Introduction
Chronic urticaria is an etiopathologically complex disease process in which chronic spontaneous urticaria (CSU) and chronic inducible urticaria are distinguished [1–3].
Although about 2% of the European population suffers from this disease, its etiopathogenesis still remains unknown [1, 3, 4]. There are some works indicating a significant participation of vitamin D in the etiopathogenesis of urticaria. Furthermore, in 40–50% of patients with CSU on the basis of the positive result of the autologous serum skin test (ASST), an autoimmunological background of the disease is diagnosed [5, 6]. Moreover, numerous test results confirm involvement of the coagulation system/fibrinolysis and non-infectious inflammatory factor in the pathomechanism of CSU [1, 7–14].
Aim
The aim of the study was to determine whether C-reactive protein (CRP), vitamin D, coagulation and fibrinolysis pathway as well as mean platelet volume (MPV) may play a role in pathogenesis and contribute to the severity of chronic spontaneous urticaria.
Material and methods
One hundred and forty-two patients (including 108 women and 34 men) with CSU in the active phase at the age from 19 to 87 were qualified for the study. CSU diagnosis was based on the medical history and clinical picture of skin lesions. The active phase of the urticaria was defined as the presence of more than 5 hives that occurred within 7 days before the beginning of the study. What is more, all study participants were instructed not to use any oral antihistamines, corticosteroids or antithrombotic therapy for at least 2 weeks before the beginning of the study. In each patient, on the basis of the results of consultations (dental, laryngological, urological and gynaecological), the presence of latent inflammatory foci was excluded. On the basis of the imaging tests, thromboembolism of the lower limbs was excluded in patients with higher values of D-dimers.
The activity of urticaria was assessed using the UAS-7 scale, assigning the obtained results to the following 4 groups: urticaria with a minimal course 1–6, mild 7–15, medium 16–27 and severe 28–42, according to the guidelines of the EAACI/GA(2)LEN/EDF/WAO [15, 16]. ASST test was performed in all study participants. A venous blood sample from cubital fossa was taken for analysis of the following variables: complete blood count, electrolytes (Na, K), CRP, alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatinine, γ-glutamyl transferase (GGTP), glomerular filtration rate (GFR), vitamin D, D-dimer and fibrinogen levels.
Results
During the performed study, the average UAS-7 value in 142 subjects was 19.06.
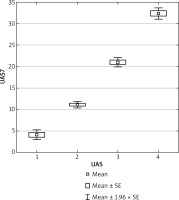
The mean values of UAS-7 in particular groups were distributed in the following way: group 1 – 4.11; group 2 – 11.11; group 3 – 21.02; and group 4 – 32.36 (Figure 1).
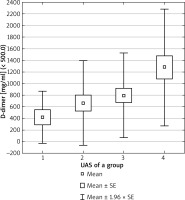
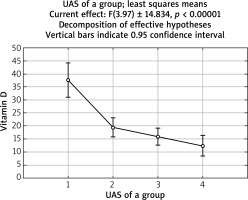
In the examined group, a statistically significant difference between groups with various activity of urticaria (defined as UAS-7 value) in D-dimer concentration average values (p < 0.05) was observed (Figure 2). We did not observe any statistically significant correlation between activity of urticaria (UAS-7 value) and fibrinogen as well as D-dimer concentration. However, a statistically significant negative correlation between activity of urticaria and vitamin D concentration (p < 0.001) was noted (Figure 3).
Ninety-two (65%) study participants had positive ASST tests and 50 (35%) were ASST negative tested.
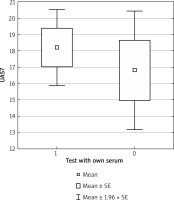
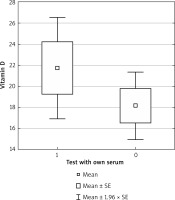
In the individuals with a positive ASST test, a statistically non-significant value of the UAS-7 scale (p = 0.58) and statistically non-significant higher vitamin D concentration (p = 0.305) were found as compared to patients with a negative ASST result (Figures 4 and 5).
No significant correlation between the mean MPV values (p = 0.88816) and the PLT number (p = 0.935136) as well as the UAS-7 value in the examined groups was confirmed.
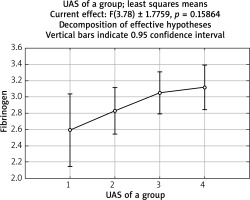
In the examined material, no statistically significant increase (p = 0.16) in fibrinogen concentration with the increased activity of urticaria was observed (Figure 6).
There was no statistically significant correlation between CRP concentration and activity of urticaria (p = 0.14).
