Introduction
Asthma has the features of airway hyperresponsiveness and reversible airflow obstruction [1–5]. It has become one of the most common airway diseases in both children and adults [6]. The incidence of childhood asthma is estimated to range from 10% to 13%, and its incidence in adults is about 4% [7–9]. The exacerbation of asthma results in an acute obstruction of expiratory airflow due to airway inflammation, bronchospasm and hypersecretion [10, 11]. If the emergent and effective treatment is not conducted for asthma exacerbation, patients suffer from hypercapnia, severe hypoxemia and subsequent respiratory failure [12].
The Global Initiative for Asthma (GINA) guideline recommends early administration of supplemental oxygen and medications such as nebulized bronchodilators and systemic corticosteroid for the management of acute severe asthma [6]. The administration approaches of oxygen mainly include nasal cannula and nonrebreather mask which are promising for chronic obstructive pulmonary disease (COPD), but show limited success in acute severe asthma [13]. High-flow oxygen therapy is the oxygen-delivering method of inspired fraction (FiO2) up to 1.0 via a high-flow nasal cannula across a range of flows from 2 to 60 l/min. It also provides positive airway pressure and decreases rebreathing from anatomic dead space, which helps reduce the respiratory effort [14, 15]. Many studies reported the effectiveness of high-flow oxygen therapy for hypoxemic respiratory failure and cardiogenic pulmonary oedema [16, 17].
Current evidence is insufficient for routine clinical use of high-flow oxygen therapy for asthmatic patients. Recently, several studies have investigated the efficacy of high-flow oxygen versus conventional oxygen therapy for these patients, but the results are conflicting [12, 18, 19].
Aim
This systematic review and meta-analysis of RCTs aims to assess the efficacy of high-flow oxygen versus conventional oxygen therapy in asthmatic patients.
Material and methods
This systematic review and meta-analysis are performed based on the guidance of the Preferred Reporting Items for Systematic Reviews and Meta-analysis statement and Cochrane Handbook for Systematic Reviews of Interventions [20–22]. No ethical approval and patient consent are required because all analyses are based on previous published studies.
Literature search and selection criteria
We have systematically searched several databases including PubMed, Embase, Web of Science, EBSCO, and the Cochrane library from inception to May 2021 with the following key words: “high-flow oxygen” AND “asthma”. The reference lists of retrieved studies and relevant reviews are also hand-searched and the above process is performed repeatedly in order to include additional eligible studies.
The inclusion criteria are as follows: (1) study design is RCT, (2) patients are diagnosed with asthma, and (3) intervention treatments are high-flow oxygen versus conventional oxygen therapy.
Data extraction and outcome measures
Some baseline information is extracted from the original studies, and they include the first author, number of patients, age, female, medical history of asthma and detailed methods in two groups. Data are extracted independently by two investigators, and discrepancies are resolved by consensus. The primary outcomes are PaCO2 and PaO2. Secondary outcomes include the intubation rate, hospital length of stay and dyspnoea score.
Quality assessment in individual studies
The methodological quality of each RCT is assessed by the Jadad Scale which consists of three evaluation elements: randomization (0–2 points), blinding (0–2 points), dropouts and withdrawals (0–1 points) [23]. One point would be allocated to each element if they are conducted and mentioned appropriately in the original article. The score of Jadad Scale varies from 0 to 5 points. An article with Jadad score ≤ 2 is considered to have low quality. The study is thought to have high quality if Jadad score ≥ 3 [22, 24].
Statistical analysis
We assess standard mean difference (SMD) with 95% confidence intervals (CIs) for continuous outcomes and odd ratio (OR) with 95% CI for dichotomous outcome. Heterogeneity is evaluated using the I 2 statistic, and I 2 > 50% indicates significant heterogeneity [25]. The random-effects model is used for all meta-analyses. We search for potential sources of heterogeneity when encountering significant heterogeneity. Sensitivity analysis is performed to detect the influence of a single study on the overall estimate via omitting one study in turn or performing the subgroup analysis. Owing to the limited number (< 10) of included studies, publication bias is not assessed. Results with p < 0.05 are considered to be significant. All statistical analyses are performed using Review Manager Version 5.3 (The Cochrane Collaboration, Software Update, Oxford, UK).
