Introduction
Androgenetic alopecia is the most common type of non-cicatricial hair loss both in male and female patients [1]. The pathomechanism of the disease involves chronic and progressive miniaturization of hair follicles, which leads to the conversion of terminal hair to vellus hair [2]. Patients who remain without treatment manifest a continuous progression of the disease. The incidence of the condition goes up with age. In women before the age of 30, female pattern hair loss (FPHL) is present in 3–6% of the population, going up to as much as 30–40% in the 50+ population, constituting a health problem as well as a psychological and social ailment. The aetiology of the condition is not fully known. A genetic background to the disease is postulated [3]. Food and Drug Administration (FDA)-registered drugs in the treatment of androgenetic alopecia (AGA) include topical minoxidil and systemic finasteride in males as well as low laser light therapy [4, 5].
Minoxidil is a pyrimidine derivative with hypotensive qualities. Its exact mechanism of action is not yet known. It is proposed that it stimulates local hair growth by increasing blood flow through the skin’s vascular bed, causing vasodilation and proliferation of blood vessels surrounding hair follicles [6], and leading to an increase in vascular endothelial growth factor (VEGF) levels [7]. It results in shortening of the telogen phase, prolongation of the anagen phase, and production of a longer hair. The initial effects, involving an increased number of vellus hair, and inhibition of hair loss, should be observed 2 months into the treatment. Most sources indicate a 6-month treatment period that leads to the full effect. In a study carried out by Goren et al., only 40% of patients who had been applying minoxidil for 16 weeks responded to the therapy with hair growth [8]. Other studies have demonstrated improvement in 40% of patients 6 months into the treatment [9]. Some of the patients fail to respond to topical minoxidil therapy, with predictive factors remaining unknown to date.
Minoxidil is available as 2% and 5% solutions. Adverse events include telogen effluvium hair loss (6–8 weeks from the beginning of treatment), which recedes over the period of several weeks, facial hypertrichosis (after the 5% minoxidil solution), scalp irritation and pruritus, headache, vertigo and tachycardia. Minoxidil should not be used during pregnancy and lactation. Minoxidil is a pro-drug that is converted by the sulfotransferase enzyme to its active form of minoxidil sulfatase in the outer sheath of the hair follicle. Minoxidil sulfatase is an active metabolite that stimulates fair follicles [8].
Trichoscopy is a valuable and dynamically developing method, which serves to diagnose as well as monitor the treatment of alopecia. With the help of video dermatoscopy, one may register and compare scalp images taken throughout the treatment. The method is used more and more frequently in the diagnostics of the vascular structure in the superficial layers of the dermis.
The CD31 antibody was used as an immunohistochemical marker which enables the assessment of cutaneous vascularization.
Aim
Taking into account minoxidil’s mechanism of action that primarily impacts the vascular bed surrounding hair follicles, this study aims to assess the vascularization of scalp areas that are affected by androgenetic alopecia, and to search for a correlation between the extent of vascularization and response to minoxidil therapy.
Material and methods
Seventy-six patients with diagnosed FPHL underwent scalp biopsy (parietal region) for a histological assessment of scalp vascularization (based on the expression of CD31 in the examined tissue material). Patients were divided into two groups: with rich and poor scalp vasculature. Rich scalp vasculature was defined as the blood vessel count exceeding the median value for the study population, i.e. more than 60 vessels per cm2, whereas poor vascularization was defined as the blood vessel count of 60 vessels per cm2 or less.
All patients were started on topical 5% minoxidil, applied once daily for a minimum of 6 months, followed by assessment and video-trichoscopic imaging, with another follow-up examination scheduled 9 to 24 months into the treatment. Patients were monitored with video trichoscopy. The working assumption was that patients with rich scalp vascularization would have better treatment results. Trichoscopic images were taken with the use of Medicam 800 video dermatoscope produced by FotoFinder (Bad Birnbach, Germany). Trichoscopy images were analysed at the TrichoLAB laboratory (Warsaw, Poland). The following parameters were assessed: average hair count per cm2 (total count), average hair thickness, percentage share of hair < 30 µm (first group thickness), percentage share of hair sized 30–50 µm (second group thickness), percentage share of hair > 50 µm (third group thickness), percentage share of single hair per follicle (single unit), percentage share of double hair in follicle (double unit), percentage share of triple hair in follicle (triple unit), cumulative hair thickness per cm2 (cumulative hair thickness density), and follicular unit density per cm2 (FU density) [10].
