Introduction
Ninety-five percent of oral neoplasms are diagnosed as squamous cell carcinoma (oral squamous cell carcinoma – OSCC). In 2017, about 390,000 new cases around the world were diagnosed (lip and oral cavity). Men suffer more often than women, especially in the sixth decade of life [1–4]. This type of cancer mainly affects people living in South-Central Asia, Central and Eastern Europe and in Malaysia [1, 2]. Smoking, betel chewing, alcohol abuse and HPV infection are the main risk factors. There is a systematic increase in the prevalence among women and patients under 50 years of age [3, 4].
In a properly functioning organism, damaged cells undergo apoptosis, a process in which various elements of the immune system participate. The main role is played by T and NK (natural killers) lymphocytes. In tumour pathology, cells gain unlimited ability to divide and survive as a result of changes in their genetic material. Lymphocyte function is the result of a balance between up-regulation (stimulation) and down-regulation (inhibition). Some types of cancers exhibit immunogenicity, i.e. the ability to induce an anti-cancer reaction. There are several molecular mechanisms involved in the regulation of the microenvironment of cancer cells and in the protection against the attack of the immune system [5]. Activation of T cells requires two signals. The first one is T cell receptor (TCR) activation – the major histocompatibility complex (MHC) on antigen-presenting cells (APC). The second signal is based on co-stimulatory molecules such as the B7-CD28 pathway [6].
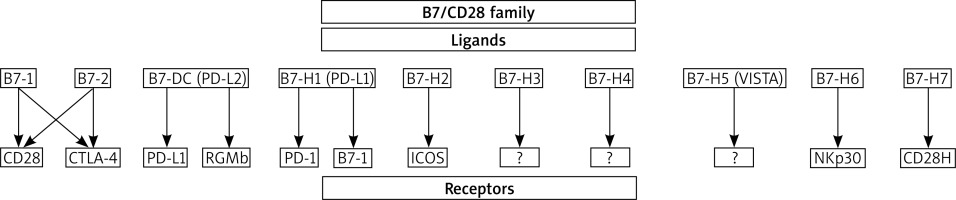
The B7/CD28 family receptors are found on cells of the immune system. These are tumour infiltrating lymphocytes (TILs). Ligands (B7 proteins) are expressed on APC cells, immune cells, epithelial cells, osteoblasts, fibroblasts and others. In addition, the presence of ligands on tumour cells was detected [7]. The B7 family includes proteins and ligands as shown in Figure 1. The B7/CD28 pathway influences the regulation of the immune response by limiting both time and strength of the inflammatory response. Although the co-stimulation mechanism of the B7/CD28 pathway is not known, monoclonal antibodies are currently used in targeted therapies of malignant tumours, autoimmune and infectious diseases [7–9].
Aim
The aim of this paper was to collect and review the B7 family proteins as prognostic factors in OSCC and to describe their role in aggressive disease progression. This particular study includes B7-H1 (PD-L1) and B7-DC (PD-L2) proteins.
Material and methods
For this review, a systematic search of the literature was conducted in the PubMed, Web of Science, Scopus, Embase, Cochrane Library, and Google Scholar databases to identify papers containing data about the B7 family proteins in OSCC. The PRISMA guidelines (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) were used [10].
PICO for study characteristics was used (P – patient/population/problem; I – intervention/exposure; C – comparison; O – outcome):
P: at least 10 patients with primary oral squamous cell carcinoma;
I: protein expression evaluation;
C: not required;
O: B7 family proteins as prognostic factors in OSCC – only B7-H1 (PD-L1) and B7-DC (PD-L2) proteins.
Inclusion criteria for papers were as follows: at least 10 patients with primary oral squamous cell carcinoma, B7 protein expression evaluation, study conducted only on humans, in the English language, prospective and retrospective studies, clinical studies and immunohistochemical evaluation of B7 protein. Articles were excluded if specimens derived from OSCC recurrences (not primary tumours), was conducted on cell lines, was not conducted on humans, the study group consisted of less than 10 patients, non-B7 family protein expression was evaluated or the study was not in English. Duplicate records as well as letters and papers that did not contain significant information were also excluded.