Results
Literature search, study characteristics and quality assessment
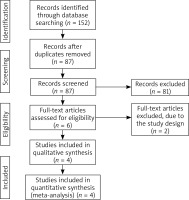
Figure 1 shows the detail flowchart of the search and selection results. 152 potentially relevant articles are identified initially and four RCTs are finally included in the meta-analysis [12, 18, 19, 26].
The baseline characteristics of four included RCTs are shown in Table 1. These studies are published between 2018 and 2021, and the total sample size is 175. The flows of high-flow oxygen range from 2 to 60 l/min. One RCT involves children with asthma [26], while the other three RCTs involve adult patients [12, 18, 19].
Table 1
Characteristics of included studies
Two studies report dyspnoea score, PaCO2, PaO2 and intubation rate [12, 18], three studies report hospital length of stay [12, 18, 26], and two studies report dyspnoea score [12, 19]. Jadad scores of the four included studies vary from 3 to 4, and all four studies have high quality based on the quality assessment.
Primary outcomes: PaCO2 and PaO2
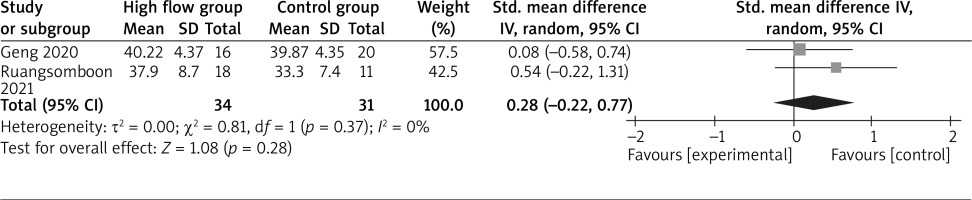
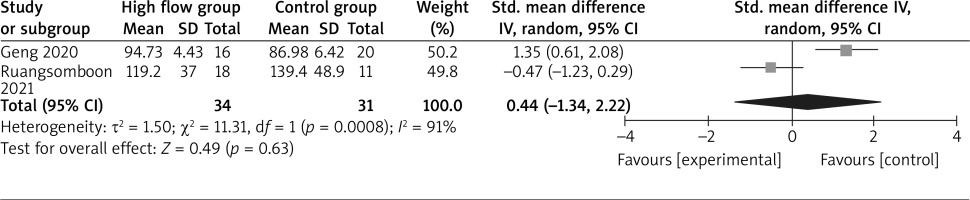
The random-effect model is used for the analysis of primary outcomes. The results find that compared to conventional oxygen therapy for asthma, high-flow oxygen has no obvious influence on PaCO2 (SMD = 0.28; 95% CI: –0.22 to 0.77; p = 0.28) with no heterogeneity among the studies (I 2 = 0%, heterogeneity p = 0.37, Figure 2) or PaO2 (SMD = 0.44; 95% CI: –1.34 to 2.22; p = 0.63) with significant heterogeneity among the studies (I 2 = 91%, heterogeneity p = 0.0008, Figure 3).
Sensitivity analysis
Significant heterogeneity remains for PaO2. There are just two RCTs, and thus we do not perform sensitivity analysis by omitting one study in each turn to detect the source of heterogeneity.
