Introduction
The continual evolution of medical science has contributed to an expanding elderly demographic, where issues such as immobility and falls pose challenges, potentially resulting in physical injuries. Consequently, there has been a heightened emphasis on the care and recuperation of wounds among the elderly, leading to an increased scholarly focus on wound dressing research. A wound surface manifests as the consequence of damage inflicted upon normal skin or tissue due to external factors like surgery, external force, heat, electric current, chemicals, low temperature, or internal factors such as disruptions in local blood supply [1].
The intricate process of wound healing involves the regeneration of adjacent tissue, formation of granulation tissue, or development of scar tissue, orchestrated by a myriad of synergistic effects. The pivotal prerequisite for expeditious wound healing lies in the restoration of the original physiological environment surrounding the wound. Protracted or aberrant repair processes may culminate in the loss of glands, skin, and hair on the wound surface, potentially resulting in infection and necrosis of tissue [2]. Categorically, wound surfaces are classified into acute and chronic stages based on their healing trajectory. Acute wounds, stemming from mechanical trauma, burns, or chemical burns, typically exhibit complete healing within a span of 2–4 weeks. Conversely, chronic wounds, characterized by a healing cycle surpassing 4 weeks and marked by recurrent incidents, are often associated with conditions such as diabetes, tumours, and aging [3]. Optimal wound healing necessitates meticulous cleaning for basic wounds and rigorous anti-infection treatment for severe wounds. However, chronic wounds, exemplified by deep burns and pressure ulcers lacking intrinsic biological factors, confront a limitation in self-healing capabilities, thereby warranting a more discerning approach in the course of wound treatment [4].
The canonical phases of wound healing encompass haemostasis, inflammation, cell proliferation, and skin remodelling. Chronic wounds are predisposed to iterative inflammatory reactions, resulting in heightened levels of cell exudate, neutrophils, surface bacteria, inflammatory factors, and toxic proteins [5–7]. The intricate nature of the wound healing process involves a nexus of cellular events, biomarkers, and specialized wound dressings to facilitate effective tissue repair [8]. Matrix metalloproteinases assume a pivotal role in promoting cell migration, reorganization, and degradation of the extracellular matrix. Excessive production, however, can lead to unwarranted matrix degradation. The surplus of cell osmotic fluid can provoke contact dermatitis, inducing skin swelling, infection, and malodorous manifestations. Consequently, the treatment paradigm for chronic wounds requires the implementation of professional wound dressings designed to absorb surplus wound exudate [9].
The aging of the population induces a cascade of physiological transformations, encompassing weakened skin and sluggish blood circulation, ultimately resulting in delayed wound healing among the elderly. To address this concern, a diverse array of wound dressings has been innovatively developed, including both dry and wet dressings. These dressings not only serve to cover wounds but also actively contribute to the promotion of the healing process.
Historically, prior to the 19th century, wounds were typically bound with permeable cloth, offering limited interference with wound drainage. However, these dressings had the drawback of adhering to wounds, causing discomfort and potential damage during dressing changes, hindering the healing trajectory. In 1800, the introduction of the “Gamgee” wound dressing marked a significant advancement, allowing for prolonged maintenance of wound dryness and stabilization. Subsequently, in 1962, another breakthrough emerged when a researcher proposed that maintaining a moist environment could expedite wound healing by twofold compared to traditional dressings while minimizing scar formation [10].
The contemporary landscape of wound care has seen the advent of nanofiber wound dressings, representing a novel approach to medical wound management. In contrast to traditional dressings, nanofiber wound dressings exhibit superior gas permeability and hydrophilicity, establishing an optimal tissue environment conducive to accelerated wound healing. Furthermore, nanofiber wound dressings demonstrate commendable biocompatibility, posing no discernible toxicity or irritation to the human body. Research has additionally highlighted their capacity to offer enhanced wound coverage and support, sustained release of bioactive substances, and antibacterial effects, all of which contribute to the facilitation of wound healing [11].
