Introduction
The most common malignant neoplasm of the oral cavity is squamous cell carcinoma (oral squamous cell carcinoma – OSCC), which accounts for over 95% of these lesions. The lesions are more common in men over 60 years of age. Oral cancer is characterised by rapid spread and distant metastases. The outcome is still unsatisfactory. Identification of new markers may be of use in prevention, diagnostics, prognosis and choice of a targeted therapy [1–4].
The B7/CD28 family proteins are involved in the regulation of the immune response by limiting both time and strength of the inflammatory response [5, 6]. The B7 family includes proteins: B7-1, B7-2, B7-H2, B7-DC, B7-H1, B7-H2, B7-H3, B7-H4, B7-H5, B7-H6 and B7-H7. The group of receptors includes proteins: CD28, CTLA-4, ICOS, PD-1 and TMIGD2 (Table 1). They are potential objects of research when introducing new therapeutic agents [7–9].
Table 1
B7/CD28 family
Aim
The aim of this paper was to systematically review the literature and to summarise correlations between B7 family proteins and prognosis in OSCC.
Methods
For this review, a systematic search of the literature was conducted in the PubMed, Web of Science, Scopus, Embase, Cochrane Library, and Google Scholar databases to identify papers containing data about B7 family proteins in OSCC. The PRISMA guidelines (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines were used [10].
PICO for study characteristics was used (P – patient/population/problem; I – intervention/exposure; C – comparison; O – outcome):
P: at least 10 patients with primary OSCC;
I: protein expression evaluation;
C: not required;
O: B7 family proteins as prognostic factors in OSCC – only B7-1, B7-2, B7-H2, B7-H3, B7-H4, B7-H5 (VISTA), B7-H6 and B7-H7.
Inclusion criteria for papers were as follows: at least 10 patients with primary OSCC, B7 protein expression evaluation, study conducted only on humans, in the English language, prospective and retrospective studies, clinical studies and immunohistochemical evaluation of B7 protein. Articles were excluded if specimens derived from OSCC recurrences (not primary tumours), research was conducted on cell lines, was not conducted on humans, the study group consisted of less than 10 patients, non-B7 family protein expression was evaluated or the study was not in English. Duplicate records as well as letters and papers that did not contain significant information were also excluded.
A retrospective analysis of articles on the B7 family proteins as risk factors in OSCC published from 2012 to 22 May 2020 was performed. Key words: “B7 family”, “CTLA-4/B7-1/B7-2 pathway”, “B7-H2/CD275/ICOSLG/B7H2/B7RP-1/B7RP1/GL50/ICOS-L/ICOSL/LICOS/inducible T cell co-stimulator ligand”, “B7-H3/CD276/4Ig-B7-H3/B7H3/B7RP-2”, “B7-H4/B7x/B7s/VTCN1/B7H4/B7S1/B7X/B7h.5/PRO1291/VCTN1/V-set domain containing T cell activation inhibitor 1”, “B7-H5/V-domain Ig suppressor of T cell activation/VISTA/VSIR/B7H5/GI24/PP2135/SISP1/DD1alpha/C10orf54/chromosome 10 open reading frame 54/PD-1H/V-set immunoregulatory receptor”, “B7-H6/NCR3LG1”, “B7-H7/BTNL2/BTL-II/HHLA2/B7H7/B7-H5/B7y” and oral “cancer/OSCC/oral squamous cell carcinoma” were used. Articles were screened and sorted based on titles and abstracts. Then, articles were evaluated for eligibility. Data extracted from those records were analysed in detail. The following pieces of information were collected: total patient number, occurrence of B7 family alterations in OSCC, correlations with age, gender, grading, primary tumour size (T stage), nodal metastases (N stage), staging, prognostic significance and diagnostic methods (immunohistochemistry and other methods). For randomized studies, the Cochrane Collaboration tool was used [11] and the methodological index for non-randomized studies (MINORS) was used [12]. The ideal global score for non-comparative studies is 16 and for comparative is 24 [12].
Results
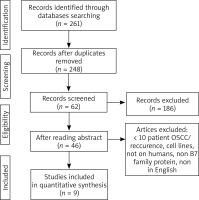
In the first step of selection 261 references were identified. 248 records were selected after the exclusion of the duplicates. Eventually, 9 articles were included in the systematic review (Figure 1) [13–21]. Three studies addressed B7-1 protein, one article was on B7-2, 2 studies involved B7-H3, one article was on B7-H4, one study involved B7-H5, one study involved B7-H6 and one involved B7-H7 (some articles were duplicated). Currently, B7-H2 protein is not described in OSCC. The first identified study was published in 2012. Table 2 shows the articles included in the analysis [13–21].