Improvement was defined as an increase in the values of the following parameters: total count, average thickness, second and third group thickness, double unit, triple unit, cumulative hair thickness density, and FU density. Improvement was also demonstrated as a decrease in the percentage values of the following parameters: single unit, and first group thickness. Inclusion criteria included clinical features (based on the 3-stage Ludwig scale, and on the 5-stage Sinclair scale) as well as dermatoscopic (trichoscopic) and histological signs of androgenetic alopecia. An exclusion criterion was another cause of hair loss, e.g. hypothyroidism, anaemia, etc. The use of an additional form of systemic treatment, e.g. with spironolactone or finasteride, was also an exclusion criterion. All patients gave their written consent for the biopsy procedure for the purpose of histological examination. The Jagiellonian University Bioethics Committee approved the procedure. The number of the approval: 1072.6120.24.2019.
Statistical analysis
Normality of quantitative variable distribution was analysed with the Shapiro-Wilk test. Variables with distribution other than normal were presented as medians and first and third quartiles (Q1–Q3). Qualitative variables were presented as percentages. Comparative analysis was performed with the use of Pearson’s χ2 test for categorical data, and with the use of the Mann-Whitney U test for non-normally distributed continuous variables. Response to treatment was assessed with the use of repeated measures analysis of variance. In cases of statistical significance, post-hoc Scheffe tests were performed. The level of statistical significance was set at p < 0.05.
All calculations were performed using Statistica version 13 software (2017; TIBCO Software Inc.).
Results
The average age of study participants was 49. The median duration of hair loss was 4 years (Q1–Q3: 1–10 years).
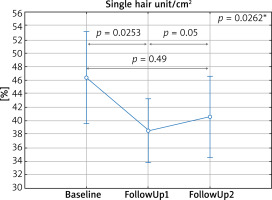
Without dividing patients into groups, a significant increase in the total count was observed 6 months into the treatment as compared to the baseline visit (Figure 1), and a decrease in the number of single hair per follicle as compared to baseline (Figure 2).
Figure 1
Average hair count per cm2 (total count) depending on the time of measurement. Baseline – first visit, FollowUp1 – visit after 6 months, FollowUp2 – visit after 9–24 months from the baseline. *In the post-hoc test a statistically significant difference between Baseline and FollowUp1 was demonstrated
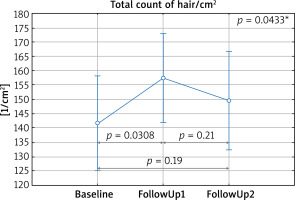
Figure 2
Percentage share of single hair per follicle (single unit) depending on the time of measurement. Baseline – first visit, FollowUp1 – visit after 6 months, FollowUp2 – visit after 9–24 months from the baseline. *In the post-hoc test a statistically significant difference between Baseline and FollowUp1 was demonstrated
A decrease in the number of single follicular units was observed in 50.67% of patients. No decrease in the total hair count was reported for 68% of patients, out of which 84.30% of patients experienced additional hair growth.
Based on the histological examination, and measurement of the number of vessels in the scalp with the use of the CD31 antibody, patients were divided into two groups. 44% of patients were included in the rich vascularization group, and the remaining ones were qualified as those with poor scalp vascularization.
When divided into different scalp vascularization groups, patients with rich scalp vascularization and those with poor scalp vasculature responded equally well to the topical treatment with minoxidil.
With respect to other comparable parameters, including hair thickness, density of follicular units, percentage share of single, double and triple units, cumulative hair thickness density, and percentage shares of different-thickness hair, no statistically significant differences were observed.
