INTRODUCTION
Generalized pustular psoriasis (GPP) is a rare variant of pustular psoriasis. It was first described in 1910 by von Zumbusch. It is characterized by acute, subacute and chronic episodes of erythematous lesions with overlying sterile pustules. Global estimates of GPP prevalence vary considerably between countries, ranging from 1.76 to 124 patients per million population [1]. GPP flares are commonly triggered by medications, infections, trauma, or surgical procedures [2]. GPP flares are frequently accompanied by systemic symptoms, including fever, malaise and leucocytosis and may lead to hospitalization and life-threatening complications [1]. Treatments for GPP include various therapies, most of which are approved for other indications and used off-label for GPP. They can be broadly classified into three categories: biologics, non-biologic systemic therapies and other therapies (Table 1) [2, 3]. However, their use is based on current treatments for plaque psoriasis, and there is limited evidence for GPP-specific efficacy of anti-psoriatic drugs, including biologics [1].
Table 1
Therapies used in generalized pustular psoriasis
| Biologics | Non-biologic systemic therapies | Other therapies |
|---|---|---|
A minority of GPP cases are caused by rare and damaging mutations in the IL-36RN gene, which encodes an anti-inflammatory protein known as the interleukin 36 receptor antagonist [4]. Spesolimab, a first-in-class monoclonal antibody targeting the interleukin-36 receptor, is currently approved for the treatment of acute GPP flares by both the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) [5].
OBJECTIVE
This study is aimed to describe the case of a patient with inflammation of the salivary glands occurring as a potential adverse effect of acitretin therapy for GPP.
CASE REPORT
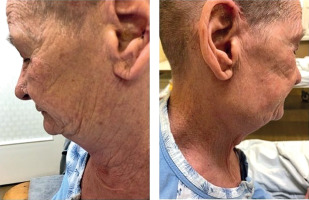
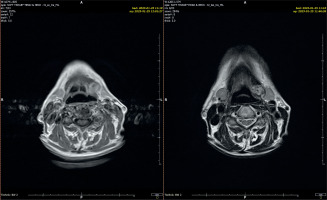
A 72-year-old female patient was admitted to the Dermatology Department due to sudden onset of widespread rash and the eruption of primary, sterile, macroscopically visible pustules on non-acral skin following a recent neurosurgical procedure. The patient’s condition was complicated by elevated inflammatory markers (white blood cells (WBC) = 12.79 × 10³/µl, C-reactive protein (CRP) = 27.7 mg/dl) and decreased levels of both total (7.96 mg/dl) and ionized calcium (0.87 mmol/l), as revealed by laboratory tests. The patient had a history of generalized pustular psoriasis with periods of remission and exacerbation. Based on the clinical presentation of the skin lesions, medical history, additional test results, and the diagnostic criteria established in 2017 by the European Rare and Severe Psoriasis Expert Network (ERASPEN) (which include primary sterile pustules visible on non-acral skin, systemic inflammation, and a recurrent disease course), an exacerbation of GPP was diagnosed. Given the severity and progression of the skin lesions, as well as the patient’s overall deteriorated condition, a comprehensive treatment approach was initiated. Based on the clinical status, drug availability, and expert recommendations, treatment with acitretin and glucocorticosteroids was introduced. Acitretin was administered at an initial dose of 20 mg/day (0.5 mg/kg), and methylprednisolone at 8 mg/day. A rapid and favorable clinical response was observed, with marked reduction of pustular lesions. However, on the fifth day of pharmacotherapy, the patient developed notable bilateral swelling in the submandibular and preauricular regions (Figure 1). This swelling was firm and accompanied by a mild increase in inflammatory markers (WBC = 15.95 × 10³/µl, CRP = 5.1 mg/dl). An ultrasound examination confirmed inflammation in all salivary glands, with no evidence of enlarged lymph nodes in cervical regions. In response to these findings, an otolaryngology consultation was obtained. Common inflammatory and postoperative infectious causes, including mumps, were excluded, and serologic testing yielded negative results. Additional serologic tests were performed for cytomegalovirus, Epstein-Barr virus, and bacterial pathogens; all results were negative. Autoimmune markers, including ANA and anti-SSA/SSB antibodies, were negative, thereby excluding Sjögren’s syndrome and other related connective tissue diseases. Throughout the hospitalization, the patient received optimal intravenous and oral fluid therapy. Considering the clinical presentation, the findings of the otolaryngology consultation, and a comprehensive review of the patient’s medications, an adverse reaction to acitretin was suspected. Given the diagnosis, acitretin was discontinued, while glucocorticosteroid therapy was continued. This adjustment led to the resolution of the swelling and other symptoms. A follow-up ultrasound examination conducted 5 days after acitretin discontinuation revealed that the inflammation in the salivary glands had completely subsided. Magnetic resonance imaging performed 7 days after the episode revealed symmetrical parotid and submandibular glands without focal lesions (Figure 2). To further the diagnostics, a salivary gland biopsy was performed, yielding a histopathological result of groups and clusters of salivary gland cells with a monomorphic appearance in acinar arrangements. No nuclear atypia was observed. Numerous macrophages and microcalcifications were present in the background. The cytological findings did not support a diagnosis of neoplastic proliferation.