A retrospective analysis of articles on the B7 family proteins as risk factors in OSCC published from 2011 to 22 May 2020, was performed. Key words: “B7 family and oral cancer/OSCC/oral squamous cell carcinoma”, “PD-1/PD-L1/PD-L2 pathway and oral cancer/OSCC/oral squamous cell carcinoma”, “B7-H1/PD-L1/CD274/PDCD1LG1/B7H1/B7-H/PDCD1L1/PDCD1LG1/PDL1 and oral cancer/OSCC/oral squamous cell carcinoma”, “B7-DC/CD273/PDCD1LG2/B7DC/Btdc/PDCD1L2/PDL2/bA574F11.2 and oral cancer/OSCC/oral squamous cell carcinoma”, “PD1 signal transduction and oral cancer/OSCC/oral squamous cell carcinoma” were used. Articles were screened and sorted based on titles and abstracts. Then articles were evaluated for eligibility. Data extracted from those records were analysed in detail. The following pieces of information were collected: total patient number, occurrence of B7 family alterations in OSCC, correlations with age, gender, grading, primary tumour size (T stage), nodal metastases (N stage), staging, prognostic significance and diagnostic methods (immunohistochemistry and other methods). For randomized studies, the Cochrane Collaboration tool [11] and the methodological index for non-randomized studies (MINORS) were used [12]. The ideal global score for non-comparative studies is 16 and for comparative is 24 [12].
Results
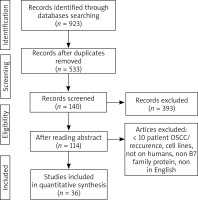
In the first step of selection 923 references were identified. 533 records were selected after exclusion of the duplicates. Eventually, 36 articles were included in the systematic review (Figure 2) [13–48]. Three studies involved PD-L2 while 35 articles were on PD-L1. The first identified study was published in 2011. Table 1 shows the articles included in the analysis [13–48].
Table 1
B7 family genetic alterations in oral squamous cell carcinomas: a review of current studies
| No. | Reference | Study type | Total patient number | Occurrence of protein expression n patients (%) | Correlations | Methods | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age | Gender | Grading | Primary tumour size (T stage) | Nodal metastases(N stage) | Staging | Prognostic significance | ||||||
| B7-H1 (PD-L1): | ||||||||||||
| 1 | Cui et al., 2020 [13] | O, R, C | 34 | No data | NE | NE | NE | NE | NE | NE | NE | IHC |
| 2 | Meehan et al., 2020 [14] | O, R, C | 67 | No data | NE | NE | NE | NE | NE | NE | NS | IHC |
| 3 | Quan et al., 2020 [15] | O, R, C | 159 | No data | NE | NE | NE | NE | NE | NE | OS – NS p = 0.742 | IHC |
| 4 | Wilms et al., 2020 [16] | O, R-P, C | 101 | 80 (79.2%) | NS p = 0.494 | S p = 0.019 | NE | NS p = 0.929 | NS p = 0.286 | NS p = 0.888 | OS – S p = 0.021 DFS – S p = 0.020 | IHC |
| 5 | Zhao et al., 2020 [17] | O, R, C | 46 | 30 (65.2%) | NS p = 0.829 | NS p = 0.956 | NS p = 0.806 | NS p = 0.052 | S p = 0.009 | S p = 0.011 | NE | IHC |
