Introduction
The term ‘allergy’ was introduced by Clemens von Pirquet in 1906; however, the earliest records of allergic conditions date back to approximately 2,650–2,600 BC, when the king of Egypt died due to an insect sting. In the 6–5th century BC, Hippocrates observed allergic symptoms, in the form of gastrointestinal dysfunction and urticaria, following cow milk consumption. The earliest records of asthma symptoms and the recommended treatments date nearly as far back, to the medical treatises by Celsus and by Aretaeus of Cappadocia from the first century AD [1–5].
The immense current (20th and 21st century) worldwide interest in the epidemiology of allergic conditions is a result of the growing incidence, bordering on pandemic proportions, and reaching unprecedented levels. According to the White Book on Allergy published in 2011, the prevalence of allergic diseases increases dramatically all over the world, in both developed and developing countries. Such conditions include bronchial asthma; allergic rhinitis; anaphylaxis; drug, food, and insect-venom allergies; eczema; urticaria; and angioedema. The increased incidence of allergy is particularly pronounced in children, who have been affected the most by this trend for the last two decades [6].
First epidemiological studies
One of the first survey-based epidemiological studies was conducted in 9,000 families in the United States in the period 1928–1931. The resulting analyses showed 0.5% of children aged 5–9 years to suffer from bronchial asthma. Subsequent studies, conducted in the period from 1976 to 1980, demonstrated an increased prevalence of asthma in children and adolescents aged 3–17 years. Asthmatic symptoms were reported in 9.5% of children among a total of 7,300 respondents. Fourteen years later, 15.9% of children aged 5–17 years had been diagnosed with bronchial asthma [7]. In comparison with the 1983 data, the prevalence of asthma had increased by 6.7% in this population [8]. By the beginning of the 21st century, the total number of bronchial asthma patients in the United States had increased by 60% in comparison with the data from the early 1980s [9]. Meanwhile, the rates of allergic rhinitis ranged from 9% to 42% in the study population [10]. The current incidence is very high in the population of under-18-year-olds, 41% of whom suffer from asthma, 11.6% from atopic dermatitis, 10% from allergic rhinitis, 8.4% from hay fever, and 5.4% from food allergies [11].
In 1949, a team led by doctor Obtułowicz conducted one of the first epidemiological studies in Poland in patients from three internal medicine departments of Wroclaw clinics. Asthma was observed in 0.5–3.5% of respondents [12]. A subsequent study, called Polish Multicentre Study of the Epidemiology of Allergic Diseases (PMSEAD), which was conducted under the auspices of the Polish Society of Allergology in the period from 1998 to 1999, showed allergic conditions to be a growing problem in the Polish population. According to the results of that study, 8.6% of 3–16-year-olds and 5.4% of adults were affected with asthma. Allergic rhinitis was detected in 12% of adults and 11% of children [13, 14]. Subsequent studies were conducted in the form of surveys in the Lublin Voivodship in 1995–2001. Those studies indicated a distinct rise (from 3.4% in 1995 to 9.6% in 2001) in the prevalence of asthma in children aged 8–13 years residing in rural areas [15]. Studies conducted in a group of 404 children from central Poland indicated a significantly higher prevalence of asthma in school-age children from large cities in comparison to the figures in children from rural areas (16.42% vs. 1.97%) [16]. Observing the International Study of Asthma and Allergies in Childhood (ISAAC) study protocol, a population of children aged 6–7 years and adolescents aged 13–14 years was assessed in Krakow and Poznan in the years 1994–1995 and 2001–2002. A comparison of study results from the two periods showed considerably increased rates and symptoms of allergic rhinitis and conjunctivitis both in 6–7-year-olds from Krakow and Poznan (by 33.6% and 31.3%, respectively) and in 13–14-year-olds from Krakow and Poznan (by 41.4% and 39.6%, respectively). Moreover, the evaluated population showed a significant increase in the symptoms of asthma, with symptoms in 6–7-year-olds from Krakow and Poznan having increased by 14.3% and 8.1%, respectively, and symptoms in 13–14-year-olds from Krakow and Poznan having increased by 7.5% and 7.9%, respectively [17, 18]. A pilot study from 2003 was to assess the validity of the questionnaire that had been adapted for Poland from its original version developed for the European Committee Respiratory Health Survey II (ECRHS II). The study involved 1,000 adult urban respondents from Świdnica (Lower Silesian Voivodship). The results demonstrated a two-fold increase in the rates of allergic conditions in comparison with the figures from the mid-1990s [13]. More recent studies have focused not only on the risk factors for allergic conditions, but primarily on the quality of life and the direct and indirect costs to the patients.
