Approximately 20–25% of deaths due to trauma in the first four decades of life result from thoracic trauma [1]. Penetrating thoracic trauma is present in 33% of deaths due to thoracic trauma [2]. in a 1972 autopsy study involving 1178 trauma patients, 82% of the patients with tracheobronchial injuries died at the accident site; 30% of those who could come to the hospital also died. it was observed that 50% of these losses occurred in the first hour [3].
Between 2001 and 2005, a total of 1033 tracheal injuries were reported over five years in Germany [4]. According to the US Centers for Disease Control and Prevention (CDC), while the medical costs of approximately 12 000 patients who died in 2005 due to firearm assault injury were more than $60 million, it was reported that the loss of work and productivity was 18 billion dollars. Between 2000 and 2005, the average hospital cost for adults over the age of 18 who had a penetrating injury was estimated at £7983 in the UK [5]. it was observed that mortality in thoracic trauma patients was 36% before 1950 and decreased to 9% after 1970 [6].
This article aims to review the current knowledge and treatment algorithms regarding such injuries.
HISTORY
The first published tracheobronchial injury case appeared in the medical literature in 1848 as an autopsy finding of a male patient who died after a carriage accident causing a left main bronchus rupture. The first surgically treated case, by Hotz in 1913, was a left main bronchus rupture in a 2-year-old girl who had been trapped under a train carriage. Then came the first published long-term survival report by Krinitzki [3] in 1927 of a 30-year-old woman who had experienced rupture of the left main bronchus after a tracheo-bronchial injury 20 years earlier. Nissen successfully performed a left pneumonectomy on a 12-year-old girl in 1931 for traumatic stricture in the left main bronchus. Hood and Sloan [7] published their series of 7 cases in 1959 and argued that primary repair of the trachea is a method that is technically applicable and preferable in emergencies. Two years later, Shaw [8] published his article defending primary repair as a treatment strategy in tracheobronchial injuries.
ANATOMY
The cardiac box margins are formed by the clavicle above, xiphoid below, and middle clavicular line laterally. Main structures, such as lungs, major airway structures, heart, major vessels, and spinal cord, are located in the thorax. Trauma in this area is associated with the risk of penetrating heart injury, cardiac tamponade, and rapid development of clinical decompensation. while the risk of death in injuries with a sharp object is normally 1–8%, it reaches 25–28% when it includes the cardiac box, and most of those patients die before they can reach the hospital [9]. Mortality rates were 8% for the left main bronchus, 16% for the right main bronchus, and 26% for the trachea [10].
The trachea can be examined in two parts, cervical and thoracic. The cervical trachea refers to the part of the trachea above the sternal notch. The thoracic part of the trachea is 5–8 cm in lenght and is defined as the trachea from the sternal notch to the carina (approximately at the level of the 4th thoracic vertebra). when the head is being fully extended, almost the entire trachea slides in the mediastinum, and 1/2 to 2/3 of it rises above the jugular notch.
Ischemic complications during airway resection are rare, as the trachea is supplied segmentally from the innominate-subclavian system by the inferior thyroid artery and bronchial arteries [11].
DEFINITION AND INCIDENCE
Penetrating thoracic injury is defined as an injury that develops in a confined area between the lower neck and lower costal margin and penetrates the body through the skin barrier. Tracheobronchial rupture is defined as traumatic injuries in the cricoid cartilage and of the trachea and main bronchi. Pene trating trauma is usually a result of a firearm or stab wound. Non-penetrating trauma, on the other hand, is caused by blunt trauma mechanisms such as motor vehicle accidents, falls, bumps, explosions, and burns and mainly occurs in the distal trachea and right main bronchus [12].
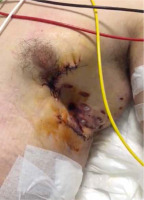
The majority of thoracic trauma cases are detected as blunt trauma. Blunt traumas usually affect mediastinal-thoracic trachea; however, approximately 75% to 80% of penetrating airway injuries are detected in the cervical trachea [13]. The probable reason for this is that the mediastinal trachea is located quite deep. However, when trauma affects the mediastinal trachea, it can be more acute and life-threatening since it also affects large vascular structures at the same time (Figure 1).