Table 2
B7 family genetic alterations in oral squamous cell carcinomas: a review of current studies
| No. | Reference | Study type | Total patient number | Occurrence of protein expression n patients (%) | Correlations | Methods | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age | Gender | Grading | Primary tumour size (T stage) | Nodal metastases (N stage) | Staging | Prognostic significance | ||||||
| B7-1: | ||||||||||||
| 1 | Rah et al., 2018 [13] | O, R, C | 60 | No data | S p = 0.016 | NS p = 0.521 | NE | NS p = 0.889 | NS p = 0.233 | NS p = 0.655 | ReFS – S p = 0.038 DFS – NS p = 0.156 | IHC |
| 2 | Zhang et al., 2017 [14] | O, P, C | 40 | 22 (55%) | NE | NE | NS p > 0.050 | NE | S p = 0.036 | S p = 0.020 | NE | IHC |
| 3 | Dayan et al., 2012 [15] | O, R, C | 64 | No data | NE | NE | NE | NE | NE | NE | NE | IHC |
| B7-2: | ||||||||||||
| 1 | Zhang et al., 2017 [14] | O, P, C | 40 | 23 (57.5%) | NE | NE | NS p > 0.050 | NE | S p = 0.024 | S p = 0.041 | NE | IHC |
| B7-H2: | ||||||||||||
| NE | ||||||||||||
| B7-H3: | ||||||||||||
| 1 | Chen et al., 2015 [16] | O, R, C | 72 | 48 (67%) | NS p > 0.050 | NS p > 0.050 | NE | S p = 0.0001 | NS p > 0.050 | S p = 0.004 | OS – S p = 0.005 | IHC |
| 2 | Zhang et al., 2015 [17] | R, CSS | 76 | 51 (67.1%) | NE | NE | S p = 0.011 | NS p = 0.135 | S p = 0.026 | NS p = 0.135 | NE | IHC |
| B7-H4: | ||||||||||||
| 1 | Wu et al., 2016 [18] | O, R, C | 165 primary OSCC 12 recurrent OSCC 10 OSCC with pre-surgical radiotherapy 17 OSCC with pre-surgical chemotherapy | 165 (100%) | NE | NE | S p < 0.010 | NS p = 0.0683 | S p < 0.050 | NS p = 0.068 | PS – S p < 0.050 | IHC |
| B7-H5 (VISTA): | ||||||||||||
| 1 | Wu et al., 2017 [19] | O, R, C | 165 primary OSCC 12 recurrent OSC 10 OSCC with pre-surgical radiotherapy 17 OSCC with pre-surgical chemotherapy | 165 (100%) | NE | NE | NS p > 0.050 | NS p > 0.050 | S p < 0.050 | NS p > 0.050 | OS – S p = 0.036 | IHC |
| B7-H6: | ||||||||||||
| 1 | Wang et al., 2017 [20] | O, R, C | 50 | 24 (48%) | NS p > 0.050 | NS p > 0.050 | S p = 0.0186 | NS p > 0.050 | NS p > 0.05 | NS p = 0.460 | DFS – S p = 0.005 OS – S p = 0.007 | IHC |
| B7-H7: | ||||||||||||
| 1 | Xiao et al., 2019 [21] | O, R, C | 201 | No data | NE | NE | NS | NS | NS | NE | OS – S p = 0.031 | IHC |
[i] R – retrospective study, P – prospective study, O – observational study, C – cohort study, CSS – cross-sectional study, S – significant, NE – not examined, NS – not significant, OS – overall survival, PS – poor survival, PP – poor prognosis, DSS – disease-specific survival, DSD – disease-specific death, DFS – disease-free survival, PFS – progression-free survival, RFS – recurrence-free survival, ReFS – relapse-free survival, IHC – immunohistochemistry, RT‐qPCR – reverse transcription polymerase chain reaction.
Observational studies were the most commonly presented papers. There were no randomized controlled trials. 88.89% were retrospective (n = 8) and 11.11% of the studies were prospective (n = 1). Studies were mostly cohort-based (88.99%, n = 8), rarely cross-sectional (2.23%, n = 1) (Tables 3 and 4).
