Introduction
Pemphigus is a heterogeneous group of autoimmune acantholytic illnesses expressing possibly misleading clinical peculiarities and running a potentially lethal course [1]. In humans there are seven desmosomal cadherins, three desmocollins (DSC1–3) and four desmogleins (DSG1–4). Of those, DSG1 and DSG3 are widely regarded as targets for IgG pathogenic antibodies in “classical” variants of pemphigus. DSG1 is targeted in the pemphigus foliaceus (PF) circle of diseases, whereas DSG3 or both DSG3 and DSG1 are targeted in the pemphigus vulgaris (PV) circle of diseases [2, 3]. The knowledge accumulated worldwide over the years undoubtedly indicates that the intricate networking of intrinsic and extrinsic factors [4–7] brings about the clinical unmasking of those illnesses.
The imaging direct immunofluorescence (DIF) of perilesional tissue, adapted for use in autoimmune blistering diseases in the 1960s [8], detecting deposits of immunoreactants having a chicken wire or fishnet appearance is universally accepted as the gold standard in diagnosing pemphigus [9]. Nevertheless, triaging DIF in intraepidermal bullous dermatoses is advisable [10]. Of note, IgG4 deposition can have an appearance resembling dew drops on the spider web [11]. DIF of plucked scalp hair, for the detection of deposits of immunoreactants, including IgG4, in the outer root sheath, was proposed to reduce tissue traumatization [12, 13]. DIF images can be obtained using fluorescence microscopy systems with different light sources [14], and DIF procedure can be automated [15]. Moreover, it has been argued that an IgG4 enriched antitotal IgG fluorescein isothiocyanate (FITC) conjugate can be used for routinely diagnosing autoimmune blistering diseases with DIF [16]. It should be stressed that identifying exclusively C3 deposits should not be regarded as diagnostic since C3 activation may be triggered without antigen-antibody binding. Artificial intelligence approaches would provide help for interpreting subjective DIF images.
In a minimal laboratory workup of pemphigus, DIF should be combined with serum biochemical-molecular testing for circulating antibodies against DSG1 and DSG3. Nowadays, the multiplex approach to this issue is gaining popularity and two techniques following their validation in various ethnicities, namely multiplex ELISA and multiplex indirect immunofluorescence are available [17–19].
Aim
Our aim here was to explore the issue of the relationship between detecting IgG deposits in DIF and revealing IgG antibodies to particular DSG isoforms in ELISA techniques in treatment-naïve patients with pemphigus diagnosed in a single tertiary referral centre, and to discuss obtained data in relation to the pertinent literature on etiopathogenesis of pemphigus. Our novel observation is that, in the light of our monocentre experience, the IgG tissue deposits in treatment-naïve patients with pemphigus diseases are accompanied by serum IgG antibodies to DSG1 rather than to DSG3. DSG1 may bind IgG more efficiently than DSG3 since DSG1 has a longer cytoplasmic region compared to that of DSG3.
Material and methods
Patients
The retrospective analysis of documentation of patients with pemphigus seen in a single tertiary referral centre in the years 2016–2020 revealed 19 treatment-naïve patients (11 females, 8 males; age range: 39–88 years; mean age: 63 years) who exhibited pemphigus IgG deposits, accompanied by other types of immunoreactants in various combinations, with DIF of perilesional tissue. This IgG deposition-positive group studied in detail consisted of 9 (47.37%) PF, 9 (47.37%) mucocutaneous PV (mcPV) and 1 (5.26%) mucosal-dominant PV (mPV) cases. The patients with IgG antibodies against DSG1 and IgG antibodies against DSG3 served as mutual control subgroups.
In total, 56 individuals with newly diagnosed pemphigus diseases, who had pemphigus-compatible DIF readings, were identified in those years. In detail, 14 (25.00%) PF, 24 (42.86%) mcPV and 18 (32.14%) mPV cases were diagnosed. All patients had ELISA results corresponding to their pemphigus diseases. Thirty-seven patients exhibited in DIF pemphigus IgG4 and/or IgG1 deposits, but not IgG deposits. This IgG deposition-negative group consisted of 5 (13.51%) PF, 15 (40.54%) mcPV and 17 (45.95%) mPV cases. Therefore, these patients were not evaluated in depth in our study, except for statistically analysing the proportions of PF and mPV patients in IgG deposition-positive and IgG deposition-negative groups.