Aim
This study aims to investigate the application of nanofiber wound dressing in the context of wound care for elderly patients. The primary objectives involve assessing the biocompatibility and therapeutic efficacy of nanofiber wound dressing. The aim is to substantiate that nanofiber wound dressing proves suitable for the wound care of elderly patients, effectively alleviating the pain associated with delayed wound healing and infections. This research presents nanofiber wound dressing as a novel, secure, and efficacious treatment modality for addressing the specific challenges of wound care in the elderly population.
Material and methods
Study subjects
In a prospective study conducted at the Affiliated Hospital of the Shaanxi University of Chinese Medicine from January 2023 to September 2023, a total of 86 elderly patients with wounds were enrolled. The participants were randomly assigned to either the control group or the experimental group, with each group comprising 43 patients.
Inclusion criteria
Patients meeting specific inclusion criteria were considered eligible for the study. These criteria encompassed individuals with conditions such as diabetic foot, pressure ulcers, burn wounds, and other ailments posing challenges to natural wound healing. The recruitment period spanned from January 2023 to September 2023, targeting those experiencing chronic wounds persisting for an extended duration. The study emphasized voluntary participation, requiring individuals to willingly agree to be part of the research. Additionally, participants were required to possess unobstructed communication abilities to facilitate effective interaction during the study. Normal pain perception was a crucial criterion to accurately assess the impact of nanofiber wound dressing without confounding factors related to altered pain perception.
Exclusion criteria
Conversely, exclusion criteria were defined to exclude certain individuals from the study. This included individuals below the age of 60, those with acute wounds, and those outside the specified elderly age group. Patients who refused to participate or withdrew consent were excluded, ensuring commitment throughout the research. Exclusion extended to individuals with incomplete data, those with prior exposure to nanofiber dressing treatments, and those concurrently participating in other advanced wound healing treatments. Severe allergies to dressing material were additional exclusion criteria. All procedures in this study received approval from the ethics committee of the hospital, and informed consent was obtained from all subjects.
Care methods
Upon admission, patients in both groups received routine care services, with regular monitoring of body temperature, blood glucose, blood pressure, and other relevant parameters. For wound treatment, isotonic saline was employed for initial wound cleaning, followed by the use of medical alcohol or disinfectant for further cleaning and disinfection. Sterile tweezers and scissors were used to remove necrotic tissue around the wound. Patients with large blisters due to burns underwent controlled drainage, with preservation of intact blisters to prevent infection. Subsequently, isotonic saline was used to rinse the residual disinfectant, and the wound and surrounding skin were dried with gauze. Sterile cotton swabs were then used to evenly apply the designated treatment drugs to the wound. Dressings were changed every 2 days or more frequently based on the dressing’s exudation. Gentle handling during dressing changes aimed to prevent secondary tissue damage caused by tearing the wound. During each dressing change, wound exudate was collected and cultured in a dish, with observations and recordings of plaque formation. Detailed records of patient’’ wound healing status were maintained.
Control group
After disinfection and debridement, wounds in the control group were evenly treated with traditional dressing. The dressing extended 2 to 4 cm beyond the wound, with the outer layer covered by sterile gauze. Sterile gauze was then fixed on the wound using a bandage or tape.
Experimental group
The experimental group underwent nanofiber wound dressing (20163640190) in addition to conventional treatment after disinfection and debridement. Similar to the control group, the dressing extended 2 to 4 cm beyond the wound, and the outer layer was covered with sterile gauze, fixed on the wound using a bandage or tape
Observation indicators
After admission, the patient’s age, gender, wound factors, wound area, wound healing time, wound healing area, degree of wound infection (positive rate of bacteria), therapeutic effect, and visual analogue scale (VAS) score following treatment were recorded.
The evaluation criteria of treatment were cure, effective, and ineffective. Complete healing was achieved when the patient’s wound healed completely and new granulation began to grow in the tissue under the wound. Effective healing was defined as wound healing area > 1/3. ≤ 1/3 was considered invalid. Total effective rate = (cured + effective) number/total sample number of the group × 100%.
During the treatment, VAS [12] was used to evaluate the degree of pain before treatment and nursing, 8 days of care, and 16 days of care, with a full score of 10. The lower the patient’s pain, the smaller the score.