Table 3
Results of MINORS for non-comparative studies
| No. | Reference | MINORS for non-comparative studies | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | Score | ||
| 1 | Zhang et al., 2017 [14] | 2 | 2 | 2 | 1 | 0 | 1 | 2 | 0 | 10 |
| 2 | Dayan et al., 2012 [15] | 2 | 2 | 2 | 2 | 0 | 1 | 2 | 0 | 11 |
[i] 1 – a clearly stated aim, 2 – inclusion of consecutive patients, 3 – prospective data collection, 4 – endpoints appropriate to the aim of the study, 5 – unbiased assessment of the study endpoint, 6 – follow-up period appropriate to the aim of the study, 7 – loss to follow up less than 5%, 8 – prospective calculation of the study size; score: 0 – not reported, 1 – reported but inadequate, 2 – reported and adequate; the ideal global score for comparative studies is 16.
Table 4
Results of MINORS for comparative studies
| No. | Reference | MINORS for comparative studies | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | Score | ||
| 1 | Xiao et al., 2019 [21] | 2 | 1 | 2 | 2 | 1 | 1 | 2 | 0 | 1 | 1 | 1 | 2 | 16 |
| 2 | Rah et al., 2018 [13] | 2 | 2 | 2 | 2 | 2 | 1 | 2 | 0 | 2 | 2 | 2 | 2 | 21 |
| 3 | Wang et al., 2017 [20] | 2 | 1 | 2 | 2 | 0 | 1 | 2 | 0 | 1 | 1 | 1 | 2 | 15 |
| 4 | Wu et al., 2017 [19] | 2 | 2 | 2 | 2 | 0 | 1 | 2 | 0 | 1 | 1 | 1 | 2 | 16 |
| 5 | Wu et al., 2016 [18] | 2 | 2 | 2 | 2 | 0 | 1 | 2 | 0 | 1 | 1 | 1 | 2 | 16 |
| 6 | Chen et al., 2015 [16] | 2 | 0 | 2 | 2 | 0 | 1 | 2 | 0 | 1 | 1 | 1 | 2 | 14 |
| 7 | Zhang et al., 2015 [17] | 2 | 2 | 2 | 0 | 0 | 1 | 2 | 0 | 2 | 2 | 2 | 2 | 17 |
[i] 1 – a clearly stated aim, 2 – inclusion of consecutive patients, 3 – prospective data collection, 4 – endpoints appropriate to the aim of the study, 5 – unbiased assessment of the study endpoint, 6 – follow-up period appropriate to the aim of the study, 7 – loss to follow up less than 5%, 8 – prospective calculation of the study size, 9 – an adequate control group, 10 – contemporary groups, 11 – baseline equivalence of groups, 12 – adequate statistical analyses; score: 0 – not reported, 1 – reported but inadequate, 2 – reported and adequate; the ideal global score for comparative studies is 24.
The biggest study group consisted of 201 patients and the smallest – 40 patients. In total, 893 patients (excluding duplicates) were analysed in the studies. The following proteins have been described: B7-1, B7-2, B7-H3, B7-H4, B7-H5, B7-H6 and B7-H7 (Table 2). In all tested samples, the expression of proteins was found by testing proteins B7-H4 and B7-H5. The occurrence of protein expression was as follows: B7-1 – 55%; B7-2 – 57.5%; B7-H3 – about 67%, B7-H4 – 100%; B7-H5 – 100%, B7-H6 – 48% and B7-H7 – no data. Only B7-1 expression was correlated with age [13]. Grading was correlated with B7-H3 [17], B7-H4 [18] and B7-H5 [19] protein expression. Primary tumour size (T stage) was associated with B7-H3 protein [16]. Metastases in lymph nodes were correlated with B7-1 [14], B7-H3 [17], B7-H4 [18] and B7-H5 protein expression [19]. Staging was correlated with B7-1, B7-2 [14] and B7-H3 protein expression [17]. B7-1, B7-H3, B7-H4, B7-H5, B7-H6 and B7-H7 protein expressions were prognostic factors. Overall survival was significantly associated with B7-H3 [17], B7-H5 [19], B7-H6 [20] and B7-H7 [21]. Disease-free survival was correlated with B7-H6 protein [56]. Poor survival was related to B7-H4 [18]. Relapse-free survival was significantly associated with B7-1 [13]. Immunohistochemistry was used as a diagnostic method in all the studies.