DISCUSSION
Pustular psoriasis is a rare form of psoriasis, which represents approximately 1% of all clinical cases of psoriasis. It is an immune-mediated systemic skin disorder, characterized by sterile, neutrophil-rich pustules with a variety of distribution patterns. Pustular psoriasis can be further divided into specific subtypes based on the clinical presentation and location of the pustules; it can be classified as either localized, including palmoplantar pustulosis (PPP) and acrodermatitis continua of Hallopeau, or generalized, including GPP, pustular psoriasis of pregnancy, and infantile/juvenile pustular psoriasis [6]. According to the diagnostic criteria established in 2017 by ERASPEN, GPP is defined as primary, sterile, macroscopically visible pustules on non-acral skin (excluding cases where pustulation is restricted to psoriatic plaques). GPP can occur with or without systemic inflammation, with or without psoriasis vulgaris. GPP should only be diagnosed when the condition has relapsed at least once or when it persists for more than 3 months. Furthermore, in its consensus, ERASPEN proposed a classification of clinical phenotypes of pustular psoriasis (Table 2) [4]. The Japanese Dermatological Association (JDA) published diagnostic criteria for GPP in 2018, based on four factors: (1) systemic symptoms such as fever or fatigue; (2) systemic or extensive flush accompanied by multiple sterile pustules; (3) histological evidence of neutrophilic subcorneal pustules, characterized by Kogoj’s spongiform pustules; and (4) recurrence of these clinical and histological findings. In Japan, a definitive diagnosis of GPP can be made in patients who meet all four criteria, and GPP should be suspected in those who meet criteria (2) and (3) [7]. Flare-ups of GPP can be triggered by several factors [8]. In the case of the patient described, the neurosurgical procedure was likely the triggering factor. Current treatment options for GPP can be classified into biologic and non-biologic systemic agents. Based on the Japanese guidelines for the management of GPP and the Medical Board of the National Psoriasis Foundation, the most commonly used treatments for patients with GPP are retinoids (including acitretin), cyclosporine, and methotrexate [4]. GPP is distinct from plaque psoriasis, characterized by unique genetic loci and inflammatory cytokines, with IL-1 and, particularly IL-36 being predominant [9]. Since April 2024, spesolimab has been approved for the treatment of GPP flares in adults in key regions such as the USA, Japan, China and the EU and was recently approved for expanded indications in the USA and China [5]. Retinoids are biologically active derivatives of vitamin A. They modulate cellular proliferation and differentiation, normalize keratinization and cell adhesion, exert anti-acne and antiseborrhoeic activity, and display immunomodulatory and anti-inflammatory effects. In addition, they induce apoptosis, regulate extracellular matrix components, and contribute to tumour chemoprevention and therapy.
