Introduction
Psoriasis commonly increases the risk of systemic disorders including obesity, metabolic syndrome, diabetes mellitus, and cardiovascular morbidities [1–6]. In patients with psoriasis, many inflammatory factors are overproduced, including tumour necrosis factor-α (TNF-α), interleukin-6 (IL-6), IL-8, IL-12, IL-17, IL-19, and IL-23, which lead to the occurrence of metabolic syndrome [7–9]. In particular, metabolic syndrome is characterised by central obesity, high triglyceride level, low high-density lipoprotein cholesterol (HDL-C), hypertension, and glucose intolerance, and it serves as a strong predictor of coronary heart disease, diabetes, and stroke [5, 10–13]. Thus far, metabolic syndrome has not been well controlled in patients with psoriasis.
Metformin is widely used for the treatment of type 2 diabetes, and it has additional beneficial effects on weight reduction, decreased hyperinsulinaemia, improved lipid profiles, augmented fibrinolysis, and enhanced endothelial function [14–17]. In patients with psoriasis, metformin was reported to prevent the progression of metabolic syndrome, inhibit weight loss, and improve treatment efficacy [18].
Several RCTs showed that metformin may have the capability to improve the treatment efficacy for psoriasis, but the results were not well established [18–20]. We therefore conducted this meta-analysis of RCTs to evaluate the effectiveness of metformin on treatment efficacy for psoriasis patients.
Material and methods
Study selection and data collection
This meta-analysis did not require ethical approval or patient consent, and it was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-analysis statement and Cochrane Handbook for Systematic Reviews of Interventions [21, 22]. We searched PubMed, EMbase, Web of science, EBSCO and the Cochrane library up to July 2022, using the search terms “metformin” AND “psoriasis”. The inclusion criteria were as follows: (1) study design was RCT; (2) patients were diagnosed with psoriasis; and (3) intervention treatments were metformin versus placebo.
Quality assessment
The Jadad Scale was used to evaluate the methodological quality of individual RCTs [23]. This scale consisted of 3 evaluation elements: randomization (0–2 points), blinding (0–2 points), and dropouts and withdrawals (0–1 points). The score of the Jadad Scale varied from 0 to 5 points. Jadad score ≤ 2 suggested low quality, while Jadad score ≥3 indicated high quality [24].
Outcome measures
The following information was extracted: first author, publication year, sample size, age, gender, psoriasis area severity index (PASI), and methods of 2 groups. The primary outcomes were PASI 75% reduction and erythema, scaling, and induration (ESI) 75% reduction. Secondary outcomes included fasting plasma glucose (FPG), triglycerides, total cholesterol, and low-density lipoprotein (LDL).
Statistical analysis
A team consisting of 3 authors performed the statistical analyses. Odds ratio (OR) with 95% CI was applied to evaluate dichotomous outcomes, and standard mean difference (SMD) with 95% CI was used to assess continuous outcomes. I2 statistic was applied to assess the heterogeneity, and significant heterogeneity was observed if I2 > 50% [25]. The random effect model was used regardless of the heterogeneity. We conducted the sensitivity analysis by omitting one study in turn or using subgroup analysis. P ≤ 0.05 indicated statistical significance, and Review Manager Version 5.3 was used in all statistical analyses.
Results
Literature search, study characteristics, and quality assessment
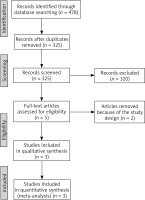
The flow chart for the selection process and detailed identification was presented in Figure 1. In total 478 publications were identified through the initial search of databases. Ultimately, 3 RCTs were included in the meta-analysis [18–20].
The baseline characteristics of the 3 eligible RCTs in the meta-analysis are summarized in Table 1. The 3 studies were published between 2016 and 2022, and the total sample size was 148. There were similar baseline characteristics between the metformin group and the control group. The doses of metformin supplement were 500 mg or 1000 mg once daily.
Table 1
Characteristics of included studies
Among the 3 RCTs, 3 studies reported PASI 75% reduction [18–20], 2 studies reported ESI 75% reduction [18, 20], 3 studies reported FPG [18–20], while 2 studies reported triglycerides, total cholesterol, and LDL [18, 20]. Jadad scores of the 3 included studies varied from 3 to 4, and all studies were considered to have high quality according to quality assessment.