Discussion
B7 protein family and the receptors
B7-1/B7-2/CTLA-4 pathway
B7-1 protein is a type I membrane protein encoded by the CD80 gene on chromosome 3 (locus 3q13.33). B7-1 has two domains: immunoglobulin constant-like domain (IgC; extracellular) and the variable-like domain (IgV), required for receptor binding. This protein is expressed on the surface of monocytes, macrophages, mast cells, activated B lymphocytes, podocytes, fibroblasts, antigen-presenting and dendritic cells. T cells are co-stimulated and activated by B7-1 protein. The function of the CD80 protein is to stimulate the signal that activates T lymphocytes. This is necessary for their survival. B7-1 protein binds to CD28, CTLA-4 and PD-L1 receptors. The binding of the CD80 protein to the CD28 receptor enables autoregulation and intercellular connection. The binding to CTLA-4 receptor allows reduction of regulation and cellular dissociation. B7.1 protein plays a role in the induction of T cell anti-tumour immunity [8, 9, 13, 15, 22]. The presence of the B7-1 protein expression has been demonstrated in numerous malignancies, such as human hepatocellular cancer [22], thyroid cancer [23], bladder cancer [24] and pancreatic cancer [25]. Chaux et al. revealed that in colorectal cancer, expression of the B7-1 and B7-2 molecules were not found. As a consequence, the immune system does not recognise cancer cells as antigenic [26, 27]. Wang et al. demonstrated that the B7-1 negative status was a prognostic factor for poor disease-specific survival in pancreatic carcinoma [25]. The literature documenting B7-1 protein in OSCC is limited. CD80 protein in OSCC was first reported in 2012 by Dayan et al. [15]. According to some studies, the percentage of common B7-1 protein range was 55% [14]. B7-1 protein expression correlated with age, recurrence, relapse-free survival [13], stage, and nodal metastases in OSCC [14].
B7-2 is a type I membrane protein encoded by the CD86 gene on chromosome 3 (locus 3q13.33). This protein is expressed on the surface of antigen-presenting cells, T cells, B lymphocytes, dendritic cells, monocytes, macrophages and mast cells. It is the source of the co-stimulatory signal. The B7-2 protein has genes parallel to B7-1 ligand in 27%. It activates T lymphocytes and is necessary for their survival. B7-2 protein binds to CD28 and CTLA-4 receptors. The binding of the CD86 protein to the CD28 receptor enables autoregulation and intercellular connection. The binding to CTLA receptor allows to reduce regulation and cellular dissociation. B7.1 protein plays a role in the induction of T cell anti-tumour immunity [7, 14, 22]. The presence of the B7-2 protein expression has been demonstrated in human hepatocellular carcinoma [27] and thyroid carcinoma [23]. B7-2 protein in OSCC was reported in 2017 by Zhang et al. In the study mentioned, the percentage of common B7-2 protein was 57.5%. This protein expression correlated with stage and nodal metastases [14].
CD28 is a co-stimulatory receptor of B7-1 and B7-2 molecules. CD28 is encoded by the CD28 gene on chromosome 2 (locus 2q33.2). This protein consists of the immunoglobulin variable-like domain (IgV; extracellular), transmembrane domain, stalk domain and a cytoplasmic domain. CD28 receptor is found on naive T lymphocytes, plasma cells, NK and NKT cells (natural killers T cells). This molecule is a receptor of B7-1 and B7-2 ligands. This protein stimulates T cell receptor (TCR). CD28 molecule is associated with T cell activation, proliferation, tolerance and survival. This protein provides a signal for production of various interleukins (e.g. interleukin 2) and amplifies the immune system response. Activated T cells stimulate B lymphocytes to produce antibodies. The pathways associated with CTLA-4 and ICOS cannot be activated without the induction of the CD28 receptor [8, 28–30]. CD28 protein affects apoptosis via Bcl-xL (anti-apoptotic protein – B-cell lymphoma-extra large). Lymphocytes are not subject to programmed death [31]. Activation of CD28 causes the induction of transcription and/or stabilization of cytokine mRNAs: IL-4, IL-5, IL-8, IL-13, interferon γ (IFN-γ) and tumour necrosis factor α (TNF-α). The lack of co-stimulation by CD28 leads to the anergy of clonal lymphocytes. Anergy means a lack of reaction by the body’s defence mechanisms to foreign substances [29, 30].