Patients who initiated their treatment earlier (up to 4 years from diagnosis) benefitted more from treatment (Table 1).
Table 1
Comparison of the results of repeated measures analysis depending on the disease duration
| Parameter | Baseline | FollowUp1 | FollowUp2 | P-value | |||
|---|---|---|---|---|---|---|---|
| Up to 4 years from diagnosis | > 4 years from diagnosis | Up to 4 years from diagnosis | > 4 years from diagnosis | Up to 4 years from diagnosis | > 4 years from diagnosis | ||
| Total count [1/cm2] (Q1–Q3) | 159 (122.00–193.00) | 126 (88.00–151.00) | 172 (130.00–210.00) | 134 (105.00–155.00) | 132 (113.00–176.00) | 147 (117.00–180.00) | 0.026 |
| Average thickness [µm] (Q1–Q3) | 55.12 (50.14–60.98) | 56.70 (51.84–60.69) | 60.25(49.06–65.01) | 57.23 (53.40–65.44) | 57.64 (52.56–64.16) | 57.70 (51.22–61.49) | 0.53 |
| Thickness 1st gr (%) (Q1–Q3) | 3.33 (1.47–7.14) | 3.03 (0–6.67) | 3.23 (0–8) | 2.38 (0–4.44) | 1.11 (0–5.41) | 4.76 (0–9.76) | 0.89 |
| Thickness 2nd gr (%) (Q1–Q3) | 29.73 (18.75–42.86) | 29.63 (23.53–34.78) | 26.47 (13.89–47.62) | 30.56 (18.52–44.00) | 25.53 (15.56–33.33) | 34.55 (25.00–38.24) | 0.59 |
| Thickness 3rd gr (%) (Q1–Q3) | 65.91 (50.00–78.79) | 66.67 (52.94–75.00) | 69.23 (47.62–84.00) | 64.71 (50.00–81.48) | 73.62 (56.67–82.22) | 61.76 (51.06–75.00) | 0.56 |
| Single unit (%) (Q1–Q3) | 37.04 (27.78–55.00) | 52.63 (31.82–66.67) | 37.50 (27.27–45.45) | 40.91 (34.78–61.11) | 42.68 (25.00–55.56) | 38.89 (27.78–52.63) | 0.64 |
| Double unit (%) (Q1–Q3) | 35 (28.57–42.86) | 33.33 (20.00–47.37) | 36.36 (31.58–43.33) | 38.10 (27.78–50.00) | 37.87 (29.41–50.00) | 41.67 (30.77–47.62) | 0.92 |
| Triple unit (%) (Q1–Q3) | 23.81 (10.53–34.78) | 11.11 (6.67–22.22) | 27.27 (11.11–35.29) | 13.64 (6.67–22.22) | 18.61 (11.76–26.32) | 22.22 (13.04–28.57) | 0.20 |
| Cum hair thick dens [µm/cm2] (Q1–Q3) | 8.70 |(7.19–11.47) | 7.28 (4.98–8.44) | 10.04 (6.99–12.57) | 7.99 (5.68–9.07) | 8.09 (6.05–10.27) | 8.35 (7.18–10.10) | 0.0102 |
| FU dens [1/cm2] (Q1–Q3) | 88.05 (71.27–96.43) | 75.47 (62.89–83.85) | 88.05(75.47–100.62) | 75.47 (62.89–83.85) | 79.66 (71.27–83.85) | 79.66 (75.47–88.05) | 0.0442# |
# No statistically significant differences observed in the post-hoc tests, Total count – average hair count per cm2, average hair thickness, first group thickness – percentage share of hair < 30 µm, second group thickness – percentage share of hair sized 30–50 µm, third group thickness-percentage share of hair > 50 µm, single unit – percentage share of single hair per follicle, double unit – percentage share of double hair in follicle, triple unit – percentage share of triple hair in follicle, cumulative hair thickness density – cumulative hair thickness per cm2, and FU density – follicular unit density per cm2.