Wound infection rate: the number of wound infection in the group/the total number of patients in the group treated × 100%. The wound secretion was collected for bacterial culture, and the infection was represented by the positive rate of bacteria.
Inflammatory factors: fasting venous blood (3 ml) was drawn from the patients before and after treatment, then collected, and centrifuged. Interleukin-1β (IL-1β) and tumor necrosis factor-α (TNF-α) were detected by THE enzyme-linked immunosorbent assay.
The adverse reaction rate: the abnormal number of patients in the group/the total number of patients in the group treated × 100%. The abnormal rate of carcinoembryonic antigen (CEA), a-fetoprotein (AFP), renal function, and abnormal liver function of the two groups were detected, and the adverse reaction rate was statistically analysed.
Results
Comparison of general data
There were 24 males and 19 females, aged from 61 to 83 years, with a mean age of 73.25 ±2.57 in the controls. The experimental group had 23 males and 20 females, aged from 60 to 85 years, with an average age of 72.78 ±2.85. The two groups were similar in age and gender (p > 0.05) (Figure 1).
Comparison of clinical efficacy
Figure 2 A shows that 24 (55.81%) cases were cured and 13 (30.23%) cases were effective in the controls, while 37 (86.05%) cases were cured and 5 (11.63%) cases were effective in the experimental group. Figure 2 B presents that the total effective rate of treatment in the experimental group (97.67%) was higher relative to controls (86.04%) (p < 0.05).
Figure 2
Contrast of the clinical treatment outcomes. A – the statistics of the cured, effective, and ineffective number, B – the comparison of the total effective rate of the two groups
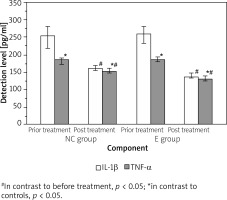
There was no obvious difference in the levels of IL-1β and TNF-α between the two groups pre-treatment (p > 0.05). Following the use of the nanofiber wound dressing, the levels of IL-1β and TNF-α in the two groups were lower as against before treatment (p < 0.05), and the differences were statistically significant when in contrast to the controls (p < 0.05) (Figure 3).
Comparison of therapeutic observation indicators
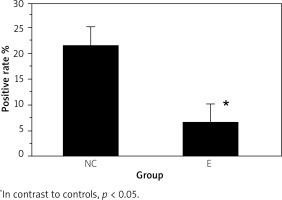
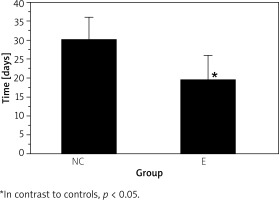
The experimental group had lower wound germiculture positive (7.14 ±2.76%) than controls (22.13 ±3.27%) (p < 0.05) (Figure 4). The wound healing time of the experimental group (18.68 ±5.78 d) was shorter as against controls (30.24 ±6.19 d) (p < 0.05), and the results are illustrated in Figure 5.
Rate of adverse reactions
The CEA, AFP, abnormal renal function, and liver function were similar in the two groups (p > 0.05) (Figure 6).
Treatment of pain
The VAS score of the experimental group (2.68 ±0.41 s) was lower as against controls (3.57 ±0.89 s) after 16 days of care with nanofiber wound dressing. This indicated that the effect of wound care using nanofiber wound dressing for exchange was more marked (p < 0.05) (Figure 7).