Direct immunofluorescence procedure
DIF of perilesional tissue was performed in all cases for detection of not only conventionally evaluated IgA, IgM, IgG and C3 deposits, but also for detection of IgG1 and IgG4 deposits. The experience of our and other laboratories is that images of IgG4 deposition can be more suitable for unequivocal interpretation compared with images of standard immunoreactants including IgG [11, 20]. For DIF staining, 4 µm cryostat sections of perilesional skin/mucosa were cut. The tissue sections were incubated in a humid chamber for 30 min at room temperature (RT) with commercially available fluorescein isothiocyanate (FITC)-conjugated rabbit polyclonal antibodies against the human IgA, IgM, IgG (code No. F 0202) and C3 (Dako, Denmark) and FITC-conjugated mouse monoclonal antibodies against human IgG subclasses: IgG1 and IgG4 (clone HP-6025) (Sigma-Aldrich, USA). The antibodies were used at a working dilution of 1 : 100 in phosphate buffer saline (PBS). The samples were then washed in PBS (pH 7.2) at RT for 15 min with gentle agitation. Then, slides were coverslipped and examined by up to three independent observers to minimize the subjectivity of evaluation. Up to three fluorescence microscopy systems were used for visualization, namely (i) the EuroStar III Plus microscope operated by Bluelight LED technology (Euroimmun, Germany), (ii) the ZEISS LSM510 system with Axiovert 200M laser-scanning confocal microscope (LCSM) (Carl Zeiss Jena GmbH, Germany) and (iii) short arc mercury lamp-operated microscope (BX40, Olympus, Japan). The LSCM analysis was done courtesy of the Electron and Confocal Microscopy Laboratory, Faculty of Biology, Adam Mickiewicz University, Poznan, Poland. The intensities of deposits on the slides were graded using the semiquantitative 4-point scale (from “-” to “+++”) at the objective magnification 40×. These fluorescent microscopy systems did not alter the readings in terms of detecting the types of immunoreactants and judging their intensity.
ELISA procedures
Two commercially available ELISA systems were used for the detection of serum IgG antibodies (Euroimmun; Lübeck, Germany), namely the monoanalyte ELISAs or the multiplex ELISA. The monoanalyte ELISAs utilized DSG1 and DSG3 recombinant proteins consisting of 5 subdomains of the extracellular domain of those molecules (manufacturer’s cut-off value of 20 RU/ml), whereas the multiplex ELISA comprised of 6 different antigens (BP180, BP230, DSG1, DSG3, envoplakin, type VII collagen). The recombinant DSG1 and DSG3 used in the latter system were the same as those in the monoanalyte ELISAs. Therefore, the results of the detection of IgG antibodies to DSG1 and DSG3 defining pemphigus diseases were not influenced by the ELISA system used. In multiplex ELISA each antigen was coated in a separate well and a semiquantitative evaluation was carried out with the manufacturer’s cut-off ratio of 1.
All measurements were made in the ELISA plate readers (Asys Expert 96 or Ledetect 96) equipped with MikroWin 2000 software by a single operator following the manufacturer’s instructions.
Statement of ethics
Study approval statement: This study protocol was reviewed and approved by the local Polish Ethical Committee of the Poznan University of Medical Sciences, approval number 560/15.
Statistical analysis
The proportions of PF and mPV patients in IgG deposition-positive and IgG deposition-negative groups as well as the relationship between detecting IgG deposits in DIF and revealing IgG antibodies to particular desmoglein isoforms in ELISA techniques was assessed with the Z test for two independent proportions using PQStat version 1.8.2.238 (Plewiska, Poland). A p < 0.05 was arbitrarily considered statistically significant.
