Introduction
The bronchodilator reversibility test is one of the most common diagnostic tests performed in diseases with symptomatic dyspnoea. The test is also a recommended method for determining the reversibility of obstruction in asthma and severity classification of chronic obstructive pulmonary disease (COPD). The reversibility test measures an increase in forced expiratory volume in 1 s (FEV1) and forced vital capacity (FVC) parameters after the administration of a bronchodilator (short-acting β-agonist – SABA). The dosage of 400 mg of a SABA, such as salbutamol or fenoterol, is recommended by the GOLD, the GINA, and the ATS/ERS guidelines [1, 2]. A standard dosage, however, applied as a rescue medicine in bronchospastic dyspnoea is 100–200 mg.
Numerous studies have shown a significant chronotropic and dromotropic influence of SABA on the heart. Insulander et al. found that salbutamol infusion caused significant electrophysiological changes at all levels of the conduction system of the heart, which was most evident in the atrioventricular (AV) node [3]. It is worth noting that even in healthy adults, inhalation of 200 mg of salbutamol causes a significantly faster heart rate and a decline in peripheral vascular resistance [4]. Burggraaf et al. [5] demonstrated that in subjects suffering from hypoxemia, salbutamol inhalation not only caused a faster heart rate but also triggered vasodilation, which could lead to pulmonary shunt and decrease in diastolic pressure [5].
Previous studies have reported the frequent incidence of unstable angina pectoris and myocardial infarction (MI) in subjects taking both short- and long-acting β-adrenergic medicines [6–9]. Lee et al. [6] found that the estimated risk of myocardial infarction among new users of β-adrenergics was significantly elevated (OR = 3.2) especially in those with cardiovascular disease (OR = 7.3) compared with subjects who had never received a β-agonist, which can be partially attributed to the impact of β-adrenergic drugs on heart rate and the occurrence of arrhythmias [6].
The reports of increased risk of acute coronary syndromes in subjects inhaling β-adrenergic drugs [6–8] raise concerns about the safe use of 400-µg short-acting, β-adrenergic drugs in the bronchodilator test in elderly subjects. Until now, the risk of significant tachyarrhythmias that can lead to MI with the use of such a dosage of SABA in elderly subjects with concomitant heart diseases has not been established.
Aim
The primary goal of this study was to examine the relationship between inhaled β-agonist used in the bronchodilator test in older subjects and the risk of cardiovascular events including tachyarrhythmia.
Material and methods
Study population
For this study, we recruited 53 non-smoking subjects (45 women) aged 77; 68–82 who were admitted to the geriatric department of Wroclaw Medical University. The subjects were hospitalised during 2017–2019 for a comprehensive geriatric assessment. They were all in a stable clinical state. Subjects suffering from respiratory diseases and/or taking inhalation drugs were excluded from the study. Assessment of medical history, physical examinations, blood biochemistry, chest X-ray, 12-lead electrocardiogram, 24-hour Holter monitoring, bronchodilator test, and echocardiography were performed.
The detailed characteristics of the participants are presented in Table 1.
Table 1
Demographic data
The research was approved by the Bioethical Committee at Wroclaw Medical University (reference number 417/2016). All subjects provided written informed consent.
Bronchodilator test
The bronchodilator test was conducted between 11 and 12 a.m. on the same day when Holter monitoring began. Before and during the test, the subjects did not perform any physical activities and did not smoke cigarettes.
Spirometry was performed before and 15 min after fenoterol inhalation using a Master Scope spirometer (Erich Jaeger GmbH, Wurzburg, Germany) according to the recommendations of ATS/ERS [2]. Particular emphasis was placed on the test repeatability and acceptability criteria [2]. In the test, a dose of 400 µg of fenoterol (Berotec made by Boehringer Ingelheim) was administered (4 puffs of 100 µg) by an MDI inhaler under the supervision of the certified staff. The GLI2012 reference values were used to interpret the results. The bronchodilator test results were presented in percentages of the predicted and initial values.