Standardized epidemiological studies
Due to their diverse methodologies, the Polish, European, and global studies conducted to date have offered no insights into the problem of allergic condition incidence in any given country or worldwide. The observed increasing trend in the incidence and prevalence of asthma, allergic rhinitis, and eczema and the effects of these conditions on patients, their families, and work environment have led to the development of a standardized ECRHS questionnaire for adults and a standardized ISAAC questionnaire for children [9, 19–21]. The data obtained with these questionnaires made it possible to compare the results of epidemiological studies conducted in individual European populations and worldwide. These standardized questionnaires ensure repeatability of data, which reveals the natural history of allergies, its population-specific dynamics, trends over time, and predicting the factors crucial for developing prevention programs and adequate organization of healthcare resources to best help allergy patients [22].
European Community Respiratory Health Survey (ECRHS)
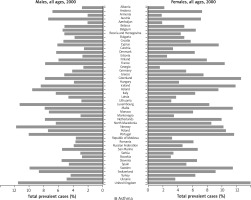
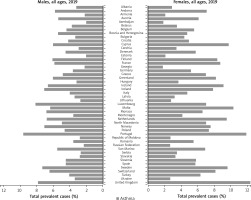
The first study with the use of an ECRHS questionnaire (ECRHS I) was conducted in 1990. The survey involved 140,000 adult respondents aged 20–44 years, from 56 centres in 25 – mainly European – countries. The year 1998 saw a launch of another, improved study ECRHS II in adults from 29 centres in 14 countries. These studies focused primarily on asthma, allergies diseases, and allergic rhinitis [23–25]. Such large epidemiological studies with the use of standardized methodology, which offered an opportunity to make comparisons between countries and continents, demonstrated a growing problem posed by allergic conditions and the factors affecting the increased incidence. The ‘allergy epidemic’ of the 21st century is a result of a surge in allergic conditions, most likely caused by genetic and environmental factors, including lifestyle (housing, family, diet, non-medicinal stimulants, drugs, hygiene, etc.). The obtained data were different for individual countries. Figures 1 and 2 illustrate the differences in the diagnosed cases of asthma in selected European countries between the years 2000 and 2019. The rates of bronchial asthma ranged from 2% to 3.3% in Estonia, Germany, Spain, Greece, Austria, Algeria, and Italy and from 8% to 11.9% in the United Kingdom, New Zealand, and Australia [25, 26]. The results of ECRHS II showed that 20.7% of young adult respondents exhibited the symptoms of asthma. The greatest discrepancy was observed between India (4.1%) and Ireland (32%). There was a slight decrease (by 0.9%) in the rates of asthma symptoms in comparison with those from ECRHS I [27]. The prevalence of allergic rhinitis in adults ranged from 11.8% of the Spanish population to 46% of the Australian population [27]. There were substantial differences in the rates of severe asthma. In this observational cohort study, primary care medical records showed that 4.2% of adults in Sweden suffer from a severe form of asthma [28]. In Denmark there has been an approximately 8% increase in the proportion of patients with severe asthma, whereas some studies show that up to 20% or even over 30% of patients with asthma have at least some evidence of severe disease [29, 30]. In Europe, the proportion of allergic rhinitis in adults aged 20–44 years has been approximately 20.9% [29]. Finnish studies from the years 1966–2017 showed the rates of allergic rhinitis rising from 0.06% to 10.70% [31]. The ECRHS also showed an association between allergic rhinitis and bronchial asthma. Bronchial asthma was reported in 6% of patients with seasonal allergic rhinitis, 9% of patients with perennial rhinitis, and in 18% of patients with both seasonal and perennial rhinitis [23]. Western lifestyle often leads to overweight and obesity, which increase the risk of asthma [32]. Retrospective studies (recorded at call) by Reijula et al. demonstrate the incidence of allergic conditions in Finnish men in the period 1926–2017. In the period from 1926 to 1961, there was a low prevalence of asthma (0.02–0.08%). Subsequently, there was an overall 12-fold increase from 0.29% in the year 1966, to 3.44% in 2001, and up to 5.19% in 2017. The prevalence of allergic rhinitis in industrialized countries has increased over the last three decades by 2–3%, with 15–30% of children and 2–10% of adults currently affected [6]. Finnish studies from the period of 1926–2017 also show an increase in the rates of atopic dermatitis in men from 0.15% in 1926 to 2.90% in 2017 [31]. Atopic dermatitis used to be limited to early childhood; however, currently it often persists into adult life. There are also newly diagnosed cases in adults [6].