Based on our study results, patients who were followed up on for more than 6 months (9–24 months) from their baseline visit failed to show further improvement with respect to the parameters under assessment.
Discussion
In 1988, FDA registered Minoxidil for topical treatment of androgenetic alopecia in the United States [6]. At that time, the 2% minoxidil formula was only registered for male patients. Presently, minoxidil is available as an over-the-counter product in the form of 2% and 5% solutions, and a 5% minoxidil foam (not available in Poland) both for men and women. Therapeutic results are observed up to 6 months into the treatment. Its efficacy has been proven, and the drug has a good safety profile, with minimum side effects [6, 11, 12]. Minoxidil improves blood flow through the scalp [13], and activates potassium channels, which stimulates the transition of hair follicles from the telogen phase to the anagen phase [14]. Side effects include facial hypertrichosis, contact dermatitis on the scalp, and pruritus of the scalp, resulting from the presence of propylene glycol in the minoxidil solution. Telogen hair loss may occur in the first month of treatment [15, 16].
Minoxidil is considered as first-line treatment in androgenetic alopecia. The 2% preparation is applied twice daily, and the 5% solution is applied once daily [6]. In a study carried out by Katzer et al., it was indicated that 89% of patients receive it at their first appointment [5].
According to the relevant literature, ca. 40% of patients experience improvement following topical treatment, and systemic therapy is applied in the remaining patients.
Drawing on the vascular dilation effect, the present study works on the hypothesis that patients who have richer scalp vascularization will benefit more from topical treatment than those with poorer scalp vascularization. The conducted assessments, however, refuted the above-mentioned thesis. Improvement with respect to the total hair count parameter in the studied parietal area, and with respect to the decrease in the number of single follicular units, failed to demonstrate statistically significant differences between the two study arms. Perhaps the reason behind the observed results is the fact that minoxidil’s mechanism of action is independent of the extent of scalp vascularization.
It may, however, be related to vasculature in a different way, i.e. it might be found to stimulate the formation of new blood vessels in patients with poorer vascularization at baseline, and thus improve their scalp blood supply, which was not analysed in this study, though.
One should also remember about several limitations of the study. A relatively small group of patients was subjected to assessment. Treatment efficacy assessment based on video trichoscopy also comes with some downsides, e.g. related to the accuracy of repeated measurements in the same location. Patient compliance is also an issue as the amounts of the drug applied by different patients are not always comparable.
The goal of the treatment applied was to inhibit hair loss, and accomplish gradual hair growth. In 68% of patients hair loss stopped 6 months into the treatment, while in 57.33% of patients, there was additional hair growth after 6 months of regular minoxidil use. Follow-up that went beyond the 6-month period showed no further improvement, but the achieved results were maintained. The above-mentioned treatment appears to be well grounded, irrespective of the extent of scalp vascularization.
In the studies carried out to date, it has been proven that the efficacy of minoxidil therapy is correlated with the patient’s age as well as with the duration of the disease [5]. Our study confirms the previous findings, thus reflecting clinical observations too, that patients whose treatment is aimed at inhibition of hair loss benefit more from minoxidil therapy than patients whose therapy is targeted at an additional stimulation of hair growth [9].
The video trichoscopy method used to monitor treatment results is ever more frequently applied in the diagnostics and monitoring of hair loss. It is a non-invasive technique, which replaces trichogram assessment, and makes it possible to minimize the number of skin biopsies (Figures 3–5).
Conclusions
Minoxidil is equally efficacious in patients with rich and poor scalp vascularization. Its efficacy was proven 6 months into the treatment. No significant further effects were observed, though, in the period of 6–24 months into the treatment. According to the present study, minoxidil helps accomplish the desired therapeutic outcome after a 6-month-long therapy, with longer treatment bringing no further benefits, but shown to maintain the previous therapeutic effect. The efficacy of minoxidil was demonstrated in patients irrespectively of the duration of the clinical symptoms.