Introduction
Chronic spontaneous urticaria (CSU) is a disorder characterized by the spontaneous development of wheals (hives), angioedema, or both, which last for more than 6 weeks [1]. Its prevalence is up to 1% of the population at any time [2]. Non-sedating, second-generation H1 antihistamines (H1AH) are accepted as a first-line treatment by the European Academy of Allergy and Clinical Immunology, the Global Allergy and Asthma European Network, the European Dermatology Forum, and the World Allergy Organization guidelines. Dosage may be increased up to four-fold if needed [1].
Omalizumab, which is a recombinant, humanized monoclonal antibody, blocks IgE binding to high-affinity receptors on effector cells, and inhibits IgE-mediated cellular responses. It is currently used worldwide for the treatment of allergic asthma and also licensed for the treatment of CSU in patients who do not respond properly to increased doses of H1AH and remain symptomatic in Europe and the United States of America.
When the current literature is reviewed, it can be seen that publications concerning treatment with omalizumab in CSU have increased especially in the last decade, and current studies on efficacy, safety, and dosage are still being added to the literature [3]. Since the efficacy, safety, and effective dosages of drugs can be affected by many factors, studies of different races and regional patient groups can be followed in the literature [4, 5].
Aim
In this retrospective single-centre observational study, we only intended to provide data for the population in the north-western region of Turkey in order to identify outcomes obtained with omalizumab using the data from our clinic with 175 CSU patients refractory to conventional therapy.
Material and methods
This was a single-centre retrospective study, which was performed in the allergic disease outpatient clinic of the Dermatology and Venereology Department of the Sakarya University Training and Research Hospital. Patients diagnosed with CSU and treated with omalizumab from September 2015 to September 2020 were recruited to the study. The research protocol was submitted and approved by the Sakarya University Ethics Committee and the research was performed following the ethical regulations of the Declaration of Helsinki and in adherence to Turkish law and regulations.
Patients treated with omalizumab continuously for at least 6 months were included in the study. The standard treatment schedule was subcutaneous omalizumab 300 mg every 4 weeks at the outpatient clinic under surveillance. Demographic data, family history, co-morbidities, disease duration, serum concentration of total IgE, weekly urticarial activity score (UAS7), treatments administered concurrently with omalizumab treatment, and side effects observed during omalizumab treatment were recorded retrospectively from patient records. Disease duration was categorized as: < 1 year; 1 year to < 5 years; 5 years to < 10 years; and ≥ 10 years.
Efficacy of the drug was assessed by subjective clinical response: results were collected before omalizumab treatment and at the 3rd month and 6th month of omalizumab treatment from patient charts. The patients were classified as severe CSU (UAS7: 28–42), moderate CSU (UAS7: 16–27), mild CSU (UAS7: 7–15), and well-controlled CSU (UAS7: 0–6) according to UAS7 scores. In addition, a decrease of more than 90% in the UAS7 scores was recorded as a complete response and was compared with the current scientific literature.
Statistical analysis
Statistical analysis was performed using SPSS for Windows version 20.0 (SPSS Inc., Chicago, IL, USA). All data were reported as mean ± standard deviation. Descriptive statistics, Kruskal Wallis analysis, and χ2 test were used for statistical analyses. A p-value less than 0.05 was considered significant.
Results
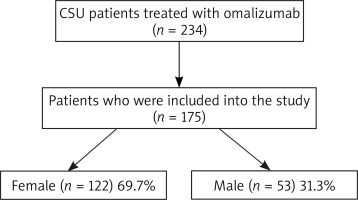
In all, 234 patients with CSU were evaluated. Fifty-nine patients were excluded from the study due to missing clinical records and finally 175 CSU patients were included in the study (Figure 1). One hundred and twenty-two patients were female (69.7%), and 53 were male (31.3%). The mean age was 42.0 ±15.2 (range: 14–85 years). A history of urticaria was documented in first-degree relatives in 9.1% of the patients. Hypertension, the most frequent comorbidity, was present in 12.5% of patients, diabetes mellitus in 10.2%, history of atopy in 6.8%, and Hashimoto’s thyroiditis in 4.5%. These were the most common co-morbidities. Other co-morbidities included vitiligo, multiple sclerosis, chronic sinusitis, gastroesophageal reflux, hyperlipidaemia, anxiety disorder, hereditary spherocytosis, and thalassemia carrier status.
In the comparison using Kruskal-Wallis analysis of the most common comorbid diseases (hypertension, diabetes mellitus, atopy, and Hashimoto’s thyroiditis) and the treatment responses, we did not find a comorbidity-specific difference between the initial, 1st month, and 6th month UAS7 scores (initial p = 0.284, 1st month p = 0.671, 6th month p = 0.431).
Initial serum concentrations of total IgE were less than 100 in 47.4% of the patients and greater than 100 in 52.6% of the patients. The correlation between IgE levels and treatment response rates were evaluated based on the UAS7 scores. We did not find any statistically significant correlation between the parameters (initial p = 0.520, 1st month p = 0.931, 6th month p = 0.343). We did not find any statistically significant correlation between the parameters in terms of the relationship between age and treatment response rates (initial p = 0.919, 1st month p = 0.838, 6th month p = 0.855). Also, there was no statistical correlation between the parameters of sex and disease activity (initial p = 0.532, 1st month p = 0.605, 6th month p = 0.891).