| 6 | Ahmadi et al., 2019 [18] | O, R, C | 255 | 70 (27.5%) | NS p = 0.610 | S p = 0.005 | NS p = 0.760 | NS p = 0.260 | NS p = 0.660 | NE | OS – NS p = 0.482 DSS – NS p = 0.864 DFS – NS p = 0.731 | IHC |
| 7 | Chen et al., 2019 [19] | O, P, C | 41 | 40 (97.6%) | NS p = 0.088 | NS p = 0.857 | S p = 0.010 | NS p = 0.9414 | NS p = 0.147 | NE | OS – NS p = 0.696 DFS – NS p = 0.210 | IHC |
| 8 | de Vincente et al., 2019 [20] | O, R, C | 125 | 4 (3.2%) | NE | NS p = 0.530 | NS p = 1.000 | NS p = 0.580 | NS p = 0.570 | NS p = 0.350 | DFS – S p = 0.030 | IHC |
| 9 | Kouketsu et al., 2019 [21] | O, R, C | 106 | 73 (68.9%) | NS p = 0.999 | NS p = 511 | NE | S p = 0.018 | NS p = 0.472 | S p = 0.010 | OS – NS | IHC |
| 10 | Takahashi et al., 2019 [22] | O, R, C | 77 | 46 (60%) | NS p = 0.880 | NS p = 0.360 | NS p = 0.420 | NS p = 0.750 | NS p = 0.790 | NS p = 0.580 | PFS – NS p = 0.540 OS – NS p = 0.920 | IHC |
| 11 | Tojyo et al., 2019 [23] | O, R, C | 48 | 44 (91.7%) | NS p = 1.000 | NS p = 1.000 | NS p = 0.540 | NS p = 0.520 | NS p = 1.000 | NE | DFS – NS p = 0.185 | IHC |
| 12 | Hanna et al., 2018 [24] | O, R, C | 81 (32 females) | 28 females (87%) | NE | NE | NE | NE | NE | NE | Female OS – S p < 0.001 | IHC |
| 13 | Maruse et al., 2018 [25] | O, R, C | 97 | 63 (64.9%) | NS p > 0.050 | NS p > 0.050 | NS p > 0.050 | NS p > 0.050 | S p = 0.050 | NS p > 0.050 | NS p > 0.050 | IHC |
| 14 | Stasikowska-Kanicka et al., 2018 [26] | O, R, C | 78 | 62 (79%) | NE | NE | NE | NE | NE | NE | PP – S p < 0.011 | IHC |
| 15 | Stasikowska-Kanicka et al., 2018 [27] | O, R, C | 70 | 67 (96%) | NE | NE | NE | NE | NE | NE | NE | IHC |
| 16 | Udeabor et al., 2018 [28] | O, R, C | 20 | No data | NE | NE | NE | NE | NE | NE | NE | IHC |
| 17 | Wirsing et al., 2018 [29] | O, R, C | 75 | 45 (60%) | NE | NE | NE | S p = 0.024 | NS | NE | DSD – NS p = 0.207 | IHC |
| 18 | Ahn et al., 2017 [30] | O, R, C | 68 | 45 (66%) | NS p > 0.050 | NS p > 0.050 | NE | S p = 0.002 | NS p = 0.648 | S p = 0.010 | DFS – NS p = 0.070 OS – S p = 0.039 | IHC |
| 19 | Feng et al., 2017 [31] | O, R, C | 119 | No data | NE | NE | NE | NE | NE | NE | OS – S p = 0.007 | IHC |
| 20 | Foy et al., 2017 [32] | O, R, C | 44 | No data | NE | NE | NE | NE | NE | NE | NE | IHC |
| 21 | Groeger et al., 2017 [33] | O, P, C | 15 | 15 (100%) | NE | NE | NE | NE | NE | NE | NE | IHC |
| 22 | Hirai et al., 2017 [34] | O, P, C | 24 | No data | NE | NE | NS 0.145 | NS p = 0.873 | NS p = 0.542 | NE | NE | IHC |
| 23 | Kogashiwa et al., 2017 [35] | O, R, C | 84 | 44 (52%) | NS p = 0.492 | S p = 0.010 | NE | NS p = 0.613 | NS p = 0.734 | NS p = 0.235 | OS – S p = 0.006 PFS – S p = 0.024 | IHC |
| 24 | Kubota et al., 2017 [36] | O, R, C | 46 | No data | NE | NE | NE | NE | NE | NE | NE | IHC |
| 25 | Mattox et al., 2017 [37] | O, R, C | 53 | 39 (73%) | NE | NE | NE | NE | NE | NE | OS – NS p = 0.830 | IHC |