Primary outcomes: PASI 75% reduction and ESI 75% reduction
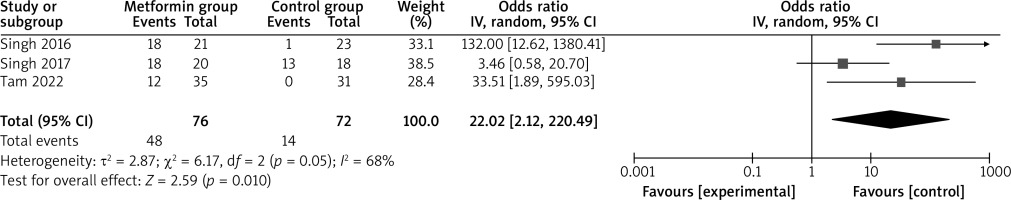
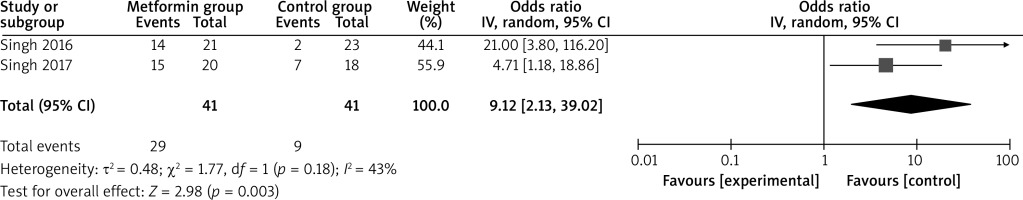
Compared to the control group for psoriasis, metformin intervention was associated with significantly increased PASI 75% reduction (OR = 22.02; 95% CI: 2.12 to 228.49; p = 0.01) with significant heterogeneity among the studies (I2 = 68%, heterogeneity p = 0.05, Figure 2) and ESI 75% reduction (OR = 9.12; 95% CI: 2.13 to 39.02; p = 0.003) with low heterogeneity among the studies (I2 = 43%, heterogeneity p = 0.18, Figure 3).
Sensitivity analysis
Significant heterogeneity was observed for PASI 75% reduction. As shown in Figure 3, the study published by Singh in 2017 showed a result that was almost out of range of the others and probably contributed to the heterogeneity [20]. After excluding this study, the results suggested that, compared with control intervention for psoriasis, metformin intervention was still associated with substantially increased PASI 75% reduction (OR = 76.31; 95% CI: 12.38 to 470.39; p < 0.00001), and no heterogeneity remained (I2 = 0, p = 0.47).
Secondary outcomes
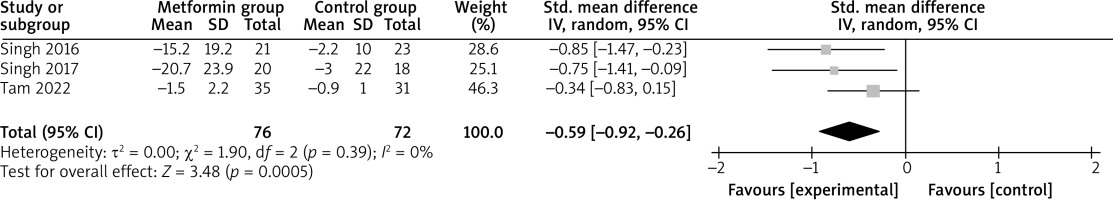
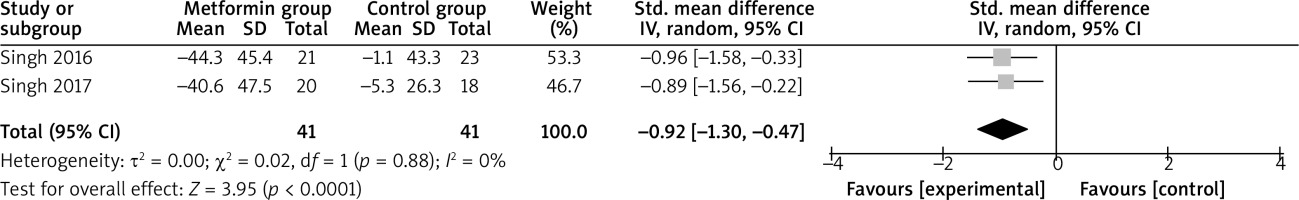
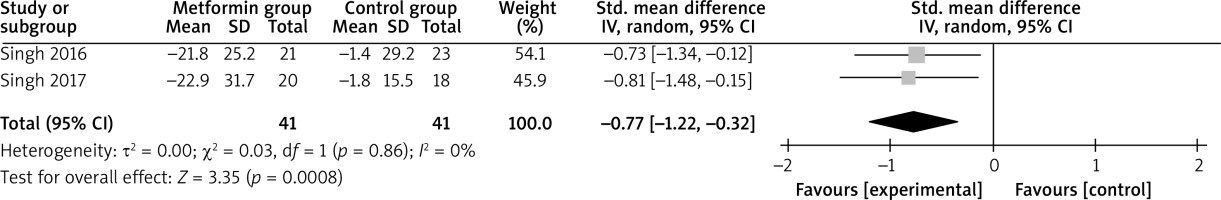
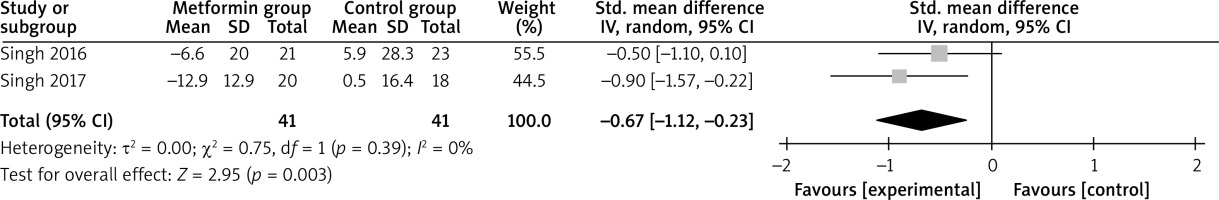
Compared with control intervention for psoriasis, metformin intervention resulted in substantially reduced FPG (SMD = –0.59; 95% CI: –0.92 to –0.26; p = 0.0005; Figure 4), triglycerides (SMD = –0.92; 95% CI: –1.38 to –0.47; p < 0.0001; Figure 5), total cholesterol (SMD = –0.77; 95% CI: –1.22 to –0.32; p = 0.00008; Figure 6), and LDL (SMD = –0.67; 95% CI: –1.12 to –0.23; p = 0.003; Figure 7).
Discussion