The International Study of Asthma and Allergies in Childhood (ISAAC)
The first large-scope epidemiological ISAAC was conducted in 6–7-year-olds and 13–14-year-olds in 57 countries in the mid-1990s. The video questionnaire study population comprised 257,800 children aged 6–7 years and 768,591 adolescents aged 13–14 years. Allergic rhinitis symptoms were reported in 7.5% of this population. Another of these studies took place in the years 2002–2003 and involved 193,404 children aged 6–7 years residing in 38 countries and 304,679 adolescents aged 13–14 years residing in 56 countries [27]. The highest rates of bronchial asthma were observed in Costa Rica (37.6%), New Zealand (22.2%), the United Kingdom (20.9%), and Australia (20%), with the lowest rates of asthma found in Albania (5%) and Indonesia (8%). The rates of wheezing in 14-year-olds living in Albania, China, Romania, or Russia (2.1–4.4%) were nearly 15 times lower than those in Australia, New Zealand, Ireland, or the United Kingdom (29.1–32.2%), with a similarly wide difference in wheezing rates in younger children (ranging from 4.1% in Indonesia to 32.1% in Costa Rica) [33]. The proportion of patients with severe asthma seems to be lower in children than in adults. For example, only 7 out of 329 twelve-year-olds in Sweden had severe asthma, as defined by the World Health Organization (WHO), which suggests that its prevalence is 0.23% in the general population and 2.1% in children with asthma. Sixty-seven out of 616 Norwegian children had asthma, with the estimated proportion of severe asthma equal to 0.5% in the general population and 4.5% in 10-year-olds [34, 35]. The most recent studies conducted in Denmark demonstrate that genetics, socioeconomic status, and smoking are important risk factors for asthma. Moreover, study results indicated that exposure to high levels of exhaust-pollution in the air affects the development of asthma in children [35]. Studies demonstrated that obesity in girls is a risk factor for asthma [28]. The mean proportion of atopic dermatitis in 6–7-year-old children was 7.9%, ranging from 0.9% in India to 22.5% in Ecuador. The prevalence of atopic dermatitis in European adolescents aged 13–14 years ranges from 1.5% in Latvia to 15% in Bulgaria, Denmark, Finland, and Hungary, with high rates also reported in Asia and Latin America. The mean prevalence of atopic dermatitis in 13–14-year-old adolescents was at 7.3% and ranged from 0.2% in China to 24.6% in Colombia; with a high prevalence also reported in Africa and Latin America. Moreover, atopic dermatitis was more common in girls than in boys in both evaluated populations. The high mean rates of atopic dermatitis in Western and Northern Europe seem to be higher than in countries in the east, which is most likely due to socioeconomic conditions. In some rapidly developing countries of Central and Eastern Europe, such as Poland and Hungary, the rates of atopic dermatitis considerably exceed the global rates of approximately 7.3%. The differences shown by the epidemiological studies mentioned above are visible not only between countries but also between different areas within the same country. A similarly non-uniform distribution of the disease can be seen worldwide [33, 36–42]. Allergic rhinitis was reported in 14.6% of 13–14-year-olds and in 8.5% of the evaluated 6–7-year-olds [33, 38]. The rates of allergic rhinitis and eczema symptoms have increased in many study centres, except those that had initially shown high levels of asthma in adolescents. The mean rate of allergic rhinitis was 7.5% of the evaluated adolescents, with the condition reported at the lowest rates in Albania (1.4%) and the highest in Portugal and Nigeria (above 35%) [21, 33]. The most recent of the ISAAC studies showed that the rates of rhinitis with itching and watery eyes vary between different centres from 0.8% to 14.9% in 6–7-year-olds and from 1.4% to 39.7% in 13–14-year-olds. The highest prevalence rates were reported in Africa (18.0%) and Latin America (17.3%) and the lowest in Northern and Eastern Europe. These studies also showed that countries with a low (< 5%) prevalence of asthma, e.g. Indonesia, Albania, Georgia, and Greece also showed a low prevalence of allergic rhinitis. Countries with a very high (> 30%) prevalence of asthma, such as Australia, New Zealand, and the United Kingdom also showed high rates of allergic rhinitis (15–20%) [39]. The reasons for such a large geographical diversification include urbanization, the degree of industrial development, the related air pollution as well as racial and cultural differences, hygiene and lifestyle.