| 26 | Takakura et al., 2017 [38] | O, R, C | 10 (patients without chemothe- rapy) | 8 (80%) | NE | NE | NE | NE | NE | NE | NE | IHC |
| 27 | Troeltzsch et al., 2017 [39] | O, R, C | 88 | 26 (29%) | NS p = 0.349 | NS p = 0.579 | NS p = 0.157 | NS p = 0.831 | S p = 0.039 | NE | DSS – NS p = 0.937 | IHC |
| 28 | Weber et al., 2017 [40] | O, R, C | 45 | 35/43 (1.:81.4% increased PD-L1_4); 32/43 (2.:74.4% increased PD-L1_2) | NE | NE | 1. G1 vs. G3 – S p = 0.020 2. G1 vs. G3 – S p = 0.010 | 1. NS p = 0.370 2. NS p = 0.487 | 1. S p < 0.002 2. S p = 0.003 | NS p > 0.05 | NE | IHC |
| 29 | Satgunaseelan et al., 2016 [41] | O, R, C | 217 | 40 (18.4%) | NS p = 0.493 | S p = 0.013 | NS p = 0.060 | NS p = 0.550 | NS p = 0.900 | NE | DSS – NS p = 0.960 DFS – NS p = 0.820 OS – NS p = 0.930 | IHC |
| 30 | Straub et al., 2016 [42] | O, R, C | 80 | 36 (45%) | NS p > 0.050 | NS p > 0.050 | NS p > 0.050 | NS p > 0.050 | NS p > 0.050 | NS p > 0.050 | OS – S p = 0.010 RFS p = 0.050 | IHC |
| 31 | Chen et al., 2015 [43] | O, R, C | 218 | 139 (64%) | NE | NE | NE | NE | NE | NE | DFS – NS p = 0.020 OS – NS p = 0.110 | IHC |
| 32 | Lin et al., 2015 [44] | O, R, C | 305 | 134 (44%) | NS p = 0.124 | S p = 0.006 | NS p = 0.326 | NS p = 0.316 | NS p = 0.736 | NS p = 0.804 | OS – NS p = 0.083 | IHC |
| 33 | Oliveira-Costa et al., 2015 [45] | O, R, C | 142 | 47/97 (49%) | NS p > 0.050 | NS p > 0.050 | NS p > 0.050 | NS p > 0.050 | NS p > 0.050 | NS p > 0.050 | DSS – S p = 0.044 | IHC |
| 34 | Cho et al., 2011 [46] | O, R, C | 45 | 39 (87%) | NS p = 0.787 | NS p = 0.745 | NS p = 0.158 | NS p = 0.393 | NS p = 0.433 | NS p = 0.736 | OS – NS p = 0.501 | IHC |
| 35 | Malaspina et al., 2011 [47] | O, R, C | 39 | No data | NE | NE | NE | NE | NE | NE | NE | IHC |
| B7-DC (PD-L2): | ||||||||||||
| 1 | Weber et al., 2019 [48] | O, P, C | 48 | 28/36 (77.8%) | NE | NE | NS p = 0.130 | NS p = 0.805 | NS p = 0.960 | NE | NS p = 0.400 | RT-qPCR |
| 2 | Groeger et al., 2017 [33] | O, P, C | 15 | 15 (100%) | NE | NE | NE | NE | NE | NE | NE | IHC |
| 3 | Kogashiwa et al., 2017 [35] | O, R, C | 84 | 20 (23.8%) | NS p = 0.792 | NS p = 1.000 | NE | NS p = 0.373 | NS p = 0.449 | S p = 0.011 | PFS – NS p = 0.350 OS – NS p = 0.058 | IHC |
[i] R – retrospective study, P – prospective study, R-P – retro-prospective study, O – observational study, C – cohort study, S – significant, NE – not examined, NS – not significant, OS – overall survival, PP – poor prognosis, DSS – disease-specific survival, DSD – disease-specific death, DFS – disease-free survival, PFS – progression-free survival, RFS – recurrence-free survival, IHC – immunohistochemistry, RT-qPCR – reverse transcription polymerase chain reaction.
All presented papers were observational and cohort-based studies (Tables 2, 3). There were no randomized controlled trials. 86.11% were retrospective (n = 31), 11.11% of the studies were prospective (n = 4) and 2.78% were retro-prospective (n = 1).