Secondary outcomes
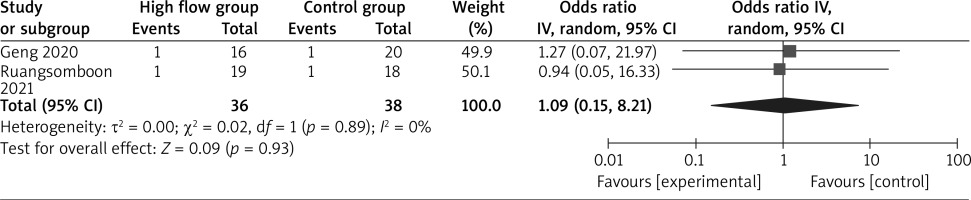
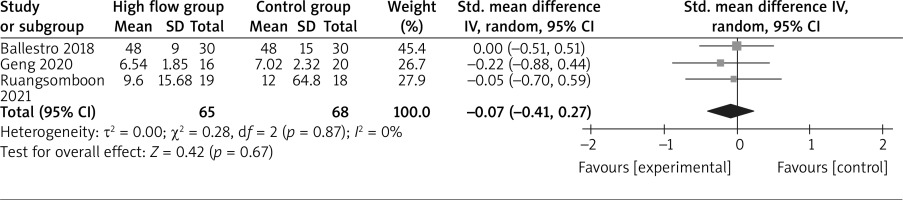
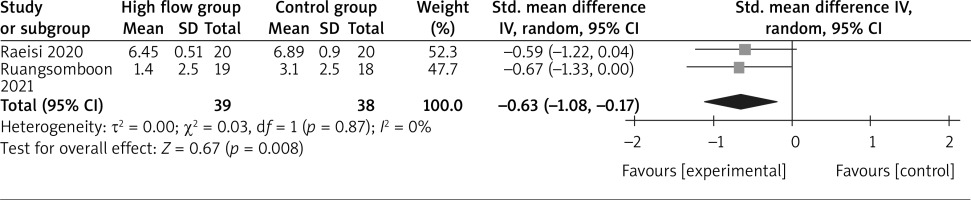
In comparison with conventional oxygen therapy for asthma, high-flow oxygen shows no obvious impact on intubation rate (OR = 1.09; 95% CI: 0.15 to 8.21; p = 0.93; Figure 4) or hospital length of stay (SMD = –0.07; 95% CI: –0.41 to 0.27; p = 0.67; Figure 5), but is associated with significantly lower dyspnoea score (SMD = –0.63; 95% CI: –1.08 to –0.17; p = 0.008; Figure 6).
Discussion
Several studies demonstrated the benefit of high-flow oxygen therapy in decreasing the arterial partial pressure of carbon dioxide in patients with hypercapnic respiratory failure [27–29]. In patients with severe asthma, high-flow oxygen therapy also showed favourable benefit in decreasing the respiratory rate and increasing their expiratory time which thereby help decrease dynamic hyperinflation. In addition, heated and humidified air could increase the comfort and reduce bronchoconstriction induced by cold air [12].
Our meta-analysis included four RCTs and 175 patients, and the results revealed that high-flow oxygen therapy was associated with a significantly lower dyspnoea score than conventional oxygen therapy for asthma, but PaCO2, PaO2, intubation rate and hospital length of stay were found to be similar between two groups. In paediatric patients with acute severe asthma, high-flow oxygen therapy was approved to improve clinical severity compared with conventional oxygen therapy [26, 30]. Its benefit in decreasing PaCO2 is confirmed in both children and adolescents [31].
In Figure 3, only adults patients are included for the meta-analysis of PaCO2 after pooling the results of two RCTs [12, 18]. These suggest that high-flow oxygen therapy may provide better efficacy to decrease PaCO2 and improve clinical severity in paediatric patients than that in adult patients. Regarding the sensitivity analysis, there is significant heterogeneity, which may be caused by different flow range and detail methods of high-flow oxygen therapy as well as various severity of asthma.
Several limitations exist in this meta-analysis. Firstly, our analysis is based on only four RCTs, and more RCTs with a larger sample size should be conducted to explore this issue. Next, there is significant heterogeneity, which may be caused by different flow range and severity of asthma. Children and adult patients are both included in this meta-analysis, which may affect the evaluation of efficacy such as the decrease in PaCO2. Finally, it is not feasible to perform the analysis of some outcomes such as modified Borg scale and respiratory rate.