Discussion
The present study aimed to investigate the effect of wound dressing based on nanofibers on wound healing in elderly patients. The findings of the present study provide convincing evidence of the efficacy of nanofiber wound dressings in the field of wound healing for elderly patients. Demographic comparison achieved through randomization ensures that observed differences in outcomes can be attributed to the intervention rather than baseline changes. Significantly higher healing rates and overall efficacy in the experimental group demonstrate the clinical benefits of using nanofiber dressings and demonstrate its potential as a superior therapeutic approach (higher rates of cure (86.05%) and effectiveness (11.63%) compared to the controls, where 55.81% were cured and 30.23% were deemed effective). In line with our study, Meamar et al. employed nanofibers with human placenta-derived mesenchymal stem cells, confirming nanofiber dressings’ effectiveness in diabetic foot wound healing [13]. The porosity and high permeability of nanofiber dressings contribute to their effectiveness in wound treatment [14]. Consistent with our study, the findings of Kolarijani et al. study validate the advantageous wound healing properties of scaffolds incorporating gold nanoparticles, which outperform sterile gas in promoting wound healing. Additionally, the study showcases the efficacy of nanofiber matrices containing gold nanoparticles in effectively treating wounds [15]. Cytotoxicity and antibacterial test results of the study by Cui et al. showed that DCH/PLA nanofibers showed good cytocompatibility with L929 mouse fibroblast cells and showed positive antibacterial activity against Escherichia coli, indicating its ability to treat of infectious wounds [16].
The analysis of inflammatory markers, IL-1β and TNF-α, before and after treatment indicates a significant reduction in both groups post-application of nanofiber dressing. The more pronounced decrease in the experimental group suggests a potential anti-inflammatory effect associated with the use of nanofiber dressings. This is a promising finding that aligns with the observed superior clinical outcomes. Kanji et al. studied the effects of polyethersulfone nanofibers on skin wounds caused by streptozotocin-induced NOD/SCID diabetic mice, and the results showed a decrease in the sustained proinflammatory activity of NF-κB and that its downstream effector molecules TNF-α, IL-1β, and IL-6 were associated in the wound bed and caused a decrease in the number of myeloperoxidase-positive neutrophils and an increase in the level of IL-10 [17].
Further investigation into microbial control and wound healing time reaffirms the positive impact of nanofiber wound dressing. The lower percentage of wound germiculture positivity and the shorter healing time in the experimental group emphasize the antimicrobial properties and accelerated wound healing facilitated by nanofiber dressings. Consistent to our study, the study of Guo et al. showed that α-lactalbumin nanofiber dressing can promote burn wound healing and reduce scar [18]. In line with our study, the results of Liu et al. study showed that electrospun chitosan composite nanofiber dressing containing Cur@β-CD/AgNPs nanoparticles consisting of silver and curcumin has synergistic effects on antibacterial activity and wound healing [19]. Also, Schulte-Werning et al. showed that chloramphenicol anti-inflammatory properties, along with high absorption capacity and antimicrobial effect, is a suitable nanofiber dressing for the treatment of infectious wounds caused by burns [20]. These results are vital markers of the dressing’s effectiveness in establishing an ideal environment for wound healing.
The present study demonstrates that there were no significant negative responses or variations in several physiological parameters across the groups, indicating that the nanofiber wound dressing is safe and well tolerated. Furthermore, the decrease in discomfort seen, as evidenced by the lower Visual Analogue Scale (VAS) score in the experimental group, highlights the possibility of enhancing patient comfort during wound care exchanges. In line with our study, Cremar et al. showed that compared to local agents, hybrid fibre dressings are effective for accelerating the healing process and recovery period by preventing the growth of bacteria and the spread of infection and reducing pain [21]. Aderibigbe et al. also showed that alginate nanofibrous wound dressing absorbs excess wound fluid, maintains a physiologically moist atmosphere, and reduces the risk of bacterial infections at the wound site [22].
Overall, the present study offers a thorough comprehension of the diverse advantages linked to nanofiber wound dressing, including its clinical effectiveness, ability to reduce inflammation, capacity to regulate microbial growth, ability to speed up wound healing, and enhancement of patient comfort. These findings provide beneficial perspectives in the field of wound care and justify future investigation in greater clinical settings.
Conclusions
Nanofiber-based wound dressings exhibit favourable attributes such as excellent biocompatibility, air permeability, suspension, biodegradability, and biological activity, establishing them as highly promising and innovative medical material. Within the context of wound care for elderly patients, nanofiber-based dressings demonstrate notable efficacy in promoting wound healing, preventing infections, reducing healing duration, and enhancing overall healing quality. Consequently, these characteristics position nanofiber-based wound dressings as a significant and promising avenue in the spectrum of wound healing care for the elderly.