Table 2
Results of MINORS for non-comparative studies
| No. | Reference | MINORS for non-comparative studies | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | Score | ||
| 1 | Quan et al., 2020 [15] | 2 | 2 | 2 | 1 | 0 | 1 | 2 | 0 | 10 |
| 2 | Ahmadi et al., 2019 [18] | 2 | 2 | 2 | 1 | 0 | 1 | 2 | 0 | 10 |
| 3 | de Vincente et al., 2019 [20] | 1 | 2 | 2 | 0 | 0 | 0 | 2 | 0 | 7 |
| 4 | Takahashi et al., 2019 [22] | 2 | 1 | 1 | 2 | 0 | 0 | 2 | 0 | 8 |
| 5 | Tojyo et al., 2019 [23] | 2 | 1 | 2 | 2 | 0 | 1 | 2 | 0 | 10 |
| 6 | Hanna et al., 2018 [24] | 2 | 1 | 2 | 2 | 0 | 1 | 2 | 0 | 10 |
| 7 | Maruse et al., 2018 [25] | 2 | 1 | 2 | 2 | 0 | 1 | 2 | 0 | 10 |
| 8 | Udeabor et al., 2018 [28] | 2 | 0 | 0 | 1 | 0 | 0 | 2 | 0 | 5 |
| 9 | Wirsing et al., 2018 [29] | 2 | 2 | 1 | 2 | 0 | 0 | 2 | 1 | 10 |
| 10 | Ahn et al., 2017 [30] | 2 | 1 | 2 | 2 | 0 | 2 | 2 | 0 | 11 |
| 11 | Feng et al., 2017 [31] | 0 | 1 | 2 | 2 | 1 | 2 | 2 | 0 | 10 |
| 12 | Foy et al., 2017 [32] | 2 | 2 | 2 | 2 | 0 | 1 | 2 | 0 | 11 |
| 13 | Groeger et al., 2017 [33] | 2 | 1 | 2 | 1 | 0 | 0 | 2 | 0 | 8 |
| 14 | Hirai et al., 2017 [34] | 2 | 1 | 0 | 0 | 0 | 0 | 2 | 0 | 5 |
| 15 | Kogashiwa et al., 2017 [35] | 2 | 2 | 2 | 1 | 0 | 1 | 2 | 0 | 10 |
| 16 | Kubota et al., 2017 [36] | 2 | 1 | 2 | 2 | 0 | 1 | 2 | 0 | 10 |
| 17 | Mattox et al., 2017 [37] | 1 | 1 | 2 | 2 | 0 | 1 | 2 | 0 | 9 |
| 18 | Takakura et al., 2017 [38] | 2 | 1 | 2 | 2 | 0 | 1 | 2 | 0 | 10 |
| 19 | Troeltzsch et al., 2017 [39] | 2 | 2 | 2 | 0 | 0 | 1 | 2 | 0 | 9 |
| 20 | Satgunaseelan et al., 2016 [41] | 2 | 2 | 2 | 2 | 0 | 1 | 2 | 0 | 11 |
| 21 | Straub et al., 2016 [42] | 2 | 2 | 2 | 2 | 0 | 1 | 2 | 0 | 11 |
| 22 | Chen et al., 2015 [43] | 2 | 2 | 2 | 2 | 0 | 1 | 2 | 0 | 11 |
| 23 | Lin et al., 2015 [44] | 2 | 1 | 2 | 2 | 0 | 1 | 2 | 0 | 10 |
| 24 | Oliveira-Costa et al., 2015 [45] | 2 | 2 | 2 | 2 | 0 | 2 | 2 | 0 | 12 |
| 25 | Cho et al., 2011 [46] | 2 | 2 | 2 | 2 | 0 | 1 | 2 | 0 | 11 |
[i] 1 – a clearly stated aim, 2 – inclusion of consecutive patients, 3 – prospective data collection, 4 – endpoints appropriate to the aim of the study, 5 – unbiased assessment of the study endpoint, 6 – follow-up period appropriate to the aim of the study, 7 – loss to follow up less than 5%, 8 – prospective calculation of the study size; score: 0 – not reported, 1 – reported but inadequate, 2 – reported and adequate; the ideal global score for comparative studies is 16.