24-hour Holter ECG monitoring
24-hour Holter ECG monitoring was performed using a 12-lead device produced by Lifecard CF (Reynolds Medical, Hertford UK). The obtained data were processed using specific software, and the reports were prepared by an experienced cardiologist. In this study, we compared the heart rate and the amount of ventricular and supraventricular extrasystoles (VE and SVE) an hour before and after the bronchodilator test.
Echocardiography examination
We used a two-dimensional, M-mode cardiovascular ultrasound system (Siemens Acuson X300). The examination was conducted in the left lateral decubitus position. All echocardiographic examinations were performed by an experienced cardiologist.
Statistical analysis
The data analysis was undertaken using Statistica 13 software (StatSoft). The Shapiro-Wilk test was performed to investigate the normal distribution of the sample. As the studied parameters were not normally distributed, non-parametric tests were used. The data were expressed as median (Me) and the interquartile range 25–75%. The Kolmogorov-Smirnov or Friedman’s ANOVA test was performed to compare two independent samples. For dependent samples, the Wilcoxon or McNemar test was performed. The correlation between the variables was analysed by means of the Spearman test. A value of p < 0.05 was considered as statistically significant for all tests.
Results
A group of 53 subjects (45 women) aged 77: 68–82 took part in the study. The subjects had not suffered from any respiratory tract diseases and had not used any inhalation medicines, including β-adrenergic drugs. The most common health problems in that population were hypertension (83% of the subjects) and coronary artery disease (CAD) (36%). In addition, 41% of the subjects were stroke survivors (Table 2). A total of 36% of the subjects used β-blockers and 13% used calcium channel blockers (Table 2).
Table 2
Patients’ comorbidity and medicines used
Echocardiography
The echocardiography revealed a normal heart ejection fraction (EF) in all the study subjects (median 65; 60–65%). Diastolic dysfunction characteristics were found in 71% of the subjects. The majority of the subjects had confirmed heart valve dysfunctions. The most commonly occurring dysfunctions were mitral valve insufficiency (84%) and tricuspid valve insufficiency (81%). The other parameters are shown in Table 3.
Table 3
Heart ultrasound examination results
Spirometry
The initial (before inhalation) spirometry showed a moderately severe obstruction in only 1 patient (FEV1 – 50% of predicted value). The other subjects did not have any pulmonary dysfunction. FEV1 median was 97% (85–106%) of the predicted value and FVC was 104% (68–149%) of the predicted value. After the drug inhalation, FEV1 improved by 5% (–17%; 22%), as compared to the initial value (Table 4).
24-hour Holter electrocardiographic monitoring
The Holter ECG monitoring readings taken an hour before and after the bronchodilator test were compared. In 6 (11%) subjects, atrial fibrillation was found, while the other subjects had the sinus rhythm. A slight but statistically significant increase in heart rate from 71/min to 75/min (median) was noted after the administration of fenoterol.
In 39% of the subjects, premature ventricular beats before drug administration were found. After fenoterol inhalation, such an abnormality occurred in 32% of the subjects (p < 0.001). Premature supraventricular beats were found in 72% of the subjects before the bronchodilator reversibility test, and after drug inhalation (p = 0.01) in 79%. However, when the number of ventricular and supraventricular beats during those periods was compared, no statistically significant differences were found (p = 0.42) (Table 5).
Table 5
Frequency of occurrence of additional ventricular and supraventricular beats in patients (N = 53)
Because 36% of the studied population was treated by β-blockers, which would influence the final results, we analysed them separately. Table 5 shows that the β-blocker group had a lower heart rate before (p < 0.001) and after fenoterol inhalation (p < 0.001) compared with the non-treated subjects. We found a significant increase in heart rate (p = 0.009) and the number of subjects with supraventricular beats in the β-blocker group (p = 0.02). No potentially dangerous arrhythmias or signs of CAD were found in the subjects.
