to Bet v 1. A sensitization overlap was also found among seed storage proteins. In total, 77 (52.0%) children sensitized to any peanut molecular component were sensitized to at least one seed storage protein, of which 20 (26.0%) were sensitized to all seed storage proteins. A mono-sensitization among seed storage proteins was found in 23 (29.9%) children sensitized to Ara h 1 and less frequently to other components: Ara h 2, Ara h 3, and Ara h 6 in only 1 (1.3%), 3 (3.9%) and 5 (6.5%) children, respectively (Figure 1).
Figure 1
Venn diagram showing the distribution of sensitization to structurally related molecular components
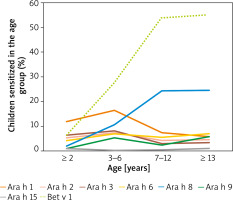
In children ≤ 2 years, most children were sensitized to Ara h 1 – 11 (12.1%). In children aged from 3 to 6 years, the sensitization to Ara h 1 remained the most prevalent in 40 (16.6%) children, followed by sensitization to Ara h 8 in 26 (10.8%). The most prevalent sensitization in children aged from 7 to 12 years was to Ara h 8 in 39 (24.5%), and sensitization to Ara h 1 was the second most common in 12 (7.5%) children. In children aged ≥ 13 years, Ara h 8 remained the most prevalent sensitizer in 21 (24.7%) children. Sensitization to Bet v 1 demonstrated a similar pattern as Ara h 8, being uncommon in children ≤ 2 years of age observed in 6 (6.6%) children, but promptly increasing in children aged 3–6 years – 67 (27.8%) and peaking at 7–12 and ≥ 13 years in 86 (54.1%) and 47 (55.3%), respectively (Figure 2).
Discussion
Peanut allergy is exceedingly common in children [1] and can cause a significant burden on society and the health care system [4]. The incidence of peanut sensitization in Lithuanian children is substantial [9], but the distribution of sensitization patterns, which are related to clinical reactivity, was unknown. This study examined the molecular sensitization patterns of Lithuanian children with possible allergic complaints; therefore, it represents the population of children admitted to the allergy clinic rather than healthy children. The sensitization patterns were examined using ALEX² Allergy Explorer multiplex allergy test, which demonstrates excellent sensitivity (94.7%) and specificity (95.7%) in diagnosing peanut allergy [12].
We found that around one-fourth of Lithuanian children with suspected allergic symptoms were sensitized to peanuts. Sensitization to clinically relevant molecules such as seed storage proteins was not uncommon. Most Lithuanian children with suspected allergic symptoms were sensitized to Ara h 8 and Ara h 1 components, and sensitization to Ara h 9 and Ara h 15 components was rare. A Norwegian study, which examined sensitization patterns in children and adults, reported Ara h 1, Ara h 2, Ara h3, and Ara h 8 components as the most frequent sensitizers in children and young adults and showed lower rates of Ara h 9 sensitization [13]. A study of peanut sensitized children and adults carried out in the United States of America (USA) reported the most prevalent sensitizations to Ara h 2, followed by Ara h 1, and lower overall sensitization to Ara h 9 [14]. In contrast, an Italian study reported Ara h 9 as the main sensitizer followed by Ara h 8 in children with a suspected nut allergy, while sensitization to Ara h 1 was less frequent [15]. It is evident that a more geographically related region like Norway demonstrates a similar sensitization pattern. Meanwhile, Italy, a geographically diverse country, presents a more varied sensitization profile, influenced by LTP sensitization and Bet v 1 sensitization. Interestingly, in the USA, which is also geographically diverse and influenced by birch pollination, the sensitization to seed storage proteins Ara h 2 and Ara h 1 was the most common in children up to young adults. These differences highlight the importance of geographical location on the peanut sensitization patterns in different countries. However, variables such as dietary preferences can likely influence the differences between different regions. Similar findings were reported in a study comparing the prevalence of sensitization to peanut molecules across three distinct regions: Spain, Sweden, and the United States [16]. Ara h 2 was the most common sensitizer in the United States and Sweden; Ara h 8 was the second most common sensitizer in Sweden. In comparison, most Spanish patients were sensitized to Ara h 9, which was not common among American and Swedish patients [16]. In this study, a geographically similar country – Sweden, has demonstrated predominant sensitization to Ara h 2; however, this study analyzed the prevalence of sensitization in symptomatic children as opposed to the sensitized population; therefore, a lower frequency of clinically insignificant sensitizations, such as Ara h 8 could have been observed in this group of patients.
In our study, 58.1% of children were mono-sensitized to one molecular peanut component; similar findings were reported in the Italian study, where 54% were sensitized to only one allergenic component [15]. Considerable overlap was seen in children sensitized to Bet v 1 and Ara h 8. Around 43% of Bet v 1 sensitized children were sensitized to Ara h 8, and all Ara h 8 children were sensitized to Bet v 1. Similarly, in the Norwegian study, half of the peanut extract sensitized patients were sensitized to Ara h 8, and almost all were co-sensitized to birch pollen extract [13]. Co-sensitization to multiple seed storage proteins was common in Lithuanian children, and monosensitization was most common to Ara h 1. Similar findings of seed storage protein co-sensitization were reported both in the Norwegian and the USA studies [13, 14]; however, unlike in our study, both studies demonstrated the most common monosensitization to Ara h 2 [13, 14].
Age-dependent sensitization patterns were observed in our study. In children up to 6 years, Ara h 1 was the most common sensitizer, whereas, in children 7 years and over, sensitization to Ara h 8 was the most frequent. In the Norwegian study, sensitization to Ara h 2 was the most common and Ara h 1 the second most common in children aged up to 5 years old, with an increasing prevalence of Ara h 8 sensitization with age [13]. Comparable results were reported in the USA study – Ara h 2 was the most common sensitizer in children and young adults, but the prevalence of Ara h 8 increased gradually throughout childhood, eventually surpassing seed storage protein sensitization prevalence in adulthood [14]. In the Italian study, Ara h 1-3 were the main sensitizers in preschool children, and sensitization to Ara h 9 and Ara h 8 was most often seen in school-age children and adolescents [15].
Generally, our study results correspond to other studies and demonstrate a predominant seed storage protein sensitization pattern in young children, as well as a growing prevalence of Bet v 1 dependent Ara h 8 sensitization with age; however, one peculiarity is a high Ara h 1 mono-sensitization rate in Lithuanian children. Although we did not have the clinical reactivity data in these children, this highlights the importance of performing sensitization testing to all molecular peanut components as seed storage protein sensitization is associated with an increased risk of severe allergic reactions upon contact with peanuts [6]. Therefore, in this subcategory of children, if clinical reactivity to peanuts is unknown, it should be clarified by performing an oral food challenge. In children presenting with Bet v 1 dependent Ara h 8 sensitization, without seed storage protein, or LTP sensitivity oral food challenges can be avoided as it is usually asymptomatic or manifests in milder reactions such as oral allergy syndrome [5].
Conclusions
One-fourth of children with suspected allergic symptoms are sensitized to at least one peanut molecular component in Lithuania. The sensitization to Ara h 8 and Ara h 1 is the most common in all age groups. Age-dependent patterns were observed in different age groups; infants and preschool children are most frequently sensitized to seed storage proteins. School-age children are most often sensitized to Bet v 1 homolog Ara h 8 molecular allergen.