Table 3
Results of MINORS for comparative studies
| No. | Reference | MINORS for comparative studies | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | Score | ||
| 1 | Cui et al., 2020 [13] | 2 | 1 | 0 | 1 | 0 | 1 | 2 | 0 | 0 | 0 | 0 | 2 | 9 |
| 2 | Meehan et al., 2020 [14] | 2 | 0 | 0 | 1 | 0 | 2 | 2 | 0 | 0 | 1 | 1 | 2 | 11 |
| 3 | Wilms et al., 2020 [16] | 2 | 0 | 2 | 1 | 0 | 2 | 2 | 0 | 0 | 1 | 2 | 2 | 14 |
| 4 | Zhao et al., 2020 [17] | 1 | 2 | 2 | 0 | 0 | 1 | 2 | 0 | 1 | 1 | 2 | 2 | 14 |
| 5 | Chen et al., 2019 [19] | 2 | 1 | 2 | 2 | 0 | 1 | 2 | 0 | 2 | 0 | 1 | 2 | 15 |
| 6 | Kouketsu et al., 2019 [21] | 2 | 1 | 2 | 0 | 0 | 0 | 2 | 0 | 2 | 1 | 1 | 2 | 13 |
| 7 | Weber et al., 2019 [48] | 2 | 1 | 2 | 2 | 2 | 2 | 2 | 0 | 2 | 0 | 1 | 2 | 18 |
| 8 | Stasikowska-Kanicka et al., 2018 [26] | 2 | 2 | 2 | 0 | 0 | 1 | 2 | 0 | 1 | 1 | 1 | 2 | 14 |
| 9 | Stasikowska-Kanicka et al., 2018 [27] | 2 | 2 | 2 | 0 | 0 | 1 | 2 | 0 | 1 | 1 | 1 | 2 | 14 |
| 10 | Weber et al., 2017 [40] | 2 | 1 | 2 | 0 | 0 | 1 | 2 | 0 | 1 | 1 | 1 | 2 | 13 |
| 11 | Malaspina et al., 2011 [47] | 1 | 2 | 2 | 0 | 0 | 1 | 2 | 0 | 1 | 1 | 1 | 2 | 13 |
[i] 1 – a clearly stated aim, 2 – inclusion of consecutive patients, 3 – prospective data collection, 4 – endpoints appropriate to the aim of the study, 5 – unbiased assessment of the study endpoint, 6 – follow-up period appropriate to the aim of the study, 7 – loss to follow up less than 5%, 8 – prospective calculation of the study size, 9 – an adequate control group, 10 – contemporary groups, 11 – baseline equivalence of groups, 12 – adequate statistical analyses; score: 0 – not reported, 1 – reported but inadequate, 2 – reported and adequate; the ideal global score for comparative studies is 24.
The biggest study group consisted of 305 patients and the smallest – 10 patients. In total, 3170 patients (excluding duplicates) were analysed in the studies. The occurrence of protein expression was as follows: PD-L1 – 18.4–100% and PD-L2 – 23.8–100%. PD-L1 protein was associated with gender [16, 18, 35, 41, 44], grading [19, 48], primary tumour size (T stage) [21, 29, 30] and metastases in lymph nodes [17, 25, 39, 48]. Staging was correlated with PD-L1 [17, 20, 30] and PD-L2 [33]. Only PD-L1 protein expression proved to be a prognostic factor. Overall survival [16, 24, 31, 35, 42], disease-free survival [16, 20, 45], progression-free survival [35], poor prognosis [26] and recurrence-free survival [42] were correlated with PD-L1 protein expression. Immunohistochemistry was the most commonly used diagnostic method.
Discussion
B7 protein family and the receptors
PD-L1 (B7-H1)/PD-L2 (B7-DC)/PD-1
B7-H1/PD-L1 protein (programmed cell death 1 ligand 1/cluster of differentiation 274/CD274/PDCD1LG1/B7H1/B7-H/PDCD1L1/PDCD1LG1/PDL1) is a type I membrane protein (mass 40 kDa) encoded by the CD274 gene on chromosome 9 (locus 9p24.1). The B7-H1 protein has genes parallel to B7-1 ligand in 21%. PD-L1 has three domains: immunoglobulin constant-like domain (IgC; extracellular), the variable-like domain (IgV; extracellular) and homology domain for PD-1 [8, 49]. Intracellular structures are poorly studied. PD-L1 molecules are more prevalent than PD-L2. The PD-L1 protein suppresses the immune system [50–52]. PD-L1 protein is found on activated T lymphocytes, dendritic cells, B lymphocytes, NK cells, monocytes, macrophages, endothelial cells, epithelial cells, fibroblasts, mesenchymal stem cells, syncytiotrophoblasts, islets of Langerhans and neurons. The PD-L1 molecule plays a crucial role in the differentiation of regulatory T lymphocytes. The increase in its status is also associated with chronic inflammation and secretion of interferon γ (IFN-γ) [8, 33, 43, 53, 54]. It can affect the results of treatment of hepatitis B and C [55]. The presence of the PD-L1 protein expression has been demonstrated in glioma, ovarian cancer, renal cancer, head and neck cancer, breast cancer, sigmoid cancer, pancreatic cancer, non-small cell lung cancer and melanoma [53, 56]. Wang et al. demonstrated that PD-L1 positive expression was a prognostic factor for poor disease-specific survival in pancreatic carcinoma [53]. An increase in the response to anti-PD-1/PD-L1 therapies has been demonstrated in the treatment of lung cancer associated with smoking. An increased expression of PD-L1 refers to solid tumours, where it can serve as a defence of the tumour against the immune system [44]. In OSCC, PD-L1 protein expression was correlated with gender [16, 18, 35, 41, 44], grade [19, 48], stage [17, 21, 30], tumour size [21, 29, 30], nodal metastases [25, 26, 39–42], distant metastases [25, 26], localisation [41], vascular invasion [28], positive TILs infiltration [39], recurrence [42], disease-specific survival [45], disease-free survival [16, 20, 45], recurrence-free survival [42], overall survival [16, 24, 31, 35, 42], progression-free survival [24] and poor survival [26]. Lin et al. suggested the possibility of using PD-L1 as a prognostic factor especially in smokers and men [44]. No correlation was found between the PD-L1 protein expression and betel chewing, alcohol consumption, perineural invasion, depth of invasion, treatment, or distant metastases [41, 44]. A high expression is associated with better overall survival [30]. In addition, the presence of PD-L1 and TILs expression has been correlated with better outcome in patients with locally advanced OSCC. In those cases, the risk of recurrence was lower and survival was improved [24, 35].