FIGURE 1
35-year-old male patient with penetrating injury. Stab wound 8 cm inferior to the left nipple
While less than 10% of blunt trauma cases require an operation, surgical intervention is necessary in as many as 15–30% of patients with penetrating injury [14]. Additionally, it was observed that the incidence of airway injury in patients with penetrating chest trauma is below 1–2% [15]. in 1998, Rossbach et al. [16] reported that airway trauma was 75% more frequently seen in the male gender. A review on the studies in the literature revealed that a significant portion of the studies were designed retrospectively since the patients with penetrating injuries died before admission to a hospital and the mortality rate of these cases was higher; furthermore, the male/female ratio was as high as 24 : 1 [17]. According to the examined studies, the mortality rate varied between 2.8 and 56.9%, not depending on whether the treatment was conservative or surgical. The mean age of patients ranged from 23.6 to 37.8 years. The most commonly seen injury mechanism was stab wound with a rate of 96.4% (Table 1) [17–33].
TABLE 1
MORPHOLOGICAL CLASSIFICATION OF TRACHEAL INJURY
The anterior trachea has been identified as the most frequently injured area during tracheo-bronchial injury, including the cartilage or ligamentous parts between the tracheal rings. The majority of tracheobronchial injuries were classified according to oesophageal injury along with mediastinitis; they usually appear 1 to 2.5 cm inside the carina, and more than 85% of these injuries are injuries of the common bronchus (Table 2) [34].
TABLE 2
Morphological classification of tracheal injury [34]
Life-threatening injuries present with airway obstruction, tracheobronchial injury, pneumothorax, haemothorax, and cardiac tamponade (Figure 2).
CLINICAL FINDINGS
The damage is proportional to the speed, energy, and characteristics of the weapon as well as the tissue penetrated. Generally, the findings are non-specific, but 15–30% of the total number of cases require surgery [35]. Penetrating airway trauma usually presents with inability of pulmonary expansion and severe air leakage even after insertion of a thorax tube. Severity of the injury is associated with size of the penetrating object and deformation capability and intensity of the exposed tissue. For example, high-velocity weapons result in more extensive damage to the soft tissues. Penetrating chest trauma can be divided into two main categories, low velocity and high velocity, depending on the speed of the penetrating object. A stab wound is an example of a low-velocity injury. High-velocity injuries include injuries from bullets fired from firearms. The identification of a high-velocity gunshot trauma is vital since the adjacent structures on the bullet’s path can possibly get injured. Additionally, it should be kept in mind that the path of the bullet is not always straight, as structures can alter their paths within the thoracic cavity.
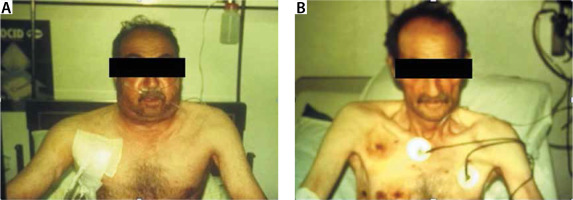
Clinical findings in tracheobronchial injuries are often nonspecific, since a 50% reduction in tracheal diameter causes dyspnoea with only significant effort. Dyspnoea and stridor at rest are observed if the tracheal lumen diameter falls below 25% [36]. Other early clinical findings include haemoptysis, localised pain, pneumothorax, subcutaneous emphysema, hoarseness, stridor, progressive dyspnoea, tachypnoea, and Hamman’s sign [37]. Hamman’s sign is a crackling noise synchronised with heartbeat and breathing, and air is seen in the mediastinal area. late clinical findings include dyspnoea, stridor, and infections of the distal lung parenchyma [38] (Figure 3).
FIGURE 3
56-year-old male patient with gunshot. A) A right anterior chest tube in a patient with subcutaneous emphysema. B) The chest tube is terminated after emphysema regression
Injuries co-existing with penetrating thoracic trauma are left recurrent laryngeal nerve (8%), abdomen (18%), spinal (7%), heart (5%), large vessel injuries (18%), oesophagus (11%), haemopneumothorax (32%) [39]. Therefore, it should be assumed that any penetrating injury to the chest may cause internal organ damage, including the heart, lungs, tracheo-bronchial tree, oesophagus, large vascular structures, diaphragm, and spinal cord [34] (Figure 4).