CTLA-4 is a protein receptor of B7-1 and B7-2 molecules and down-regulates T lymphocytes activity. CTLA-4 is encoded by the CTLA-4 gene located on chromosome 2 in locus 2q33.2. The CTLA-4 has genes parallel to the CD28 receptor in 26.9% [8, 32, 33]. This protein consists of the immunoglobulin variable-like domain (IgV; extracellular), transmembrane domain, stalk domain and a cytoplasmic domain. CTLA-4 receptor is found on T lymphocytes, dendritic cells, B lymphocytes, NK cells, NKT cells, monocytes, granulocytes and fibroblasts. Roles of CTLA-4 include inhibition of T cell activation and their response by blocking AKT protein (serine-threonine protein kinase = protein kinase B). CTLA protein skips phosphatidyl-inositol 3-kinase signalling pathway. Most CTLA-4 molecules are found in lysosomes. First, binding of B7-1 and B7-2 ligands to the CD28 receptor occurs. It activates the lymphocyte. Then, the CTLA-4 receptor is transported to the cell membrane and expressed. Activation of CTLA-4 provides a stimulus that can trigger feedback to inhibit lymphocyte activation. The presence of the CTLA-4 receptor on the tumour cell membrane avoids a specific immune response [8]. Excessive expression of the gene encoding the CTLA-4 protein may be associated with: Ewing’s sarcoma, laryngeal, hepatocellular, prostate, lung, breast, cervical [6, 32, 34–37], nasopharyngeal [38], and head and neck carcinoma [39]. In OSCC studies, the CTLA-4 gene polymorphism was detected via polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP). Single nucleotide polymorphisms (SNPs): -1722C/T, -1661A/G, -1661G/G, -1661A/A and -318C/T in the promoter region, 49A/G in exon 1, and CT60A/G in the 3’untranslated region (UTR) were assessed in patients with tobacco-related carcinoma. Genotypes: -1722CC, -1661AG and CT60AA are more frequent in patients with OSCC. The polymorphism of the CTLA-4 −1661-genotype alone and in combination with other polymorphisms may be a risk factor for oral cancer [30, 32]. In Moreira et al. study, no statistical association was found between overall survival and expression level of CTLA-4 protein [40].
B7-H2 and ICOS
B7-H2 is encoded by the ICOSLG gene on chromosome 21 (locus 21p12). The B7-H2 protein has genes parallel to B7-1 ligand in 27%. It binds to ICOS. B7-H2 was found on B lymphocytes, T cells, monocytes, mast cells, dendritic cells, macrophages, endothelial cells, epithelial cells, fibroblasts and osteoblasts. It plays an important role in cell-to-cell signalling, immune response, cell proliferation, functions of effector and Treg lymphocytes. CD275 can promote T lymphocyte production of cytokines, including IL-10, IL-4, IL-5, IL-7, IL-10, and interferon-γ. High levels of B7-H2 protein have been demonstrated in patients with colon cancer [8, 41], but have not been examined in OSCC.
ICOS is a co-stimulatory molecule and receptor of B7-H2 protein. This molecule is encoded by the ICOS gene on chromosome 2 (locus 2q33.2). The ICOS has genes parallel to CD28 receptor in 24.1% [8, 29, 30]. This protein consists of the immunoglobulin variable-like domain (IgV; extracellular), transmembrane domain, stalk domain and a cytoplasmic domain [8]. ICOS protein is expressed on activated T and B lymphocytes and binds to B7-H2 protein. It plays an important role in cell-to-cell signalling, immune response and cell proliferation. ICOS is upregulated on activated T lymphocytes. The gene polymorphism of ICOS was studied in positions +637 A/C and +1599 C/T [8, 29, 30].