B7-DC/PD-L2 protein (programmed cell death 1 ligand 2/cluster of differentiation 273/CD273/PDCD1LG2/B7DC/Btdc/PDCD1L2/PDL2/bA574F11.2) is encoded by the PDCD1LG2 gene on chromosome 9 (locus 9p24.1). The B7-DC protein has genes parallel to B7-1 ligand in 23%. PD-L2 has three domains: immunoglobulin constant-like domain (IgC; extracellular), the variable-like domain (IgV; extracellular) and homology domain for PD-1 [8, 57]. Intracellular structures are poorly studied [50–52]. PD-L2 protein is found on dendritic cells, B lymphocytes, Th2 cells, monocytes, macrophages, mast cells, hepatocytes and endothelial cells. This protein suppresses the immune system by inhibiting the T cell response through PD-1 binding. The presence of this molecule on tumour cells may cause the tumour resistance to the immune system [7, 8, 58]. PD-L2 expression was correlated with stage, but not associated with tumour size, nodal metastases, grade, progression-free survival or overall survival in OSCC [33, 35, 48].
Programmed cell death protein 1 receptor is a transmembrane protein (PD-1/cluster of differentiation 279/CD279; 50-55 kDa) encoded by the PDCD1 gene on chromosome 2 (locus 2q37.3). It consists of five domains including ITIM (immunoreceptor tyrosine-based inhibitory motif) and ITSM (immunoreceptor tyrosine-based switch motif). The IgV domain has genes parallel to CTLA-4 receptor in 21-33% and to CD28 receptor in 15.6% [8, 59]. The PD-1 receptor is found in activated T cells, B lymphocytes, NK cells, mast cells, macrophages and dendritic cells [60]. PD-1 proteins were not detected in immature T lymphocytes. The presence of PD-1 protein on TILs was revealed more frequently in comparison with peripheral blood. This was regardless of the patient’s age or HPV infection [61]. PD-1 generates an inhibitory signal that regulates the functions of T lymphocytes. This receptor has two known ligands: PD-L1 and PD-L2 [58]. Programmed cell death protein 1 receptor does not directly affect apoptosis or cell survival. PD-1 signals regulate the cellular response, but this is not completely clear. The signalling process is different in B and T lymphocytes. After binding by the PD-1 receptor, the ligand is followed by phosphorylation of tyrosine in the ITSM domain and recruitment of the SHP-2 (Src homology region 2 domain-containing phosphatase-1) and SHP-1 signal molecules (Src homology region 2 domain-containing phosphatase-2). This blocks the activation of PI-3K molecules (phosphatidyl-inositol 3-kinase) and ZAP70 (zeta chain-associated protein kinase 70) [58]. Activation of SHP-2 causes dephosphorylation of the molecule involved in TCR receptor activity and as a result reduces signal and cytokine synthesis [58]. Higher prevalence of PD-1 receptors on activated lymphocytes suggests that it is more important than the CTLA-4 receptor pathway [62]. An inflammatory reaction induces an increase in the expression of PD-L1 and PD-L2 proteins. They can serve as a feedback mechanism to reduce T cell responses in tissues and protect them from auto-aggressive damage. The binding of PD-L1 ligand to PD-1 receptor leads to the inhibition of proliferation and the reduction of IFN-γ and IL-10 cytokine secretion (interleukin 10) by about 80%, and IL-2 (interleukin 2) to the threshold below the reference values. This inhibits the proliferation of lymphocytes and promotes the survival of the cancer cell [63]. The level of PD-1 protein is high on activated lymphocytes, but it quickly decreases after removal of the antigen. However, when lymphocytes have to contend with chronic inflammation (infection or cancer), the expression of PD-1 protein is still high, which causes “exhaustion” of lymphocytes. A high oestrogen level can also induce PD-1 receptors on T cells and APC [64]. The inhibitory effect was found on Th (CD4+; helper cells) and Tc (CD8+; cytotoxic