Results
A statistically significantly higher proportion of PF cases in the IgG deposition-positive group (9 of 19, 47.37%), compared to that in IgG deposition-negative group (5 of 37, 13.51%) (p = 0.0145) was noticed. A statistically significantly lower proportion of mPV cases in the IgG deposition-positive group (1 of 19, 5.26%) compared to that in IgG deposition-negative group (17 of 37, 45.95%) (p = 0.0054) was observed.
The detailed results of DIF and ELISA examinations in the IgG deposition-positive group of 19 patients are presented in Table 1.
Table 1
Detailed results of DIF and ELISAs in the IgG deposition-positive group of 19 pemphigus patients
| IgG antibodies to desmoglein isoforms in ELISA | Immunoreactants in DIF, N | ||||
|---|---|---|---|---|---|
| IgG, IgG1, IgG4 | IgG, IgG1, C3 | IgG, IgG4, C3 | IgG, IgG1, IgG4, C3 | IgG, IgG1, IgG4, IgA, C3 | |
| DSG1 | 1 | 1 | 2 | 4 | 1 |
| DSG3 | 1 | 0 | 0 | 0 | 0 |
| Both DSG1 and DSG3 | 0 | 3 | 3 | 3 | 0 |
Among 19 pemphigus patients with IgG deposits, serum IgG antibodies against DSG1 were detected in 18 patients, whereas serum IgG antibodies against DSG3 were found in 10 patients. The statistical analysis showed that the proportion of IgG anti-DSG1 antibody-positive individuals (18 of 19, 94.74%) was statistically significantly higher than the proportion of IgG anti-DSG3 antibody-positive ones (10 of 19, 52.63%) (p = 0.0099). In none of the patients IgG there were exclusive deposits. Just 1 patient having serum IgG antibodies against DSG3, but not against DSG1, diagnosed as mPV had IgG deposits in DIF.
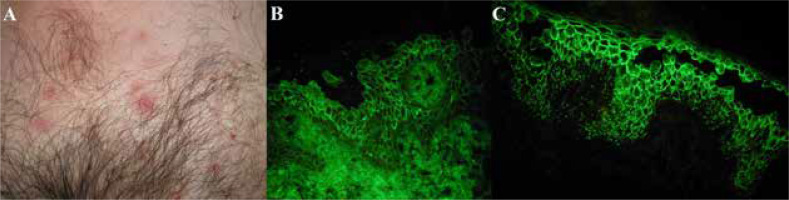
Clinical and DIF features of a representative IgG deposition-positive case are shown in Figure 1.
Figure 1
Seborrhoeic variety of pemphigus foliaceus in a middle-aged male having a markedly elevated level of IgG serum antibodies against DSG1, but a level of IgG serum antibodies against DSG3 is within normal range, with monoanalyte ELISAs. Inflammatory macules, some of them covered with yellowish scales, on the suprapubic skin (A). IgG (+) pemphigus deposits (B), and IgG4 (+++) pemphigus deposits showing dew drops on a spider web appearance (C) in DIF of perilesional skin visualized with the short arc mercury lamp-operated microscope (the original objective magnification 40×)
Discussion
The detection of IgG tissue deposition in pemphigus diseases is regarded as diagnostically obvious, but in our opinion requires more scrutiny in the light of the pathogenic IgG4-mediated autoimmune response in pemphigus [21]. In this context, the importance of IgG4-related disease in wider human pathology should be mentioned [22]. Of note, our study revealed a statistically significantly higher proportion of PF cases in the IgG deposition-positive group compared to that in IgG deposition-negative group (p = 0.0145). Conversely, a statistically significantly lower proportion of mPV cases in the IgG deposition-positive group compared to that in IgG deposition-negative group (p = 0.0054) was noticed. Despite the fact that the number of patients studied is quite small, we are convinced that the statistical analysis used enables reliable interpretation of the results. The fact that individual pemphigus lesions undergo spatial-temporal evolution may provide an explanation for discrepancies noticed by us. Simply, tissue samples for DIF may have varying tissue architecture and the distribution of desmosomal components. Such diverse topography may influence DIF results. Also, DIF results obtained in various laboratories may be prone to inconsistencies due to the variable sensitivity of FITC conjugates used. This hampers multi-centre standardization of laboratory procedures. Therefore, for the sake of clarity, clone/code numbers of the key anti-IgG and anti-IgG4 conjugates used by us were given. Interestingly in this context, Lehman et al. [20] have recently examined 12 biopsies sent to a pathology laboratory to perform DIF from