B7-H3
B7-H3 protein is a type I membrane protein encoded by the CD276 gene on chromosome 15 (locus 15q24.1). The B7-H3 protein has genes parallel to B7-1 ligand in 29%. It has two domains: IgV and IgC. B7-H3 molecule connects to the previously unknown receptor [7, 8, 42]. TLT-2 (TREML2) may be a binding partner for B7-H3. It may affect the growth of CD4 (T helper lymphocytes) and CD8 (T cytotoxic lymphocytes) T cells proliferation, and selectively increases the secretion of IFF. B7-H3 protein is found on activated dendritic cells, monocytes, macrophages, T, B and NK lymphocytes, epithelial cells, fibroblasts and osteoblasts. B7-H3 molecule is a protein that controls the immune system. There are studies suggesting stimulating effects (by T lymphocytes) as well as inhibiting the immune system (by NK cells). The B7-H3 protein induces selective production of interferon-γ. Inhibition of the immune system can occur through NFAT (nuclear factor of activated T cells), NF-κB (nuclear factor κ-light-chain-enhancer of activated B cells), and AP-1 (activator protein 1) factors. This affects the TCR, which regulates gene transcription [7, 8, 43]. The presence of the B7-H3 protein expression has been demonstrated in neuroblastoma, lung cancer [44], pancreatic cancer [45], breast cancer [46], ovarian cancer [47], endometrial cancer [48], prostate cancer [49], renal cancer [50], gastric cancer [51], colorectal cancer [52], gallbladder cancer [53], hepatocellular cancer [54], and head and neck cancer [55]. It results in poor outcome in patients with these types of cancer [7]. Potential anti-B7-H3 therapy seems to be beneficial for the treatment of central nervous system tumours. The expression of B7-H3 can be associated with a decrease in the number of active lymphocytes, a large tumour size and the presence of metastases [56]. B7-H3 protein level in OSCC was higher than in normal mucosa [17]. In OSCC, B7-H3 protein expression was associated with grade [17], tumour size [16], nodal metastases [17], and overall survival [16]. B7-H3 was correlated with poor overall survival [16]. Protein expression was not associated with location of cancer, distant metastases, recurrence, alcohol consumption, betel chewing or smoking [16].
B7-H4
B7-H4 protein is a type I membrane protein encoded by the B7-H4 gene on chromosome 1 (locus 1q13.1). The B7-H4 protein has genes parallel to B7-1 ligand in 21%. B7-H4 has two domains: IgC and IgV. Like B7-H3, B7-H4 is present in most tissues and is a control protein of the immune system. This protein was found on dendritic cells, monocytes, macrophages and mesenchymal stem cells. However, the protein expression itself is very limited [7, 8, 57]. B7-H4 inhibits the proliferation of CD4 T cells, resulting in suppressed production of cytokines (IL-2), and the proliferation of CD8 upon TCR/CD28 ligation. It binds to the receptor only on activated T cells, which can be a factor differentiating from other B7/CD28 family proteins. This suggests that the B7-H4 pathway may be another system that inhibits the cellular response of T lymphocytes. B7-H4 expression is correlated with an increased expression of IL-6 [7, 58, 59]. The presence of the B7-H4 protein expression has been demonstrated in lung cancer [60], ovarian cancer [61], cervical cancer [62], oesophagus cancer [63], gastric cancer [64], colorectal cancer [65], pancreatic cancer [66], prostate cancer [49], thyroid cancer [67] and melanoma [68]. B7-H4 protein level in OSCC was higher than in normal mucosa. B7-H4 protein expression was associated with grade and nodal metastases [18]. B7-H4 expression was an independent prognostic factor and associated with poor overall survival [18].
B7-H5 (VISTA)
B7-H5 protein is a type I membrane protein (50 kDa) encoded by the C10orf54 gene on chromosome 10 (locus 10q22.1). The B7-H5 protein has genes parallel to B7-1 ligand in 24%. VISTA has two domains: IgC and IgV. VISTA transcription is regulated by the p53 protein. It is a control protein of the immune system. There are reports that the VISTA may function as a ligand and as a receptor [7, 8, 19, 69–72]. VISTA was found in dendritic cells, T lymphocytes, monocytes, macrophages, bone marrow cells and granulocytes. It is not present on B or NK cells. VISTA has inhibitory effects on T cell activity, proliferation, cytokine production, but not on B lymphocytes. Increased levels of VISTA proteins were observed in T lymphocytes in autoimmune diseases, cancer and generalized inflammation [8, 69]. The presence of the B7-H5 protein expression has been demonstrated in gastrointestinal cancer [70–76], gastric cancer [74], lung cancer [7], pancreatic cancer [7, 75], myeloma [76] and head and neck cancer [72]. B7-H5 protein level in OSCC was higher than in normal mucosa and correlated with nodal metastases. B7-H5 expression was not an independent prognostic factor and was not associated with poor prognosis. The high level of VISTA was significantly correlated with PD-L1, CTLA-4, IL13Rα2 (interleukin-13 receptor subunit α2), PI-3K, p-STAT3 (signal transducer and activator of transcription 3), CD11b (integrin αb) and CD33 (siglec-3/sialic acid binding Ig-like lectin 3) protein expression. High levels of VISTA and low levels of CD8 were associated with poor prognosis in the primary OSCC. These results indicate that VISTA may be a potential immunotherapeutic target in OSCC [19].