cells) lymphocytes [65]. The presence of the PD-1 protein expression has been demonstrated in bladder cancer [65] and pregnancy-associated melanoma [66]. In OSCC, PD-1 protein expression was correlated with age [30], stage [30], nodal metastases [30], perineural invasion [30] and not related to the disease-free survival [30], recurrence-free survival [42] or overall survival [30, 42]. PD-1 signalling was strongly enriched in never-smokers and never-drinkers [30]. The role of the PD-1/PD-L1 pathway is not limited to the pathogenesis of tumours. This pathway is important in various diseases: insulin-dependent diabetes mellitus, lupus erythematosus, myocarditis, inflammation of the brain and spinal cord, rheumatoid arthritis and inflammatory bowel diseases [67].
B7 pathway inhibition in cancers and OSCC
The B7/CD28 pathway influences the regulation of the immune response by limiting the time and strength of the inflammatory response. Although the co-stimulation mechanism of the B7-CD28 pathway is not known, monoclonal antibodies are currently used in targeted therapies of malignant tumours, autoimmune and infectious diseases. In cancer, CTLA-4 and PD-1 receptors are blocked by ligands (B7-1 and B7-2 for CTLA-4, and PD-L1 and PD-L2 for PD-1). As a result of ligand-receptor binding, neoplastic lesions are not recognized by the immune system. The function of monoclonal antibodies is to block CTLA-4 and PD-1 receptors. As a result, T lymphocytes re-recognize tumour antigens. The immunomodulatory drugs that block the CTLA-4 protein are ipilimumab and tremelimumab, while the PD-1 is blocked by nivolumab, pembrolizumab, pidilizumab, BMS-936559, MEDI4736 (durvalumab) and MPDL3280A [49, 56, 68–72]. They are mainly used in the treatment of melanoma [66, 70], lung cancer [66, 70], genitourinary cancer [66] and prostate cancer [72]. Targeted immunotherapy in head and neck melanoma improved survival. Ipilimumab and nivolumab had a better effect together than in monotherapy [56]. Nivolumab was used in a clinical trial of 296 melanoma, non-small-cell lung cancer, prostate cancer, renal cancer, and colorectal cancer patients. The positive tumour response (expression of PD-L1 in > 5% of cells) was seen in 18% of patients with non-small cell lung cancer, 28% with melanoma and in 27% with renal-cell carcinoma [56]. In Carbognin et al. study, nivolumab, pembrolizumab and MPDL3280A were studied in patients with melanoma, lung and genitourinary cancers. The overall response rate was significantly higher in patients with positive PD-L1 expression [68].
The role of immunomodulatory drugs in the treatment of oral cancer remains unclear and requires more research. The oral squamous cell carcinoma is highly immunosuppressive. An anti-PD-1 monoclonal antibody therapy may result in better clinical efficacy in OSCC patients [73–75]. Foy et al. studied the clinical response to pembrolizumab in HPV-negative oral squamous cell carcinoma. The PD-L1 protein was overexpressed and the score of response to pembrolizumab was higher in never-smokers and never-drinkers than in smokers and drinkers, although the mutational load was lower in never-smokers and drinkers. The main difference between oral squamous cell carcinoma in never-smokers and never-drinkers when compared to smokers and drinkers, lies in the immune microenvironment, suggesting a higher clinical benefit of PD-L1 inhibition in oral cancer in never-smokers and drinkers. The immune checkpoint inhibitors can probably extend the survival of many patients [32].
Conclusions
The biology of squamous cell carcinoma is unknown. The search for new molecular markers is extremely important. Components of the B7 family are potential objects of research. Any mutations in gene encoding PD-L1 and quantitative changes in the status of PD-L1 protein may have an impact on the prognosis of oral squamous cell carcinoma.