patients with clinical features suggesting pemphigus diseases. They detected unequivocal IgG and C3 pemphigus-type deposits in only 4 biopsies. Equivocal IgG and C3 deposits were detected in as many as 8 biopsies. Evaluating IgG4 deposits in those 8 biopsies gave 6 positive results. Thus, their results are similar to ours. Nevertheless, they did not attempt to analyse their results in relation to the profile of serum IgG antibodies to DSG isoforms as we did. Anjali [23] compared the results of DSG1/3 ELISA and DIF in PV patients in clinical remission. It was revealed that the majority of such patients having positive DIF results for IgG and/or C3 were positive for anti-DSG3, but not anti-DSG1, serum antibodies. This is a contradictory observation to our data on treatment-naïve patients with pemphigus diseases. During long-term follow-up very high levels of anti-DSG3 antibodies may be observed in certain PV patients in clinical remission [24]. These discrepancies may be explained by the assumption that dynamic immune response phenomena depend on the stage of pemphigus diseases.
Still, the exciting pathological question, putting aside the limitations of laboratory methods used, is why IgG deposition in tissue of treatment-naïve patients appears to be related to the presence of serum IgG antibodies against DSG1 rather than against DSG3. We hypothesize that the pemphigus-specific explanation for this phenomenon may be the fact that DSG1 has a long cytoplasmic region compared to the DSG3 which has it remarkably truncated. Specifically, DSG1 has 5 repeat unit domains (RUD), whereas DSG3 just 2 [25]. Because of that DSG1 on keratinocytes having their surface architecture altered by the ongoing autoimmune response may bind IgG more efficiently and durably than DSG3, as well as maintain lasting downstream signalling. Ca2+-mediated signalling is important for epidermal blistering and dependent on the autoantibody profile, which indicates different roles for signalling complexes organized by DSG1 and DSG3 [26]. Still, intrakeratinocyte signalling pathways [27, 28] may be disease-nonspecific being induced by diverse triggering stimuli. Perhaps, the initiating action of IgG4 antibodies, which were reported to be predominantly detectable in active pemphigus, may facilitate subsequent more robust binding of IgG antibodies [29].
It was also suggested [30] that upon pemphigus IgG binding, DSG3 is internalized and degraded. IgG probably disrupts the assembly of functional desmosomes through rapid internalization and degradation of DSG3, which is mediated by the DSG3 cytoplasmic tail. However, the data still do not provide sufficient evidence for such a degradation pathway for DSG1 in PF patient skin. Conceivably then, the IgG-containing endosomes in PF remain in the cell and are finally lost by desquamation [31].
Our clinical experience is that PF, a disease mediated by antibodies against DSG1, but not DSG3, generally expresses more cutaneous inflammation than PV. One of the reasons for that may be that the binding of IgG against DSG1 may trigger a more prominent inflammatory response than the binding of IgG4. Finally, according to our monocentre experience, it seems that tissue binding of IgG is more prominent in treatment-naïve patients with pemphigus diseases showing cutaneous rather than mucosal involvement.
Conclusions
Analysing the results of established imaging and biochemical-molecular techniques used for routine laboratory diagnostics of pemphigus diseases, which, however, utilize peculiar reagents can provide potentially useful data for expanding our knowledge of the pathogenesis of these illnesses. The above speculations trying to interpret why IgG deposition in tissue seems to be related to the presence of serum IgG antibodies against DSG1 rather than against DSG3 require experimental verification ideally on a multicentre basis using as technologically advanced approaches [32] as feasible.