Disease duration was < 1 year in 63 (36%) patients, 1–5 years in 76 (43.4%) patients, 5–10 years in 22 (12.6%) patients, and ≥ 10 years in 14 (8%) patients. There was no statistically significant correlation between disease duration and disease activity scores (initial p = 0.401, 1st month p = 0.558, 6th month p = 0.194). The treatment schedule with omalizumab was 300 mg/month for each patient. The mean treatment period was 20 months (range: 7–60 months). The mean initial UAS7 score was 36.1 ±7.3 (range: 14–42), which was calculated as 7.3 ±9.9 (range: 0–42) at the time of the third (UAS7 3th month) injection and 3.6 ±6.6 (range: 0–35) at the sixth (UAS7 6th month) injection. The decreasing trend of UAS7 was statistically significant (p < 0.001). When the patients were classified as well-controlled CSU, mild CSU, moderate CSU, and severe CSU patients according to the UAS7 scores, 89.7% of the patients were severe CSU patients at the beginning of the study, while 93.7% of the patients were well-controlled and mild CSU patients at the 6th month (Table 1).
Table 1
Disease activity scores at different periods of the disease
Of 69.7% of the patients treated with 300 mg omalizumab every 4 weeks (standard treatment protocol), 26.6% of them were treated with the same dosage once every 8 weeks, and the remainder (7%) were treated once every 2 weeks. Two (1.14%) of the patients discontinued omalizumab treatment due to unresponsiveness. Although stated as not regularly, 30.2% of the patients treated with omalizumab still needed antihistamine treatment (53 in 175 patients). Duration of antihistamine use was 3–5 days at the end of the omalizumab treatment time period. Omalizumab treatment was stopped in 10.2% of the patients due to their well-controlled disease after an average of 14 injections. The mean disease-free follow-up period was 3 months (range: 1–25 months). According to our clinical experience, the patients with recurring disease could be quickly controlled after 3 to 6 injections. In a patient who became pregnant after the third injection, omalizumab treatment was changed from once every 4 weeks to once every 8 weeks. With this treatment protocol, successful disease control was achieved during pregnancy and breastfeeding.
Discussion
In cases of chronic spontaneous urticaria resistant to H1 antihistamines, it is recommended to stop antihistamine treatment by reducing the dose 4 weeks after starting omalizumab treatment. If the UAS7 response is above 90% at the end of the first 4 weeks, a complete response is accepted and it is recommended to discontinue antihistamine treatment. If the UAS7 response is 50–90% (partial response), it is recommended to reduce the antihistamine to the lowest effective dose, and to maintain the current antihistamine dose with a UAS7 response less than 50% (limited response or no response) [6]. Studies conducted all over the world emphasize the effectiveness of omalizumab treatment. The complete response rate is reported as 67.9% in the literature [7]. In the present study group, this rate was found to be 70.9%.
When publications on the effectiveness of omalizumab treatment are examined, some studies show that there may be a number of factors that affect the treatment response, but there are also publications showing that the examined parameters have no effect on the treatment response. Ghazanfar et al. reported that the absence of angioedema, a negative histamine release test, older age, short-term disease history, and no history of immunosuppressant use were favourable factors for a positive response to omalizumab [8]. Ertas et al. emphasized that measuring IgE levels before and after treatment could be useful for determining the treatment response. In addition, Marzano et al. also agree with the idea that high IgE levels affect treatment response positively [9, 10]. In contrast, Bulur et al. reported no significant association between treatment response and demographic data, disease-related parameters, or laboratory data 6 months after treatment with omalizumab [11]. In our study, we did not find any clinical or laboratory factors predicting response to omalizumab treatment. Improvement in disease control seemed to be independent of age, sex, disease duration, disease severity (initial UAS7), and pretreatment IgE levels.
Upper respiratory tract infections, headaches, and skin and subcutaneous tissue disorders are reported as the most common side effects of omalizumab treatment [12, 13]. A clinical study reported increased headache, arthralgia, and cough in patients receiving 300 mg omalizumab compared with those receiving placebo [14]. Bulur et al. reported that 89.4% of omalizumab-treated patients experienced no side effects, with nausea in 13 patients and myalgia in 1 patient and they stated that this may have been due to relief of urticarial symptoms and underrating of non-specific complaints [11]. Minor complications stated in the literature were observed in 12% of our patients. The very rare but most dramatic side effect reported in the literature was anaphylaxis with a rate of 0.09% [15]. It has been reported that this complication may occur by inducing both skin inflammation and anaphylaxis through the binding of immune complexes formed between omalizumab and IgE to the high-affinity IgE receptor (FcR) [16]. In the present study group, one of the patients experienced dyspnoea after the first injection of omalizumab. Although the patient did not meet the criteria of anaphylaxis, we did not continue omalizumab treatment in that patient. The minor complication rate in the present study is compatible with the literature. However, due to the case with a complaint of dyspnoea in this small patient group, we remind practitioners to be alert for this complication while using this drug.
Conclusions
The findings show that omalizumab is effective and safe for treatment of CSU with a dosing of 300 mg/month. We observed no relationship between omalizumab treatment efficacy and patient characteristics or laboratory parameters. Antihistamines were still used in one third of this patient group.