In addition to its hypoglycaemic effect, metformin can serve as an anti-inflammatory agent by activating adenosine monophosphate-activated protein kinase (AMPK) in extracellular signal-related kinase 1/2 signalling pathway, which subsequently inhibits dendritic cell and T-cell activation as well as cell proliferation [20]. These may increase the treatment efficacy of metformin supplementation for psoriasis patients [26, 27]. Our meta-analysis included 3 RCTs and 148 patients with psoriasis. The results confirmed that metformin intervention was able to improve the treatment efficacy as shown by the increased PASI 75% reduction and ESI 75% reduction.
Metformin showed a significantly improved percentage of metabolic syndrome parameters as compared to the placebo group for psoriasis patients [20]. Our meta-analysis confirmed that metformin intervention had beneficial effects on FPG, triglycerides, total cholesterol, and LDL for psoriasis patients. Metformin can act through the activation of AMPK, which not only inhibits the activation of inducible nitric oxide synthase, dendritic T cell, and monocyte/macrophage, but also activates IL-10 and TGF-β, thereby exerting the anti-inflammatory function [26]. One study reported that metformin had the capability to reduce mean IL-6 and TNF-α levels, which may be comprised by the use of methotrexate in all patients [20].
Regarding the sensitivity analysis, there was still significant heterogeneity, which may be caused by different factors. Firstly, the doses of metformin supplement were 500 mg or 1000 mg once daily, which may affect the efficacy assessment. Secondly, psoriasis was commonly associated with the incidence of metabolic syndrome, but the severity of metabolic syndrome was different. Thirdly, the various combination methods of metformin may produce some bias.
We should also consider several limitations. Firstly, our analysis was based on only 3 RCTs and more studies with large patient samples should be conducted to confirm this finding. Secondly, the methods and doses of metformin were different in the included studies and may mainly account for some heterogeneity. Thirdly, psoriasis patients with various levels of metabolic syndrome may produce some bias for efficacy assessment.