In the non-treated group of subjects, we found a statistically significant increase in the number of subjects with supraventricular beats (p < 0.001) and a larger increase in heart rate after the inhalation of fenoterol (p = 0.04) than we observed in β-blocker-treated subjects (Figure 1).
Figure 1
Heart rate before and after inhalation of fenoterol in patients treated (A) and not treated (B) with β-blockers
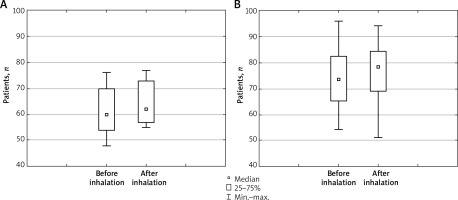
No statistically significant impact of the SABA on the occurrence of tachycardia or other arrhythmias was found.
Discussion
The aim of our study was to investigate whether 400 µg of fenoterol, used in the bronchodilator test, would trigger an increase in heart rate and the development of tachyarrhythmias that could contribute to acute coronary syndrome in elderly subjects.
Until now, much attention has been focused on the effects of SABA on the heart conduction system and peripheral vascular system [4, 10–12]. Numerous studies have shown a noticeable chronotropic effect of inhaled and intravenous drugs in the healthy population [10, 12, 13], young people suffering from asthma [2, 14], and children [11, 15] treated for the exacerbation of asthma or COPD [16–19]. Lee et al. found an increased risk of tachyarrhythmias in subjects using long-acting β-adrenergic and parasympatholytic drugs [20]. Tachycardia was caused not only by the direct effect on β-adrenergic receptors of the heart but also by indirect activation of receptors causing vessels’ dilation and reaction of the vagus nerve [12, 21]. This effect is linked to a greater risk of an acute cardiovascular incident in the population of patients treated with inhalation drugs, especially at the beginning of a therapy [6, 7, 9].
Because of these findings, a dosage of 400 mg of salbutamol or fenoterol used in the bronchodilator test in elderly subjects suffering from circulatory diseases and particularly CAD raises concerns. We conducted the β-adrenergic test in elderly subjects without respiratory tract diseases who had not used β-adrenergic drugs before. In this study, using 24-hour Holter ECG monitoring, we found that a slight post-inhalator increase in heart rate (p = 0.006) did not trigger the exacerbation of electrocardiographic signs of heart ischemia. Compared to baseline after the inhalation of fenoterol, no significant increase in the number of ventricular or supraventricular extrasystoles was observed in these subjects. However, after the drug inhalation, supraventricular extrasystoles occurred more often and ventricular premature beats less often in the studied population. No potentially dangerous arrhythmias were found among the examined subjects. However, we observed a significant increase in heart rate and the number of supraventricular beats in the non-β-blocker-treated group. Therefore, it can be concluded that using 400 mg of the short-acting β-adrenergic drug in elderly subjects is safe, but it should be performed with caution.
This study has some limitations. The group consisted of elderly subjects in a good general condition, and none of the subjects showed signs of heart failure, electrolyte imbalance, or respiratory impairments. The studies that have shown an increased risk of acute coronary syndrome [6, 7, 9] investigated populations with confirmed obstructive lung disease (asthma or COPD). This means that apart from the drug itself, other factors, such as exacerbation of the underlying disease, hypoxemia, electrolyte imbalance and other comorbidities, had an additional effect on the studied subjects. Future studies on the current topic are, therefore, recommended.
Conclusions
We observed only a slight but statistically significant increase in heart rate in subjects after the administration of 400 µg of inhaled fenoterol. However, there was a significant increase in the number of supraventricular beats in the subjects who had not taken β-blockers.
The study has shown that the older subjects did not experience potentially dangerous tachyarrhythmias after the administration of fenoterol. In subjects not treated by β-blockers or with concomitant serious heart disease, the bronchodilator test should be performed with caution.