Epidemiology of Allergic Diseases in Poland (ECAP) study
The information obtained from the previous studies study based on ECRHS and ISAAC methodology helped design the comprehensive epidemiological study called the Epidemiology of Allergic Diseases in Poland (ECAP). The main purpose of this study was to provide the missing data on the epidemiology of allergic conditions in Poland and to compare them with those from around the world. The ECAP study was conducted in the period from 2006 to 2008 and included 20,454 respondents divided into three age groups: 6–7 years, 13–14 years, and 20–44 years. The study was conducted in nine (one rural and eight urban) areas of Poland [22]. The most common chronic allergic conditions are allergic rhinitis in over 20% of respondents, symptomatic asthma in nearly 16% of respondents, atopic dermatitis in approximately 7–9% of respondents, and other allergic conditions in another several percent in the largest cities and much less commonly in pollutant-free but allergen-rich urban environment. Unfortunately, 40% of Poles struggle with allergic diseases, which are a cause of higher patient expenditures on pharmaceutical products, frequent hospitalizations, absences from school or work, and discomforts in daily living [22, 38, 40, 41–44]. It is important to mention that multi-stage epidemiological studies help identify the relationships between selected allergic conditions, with both surveys and outpatient examinations of patients with rhinitis showing many times higher rates of asthma than in populations with no rhinitis symptoms. The fact of declaring symptoms of allergic rhinitis in a survey study increased the odds of declaring asthma by over five-fold, and the odds of developing symptoms of asthma by over three-fold. Developing symptoms of atopic dermatitis also increases the risk of asthma. The results of the ECAP study demonstrated that the prevalence of allergic conditions indicates a growing trend in the entire Polish population. Allergies are most common in children in general and boys in particular [22, 23, 45]. A comparison with other European regions, Poland was shown to be, beside the United Kingdom and Germany, one of the countries most severely affected with this health problem. Analysis of data from various epidemiological studies over the last 20 years revealed the rates of allergies to be increasing steadily, with this tendency being unlikely to become reversed in Poland in the near future. Moreover, the ECAP study showed that up to 75% of patients with asthma may not be aware of their disease. Patients whose asthma was diagnosed by the doctor during the outpatient examination, did not know about her disease before [22, 45]. According to the European Academy of Allergy and Clinical Immunology (EAACI), in 15 years, nearly a half of Europeans will suffer from an allergy. In Poland, there are over 15 million people already affected. Allergic conditions make up the highest proportion of chronic diseases in children and adolescents, and these figures are most likely to increase further with each decade.
Prevention programs
The increase in the rates of allergic conditions in the evaluated populations remains the topic of interest for many researchers, physicians, and nurses, as well as for politicians, healthcare institution representatives and local governments. Therefore, it seems rational to introduce prevention programs and increase public awareness of the principles of secondary prevention, by health education, including education of healthcare professionals, introduction of innovative solutions to control the environment around the house and indoors, as well as by launching exemplary programs, such as POLASTMA conducted by the Polish Society of Allergology or the program for prevention, early detection, and detection of allergic conditions in Poland conducted by the Unit of Environmental Hazard Prevention and Allergology at the Medical University of Warsaw. Other important elements of early allergy detection are diagnostic tests, which determine the entire subsequent medical treatment and ultimately contribute towards improving the patients’ quality of life [22, 23, 26, 43]. One benefit gained from the epidemiological studies conducted so far are the developed protocols for the management, diagnostics, treatment, and prevention of allergic conditions. Allergic Rhinitis and its Impact of Asthma (ARIA) is one of the first sets of guidelines developed by the WHO based on scientific research. These guidelines have been updated based on the Evidence-Based Medicine, as provided by reports published in renowned journals [27–29]. Despite the fact that allergic rhinitis and atopic dermatitis are more prevalent, it is asthma that has been the focus of most epidemiological studies on allergies. Holst et al. showed that 72.9% of reports provided data on asthma, whereas allergic rhinitis and atopic dermatitis (eczema) were considered in 15.7% and 24.5% of studies, respectively [39]. In 1993, the WHO established amendments to the Global Initiative for Asthma (GINA), an international organization whose mission is to raise awareness among healthcare professionals and to develop guidelines for the diagnosis and treatment of bronchial asthma. Over the last decades, also the EAACI developed the “Global Atlas of Allergy”, which contains information on asthma, allergic rhinitis, food allergy, anaphylaxis, atopic dermatitis, animal and insect allergies, urticaria, and occupational allergy [38]. Allergies are often referred to as an ‘epidemic of the 21st century;’ therefore, epidemiological studies conducted in Poland and globally are so important. The most important components of these studies are comparable methodology and regularity. These measures will help develop effective prevention and management programs for patients with chronic allergic conditions, to improve the quality of everyday life and reduce the costs of healthcare due to failure to detect or late detection of allergic conditions. Allergic symptoms can be effectively combated with the treatments of modern medicine. Every allergy patient, provided they follow their doctor’s recommendations, can lead a normal life undisturbed by asthma attacks, rashes, or oedema. Allergies can be controlled and treated with selected therapies that offer the highest chance of complete symptom elimination. Because of that, the World Allergy Organization (WAO) began its mission to increase the level of awareness of health risks posed by asthma and allergic conditions and to improve the situation by integrated continual education, promoting scientific studies, improving the care of allergic patients, and measures aimed at achieving effective prophylaxis. Precisely these objectives and goals and their medical rationale were published by the WAO in the White Book on Allergy in 2011–2012 and in the European Declaration on Immunotherapy EAACI, summarizing the current state of knowledge on allergy-related problems in the world and recommending measures to control them better [38, 46–49].