Table 2
Consensus definition for the diagnosis of pustular psoriasis
They are typically classified into three generations. First-generation retinoids are non-aromatic naturally occurring compounds. These include retinol, retinal, isotretinoin (13-cis-retinoic acid), tretinoin (all-transretinoic acid), and alitretinoin (9-cis-retinoic acid) among others. Second-generation retinoids include etretinate, acitretin, and motretinate. Their structures make them more lipophilic with a higher bioavailability than first-generation retinoids. Third-generation retinoids include adapalene, tazarotene, and bexarotene which are polyaromatic molecules (Table 3) [10]. Acitretin is a second-generation mono-aromatic retinoid, the major metabolite and pharmacologically active form of etretinate. Acitretin’s mechanism of action is still not clearly understood. Despite its low binding affinity, it paradoxically activates all three RAR subtypes. Evidence supports a normalization of differentiation and proliferation as well as a modification of inflammatory responses and neutrophil function [11]. It is approved by the Food and Drug Administration for the treatment of severe, pustular, generalized, and localized plaque psoriasis. It is also used for the management of conditions such as Darier’s disease, Grover’s disease, pityriasis rubra pilaris, lamellar ichthyosis, lichen planus, and lupus erythematosus [12]. The most common adverse reactions during acitretin treatment are mucocutaneous effects [10]. Acute adverse mucocutaneous effects of systemic retinoids include xerosis with pruritus, cheilitis, dry mucosa: mouth, nose (epistaxis), eyes, skin fragility, retinoid dermatitis, palmoplantar peeling, photosensitivity, sticky sensation of the palms and soles, facial swelling, granulation tissue with pyogenic granuloma-like lesions (e.g. at sites of acne cysts, periungual), intertriginous erosions with exuberant granulation tissue, nail fragility with softening, onycholysis, paronychia, Staphylococcus aureus infection, telogen effluvium, hair thinning [10, 11]. The patient developed symptoms of sialadenitis on the fifth day of acitretin therapy, suggesting a temporal association with the medication. Otolaryngological evaluation excluded local structural and inflammatory causes. Infectious etiologies, including mumps, were ruled out based on appropriate laboratory testing. After reviewing the literature available in the PubMed database. A review of the literature available in the PubMed database did not identify any previous reports of salivary gland inflammation during acitretin therapy. To the best of current knowledge, this appears to be the first reported case of salivary gland inflammation associated with acitretin use. Complete clinical and radiological remission of symptoms occurred after discontinuation of the drug. The pathophysiological mechanism in this context is unclear. The most likely mechanism involves obstruction of the salivary gland ducts secondary to the exfoliative effects of acitretin [13]. Another possible mechanism involves impaired salivary secretion induced by retinoids. In the article ”Drug effects on salivary glands: dry mouth”, Scully in 2003 noted that systemic retinoids are well known to cause xerostomia and alterations of the oral and labial mucosa, as reported for etretinate (Wishart et al., 1981) and 13-cis-retinoic acid (Hennes et al., 1984), although robust supporting data remain limited. However, a study evaluating salivary variables during 3 months of oral isotretinoin therapy demonstrated a significantly lower mean flow rate of stimulated saliva during the period of medication than at baseline [14]. An alternative explanation may be increased salivary viscosity resulting from acute erythroderma secondary to a GPP flare [15]. Moreover, calcium deficiency may lead to impaired saliva secretion. The study highlights that Transient Receptor Potential Vanilloid 4 (TRPV4), a channel involved in calcium regulation, interacts with anoctamin 1 (ANO1) to modulate fluid secretion in salivary glands. Disruptions in calcium signalling due to insufficient calcium levels could adversely affect saliva secretion by altering ion channel function [14, 16]. To summarize, the most likely mechanism appears to involve several events: obstruction of the salivary ducts due to the exfoliating and drying effects of acitretin [13, 14], extensive and severe psoriatic erythroderma resulting in significant transcutaneous water and calcium deficiency leading to reduced secretion of proteins and proteoglycans by the secretory parts of the salivary glands via adrenergic pathways [16, 17]. Based on the available evidence and the clinical course, the pathophysiological mechanism of salivary gland inflammation in this patient appears to be multifactorial. Nevertheless, an attempt can be made to rank the possible causes by the likelihood of the following listed below.
Most likely cause: A direct adverse effect of acitretin, which likely contributed to xerogenic changes, exfoliation, and obstruction of salivary ducts, consistent with known mucosal side effects of systemic retinoids [13, 14].
Contributing factors: Systemic dehydration associated with GPP-related erythroderma [15] and calcium deficiency [16, 17] may have exacerbated glandular dysfunction by impairing secretion or altering saliva composition.
Less likely causes were excluded. Postoperative complications were ruled out based on otolaryngological evaluation. Infectious etiologies, including CMV, EBV, mumps, and bacterial infections, as well as autoimmune disorders (e.g., Sjögren’s syndrome), were excluded on the basis of appropriate laboratory and serologic testing.
CONCLUSIONS
This is the first reported case of salivary gland inflammation potentially associated with acitretin therapy in a patient with GPP. Despite the widespread use of acitretin in dermatology, no similar cases have been reported in the literature so far. It remains unclear whether this represents a direct complication of acitretin therapy or the result of multiple contributing factors. Further studies and clinical observations are required to clarify the underlying mechanism of this potentially underrecognized adverse effect.