B7-H6 and NKp30
B7-H6 protein is a type I membrane protein encoded by the B7-H6 gene. This molecule has two domains: IgC and IgV. It binds to NKp30 molecule. B7-H6 protein was not found in healthy tissue, but was found on human monocytes and macrophages after LPS (lipopolysaccharides) and IFN-g stimulation. It was not observed on B or T cells. It is responsible for the activation of the immune system [7, 77]. The presence of the B7-H6 protein expression has been demonstrated in lung cancer [78], ovarian cancer [79], hepatocellular cancer [80], glioma [81], leukaemia, various sarcomas and renal cancer [82]. The literature documenting B7-H6 protein in OSCC is limited. In one of the studies, the percentage of common B7-H6 protein range was 48% and correlated with grade, overall survival and disease-free survival. The recurrence, differentiation and expression of B7-H6 protein were related to the prognosis [20].
NKp30 receptor is binding to B7-H6 protein. It is a type I protein encoded by the NCR3 I gene. NKp30 belongs to NCR family proteins (natural cytotoxicity receptors). This receptor is found on NK cells. NKp30 consists of the ectodomains: a membrane proximal stalk domain and a distal ligand-binding domain. The stalk domain can be important for binding to ligands. It has three isoforms: NKp30a, NKp30b and NKp30c. NKp30a and NKp30b have a stimulating effect and NKp30c has a suppressive effect. NKp30 recognizes B7-H6 antigens on the tumour. In a properly functioning system, NK cells destroy cancer cells directly via cytotoxicity or indirectly by cytokine secretion. The glycosylation status of NKp30 alters its binding affinity for B7-H6 [7, 83]. Expression of NKp30 protein was demonstrated in lung cancer (using flow cytometry) [78].
B7-H7 and CD28H
B7-H7 is encoded by the HHLA2 gene on chromosome 3 (locus 3q13.13). B7-H7 is a co-stimulation protein with three immunoglobulin-like domains (IgV-IgC-IgV). In contrast to other B7 family proteins, the expression of this protein is very low in healthy cells. It is present on monocytes, macrophages, B lymphocytes, mature dendritic cells, endothelial cells, epithelial cells and syncytiotrophoblast cells. There are probably two receptors of the B7-H7 – CD28H molecule with opposite functions. Binding of antigen-presenting cells (APC) to CD28H stimulates T cell proliferation and cytokine production. B7-H7 is likely to have a greater effect on NK cells (via the NKp30 receptor) than on T cells (interferon-γ, TNF-α, IL-5, IL-10, IL-13, IL-17a, and IL-22). The presence of the B7-H7 protein expression has been demonstrated in various cancer cell lines: leukaemia, neuroblastoma, melanoma, ovarian cancer, breast cancer, lung cancer, thyroid cancer, oesophagus cancer, gastric cancer, colon cancer, pancreatic cancer, hepatocellular cancer, bladder cancer and renal cancer [7, 8, 84, 85]. The studies were carried out using immunohistochemistry [7, 9]. B7-H7 expression was with poor overall survival.
CD28H is a receptor encoded by the TMIGD2 gene on chromosome 19 (locus 19p13.3). The TMIGD2 has genes parallel to CD28 receptor in 22.8%. It binds to B7-H7. CD28H is found in natural killer cells, APC cells and naive T lymphocytes in peripheral blood. Binding to the receptor can trigger two actions: stimulation and suppression [7, 8, 85].
Conclusions
The expression status of B7 proteins in OSCC cells remains unclear. There are not many scientific reports. The rapid increase in knowledge about the molecules of the B7/CD28 family opens new possibilities for the treatment and prognosis of patients. The data presented indicate the complexity of cellular and molecular processes related to the formation, development and invasion of oral cancer. Various mutations in genes encoding the B7 family and quantitative and functional changes in B7 proteins may be important in the prognosis of OSCC.