Discussion
Our aim was to determine whether some factors may play a role in pathogenesis as well as contribute to the severity of CSU. We have noticed a statistically significant negative relationship between the urticaria active score (UAS) and serum level of vitamin D. We have observed the lowest serum level of vitamin D in patients from group 4 (with the highest UAS), which is consistent with the recent studies [17–20]. However, Thorp et al. have not observed a marked association between activity of urticaria and vitamin D serum level. On the basis of analysis of connections between D-dimer, fibrinogen concentration and UAS we have shown a significant difference between groups with various activity of urticaria in D-dimer concentration average values. Moreover, we have observed a positive correlation between urticaria active score and fibrinogen as well as D-dimer serum level. Nonetheless, statistically significant relationships were not observed. Elevated D-dimer and fibrinogen plasma levels in patients with urticaria suggest activation of coagulation and fibrinolysis cascade. Our results are in line with other studies published recently [14, 21–23]. It is well-established that an increased fibrinogen and D-dimer concentration without concomitant thrombosis is, as it has been observed in our present study, connected with activation of the coagulation and fibrinolysis pathway. Such a condition is activated by chronic endothelial vasculitis during the active phase of urticaria and seems to be noteworthy [1, 14]. Taking into consideration previous studies, activation of the coagulation cascade resulting in the generation of thrombin, subsequently activates anti-coagulation and fibrinolysis. Thrombin and other proteins may play a key role in mast cell degranulation and result in urticaria. The exact mechanism how the coagulation, anti-coagulation, fibrinolysis and complement systems act in CSU pathogenesis is required to be expanded further in future studies [21–23].
Our results regarding a possible relationship between an inflammation biomarker like CRP and UAS are partly consistent with the recent data [14, 15, 21, 24, 25]. Additionally, in our study we have noticed that CRP tends to have higher values (without any inflammation points in routine imaging like abdominal ultrasonography and chest X-ray) in the relation to the severity of urticaria in groups 1–3 of UAS score. The interpretation of such a finding has been a subject of the debate in many studies published recently, and might be connected with an increased activity of mast cells in urticaria [8, 21]. However, our study might indicate that CRP increase without any present inflammation might be a symptom of active endothelial vasculitis with the forced permeability [14]. It is interesting to note that among patients with a severe form of urticaria we have found an increased fibrinogen and D-dimer concentration concomitant with the decrease of CRP values. This outcome seems to be worth considering. It might indicate that in pathogenesis of the severe form of urticaria, coagulation and fibrinolysis pathway play a greater role than inflammatory disorder like endothelial vasculitis [14, 26, 27]. According to the available data, chronic spontaneous urticaria has an autoimmunological character in over 40–50% of cases [5, 6]. In the present study we have analysed vitamin D concentration and UAS among patients with both negative and positive ASST, which is known as an indicator of chronic autoimmune urticaria. Interestingly, we have shown that the vitamin D serum level was not statistically significantly higher among ASST positive patients in a comparison with ASST negative patients. This result is not consistent with previous data of other authors where there was no relationship between the parameters or, on the other hand, a statistically significant association was observed [17, 28]. ASST positive patients presented a more severe form of urticaria as they had higher UAS-7 values than ASST negative patients. That observation is in line with previously described results [21, 29, 30].
Our results could not confirm the theory that patients with a low vitamin D serum level are predisposed to the development of autoimmune urticaria. However, a series of studies previously reported reduction of urticaria severity due to vitamin D supplementation among ASST positive patients. These results might directly support the involvement of vitamin D in the immune system [17, 31–33]. Interestingly, Chen et al. in their study showed that vitamin D inhibits plasma B cells differentiation which also results in suppressing of antibodies secretion [18]. This effect seems to be depending on the number of receptors for vitamin D on lymphocytes B, and, what is more, might be concomitant with an increased activity of previously existing lymphocytes B. This condition might clarify our finding regarding a lower vitamin D level among patients with positive ASST in comparison with ASST negative patients. Furthermore, we have analysed the association between urticaria activity and MPV. Obtained results were not statistically significant corresponding with outcomes of Nishimura et al. [34]. However, in some previous studies this relationship was established, which leaves that matter intriguing [35, 36].
We also have assayed a possible connection between platelet count and severity of urticaria, but no results worth considering were found. In conclusion, in the present study we have observed a statistically significant correlation between urticaria activity and vitamin D as well as a statistically significant difference between groups with various activity of urticaria in D-dimer concentration average values. These results might directly support the possible involvement of both coagulation and fibrinolysis pathway and vitamin D in the urticaria pathomechanism and open new perspectives for a better understanding of the pathogenesis as well as the treatment of the disease. Moreover, our study may in some measure suggest that an increased D-dimer level in patients with severe urticaria should be considered not only as an indicator of its activity. A series of studies previously showed that the elevated D-dimer serum concentration may be regarded as a useful biomarker of worse response to standard treatment by antihistaminic drugs or cyclosporines [7, 37]. Unquestionably, further prospective studies with evaluation of response to the standard treatment in patients with an increased D-dimer level in larger populations conducted at multiple centres are required to expand further our findings.