FIGURE 4
A thoracic deformity due to multiple rib fracture after blast trauma and multiple shrapnel wounds
The management and consequences of penetrating thoracic trauma are mainly proportional to the extent of the injury to internal organs, namely respiratory failure resulting from airway obstruction and bleeding. Also, the skills of the clinicians play an important role in the patient outcome here. Hypoxia, hypovolaemia, and direct effects on thoracic organs lead to decreased cardiac output in these patients. Stability criteria in the patient with penetrating thoracic trauma are accepted as systolic blood pressure of > 95 mm Hg, heart rate of < 120 beats min-1, oxygen saturation of > 95%, Glasgow Coma Scale of ≥ 14 (Table 3) [40].
TABLE 3
Stability criteria in a patient with penetrating thoracic trauma [40]
| Systolic blood pressure | > 95 mm Hg |
| Heart rate | < 120 beats min-1 |
| Oxygen saturation | > 95% |
| Glasgow Coma Scale | > 14 |
DIAGNOSIS
Rather than using a single diagnostic method, evaluation should include a combination of clinical examination, X-ray radiography, bronchoscopy, and CT assessment. if there is a strong clinical suspicion, definitive diagnosis is possible in more than 90% of the cases with the combination of chest X-ray and early bronchoscopy [41].
Findings on X-ray radiography can reveal pneumomediastinum, fallen lung, right main bronchus rupture, and subcutaneous and deep cervical emphysema [42]. However, in 10–20% of the patients, no specific finding is present in X-ray radiography. The “fallen lung sign” reflects the tendency of the separated lung to collapse down from the hilum and from the back to the side instead of collapsing inward as in other pneumothorax cases, and is regarded to be a pathognomonic finding in damage to the main bronchus [43]. Furthermore, airway injury should be suspected when an air bubble is detected inside the artificial airway [44].
COMPUTED TOMOGRAPHY AND BRONCHOSCOPY
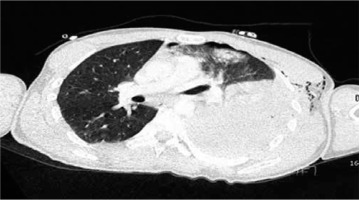
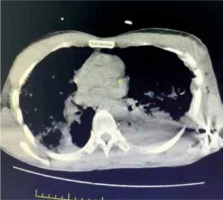
CT diagnostic success is 71%; however, with the use of multi-channel multi-detector CT, the sensitivity of CT imaging in the diagnosis of tracheobronchial injuries has increased to 94% [45]. Thorax CT is better than chest radiography in diagnosing main bronchus, lobe, or segment rupture [46]. Subcutaneous haematoma, subcutaneous air surrounding the trachea, discontinuity of the tracheobronchial tree, focal tracheal defect, tracheal wall deformity, herniation of the endotracheal tube (eTT) cuff more than 2.8 cm outside the tracheal wall, and focal intimal bronchial flap in the lumen are diagnostic. The characteristic signs of tracheal rupture on CT are: gas distribution around the fractured ends (59%), bronchial lumen stenosis or obstruction (50%), bronchial displacement, or angular deformity (50%) [47] (Figure 5).
FIGURE 5
Computed tomography imaging of a patient with penetrating thoracic trauma; bullet core, haemothorax and accompanying lung contusion and atelectasis on the left
Longitudinal tracheal rupture can be observed by virtual bronchoscopy. Regular bronchoscopy is preferred to detect the location, expansion, and depth of the rupture and to make sure the eTT cuff is inflated beyond the injured area. However, the vision may sometimes be obstructed by the presence of blood clots and tissue debris, and if there is critical airway stenosis, the bronchoscope itself may increase airway obstruction [15].
TREATMENT
The purpose of providing an adequate airway is to improve ventilation and reduce air leakage to surrounding structures by placing a cuffed eTT distally to the site of injury. Penetrating airway injuries can rapidly progress to airway obstruction and respiratory insufficiency [48]. Therefore, the concept of the “golden hour”, has been suggested to emphasise the importance of rapid and effective treatment of acutely injured patients [49]. According to most studies, mortality improves the most if the time from the moment of trauma to the time of primary repair is shorter than 60 minutes [50, 51]. while the first observations of the association between time to treatment and mortality in haemorrhagic shock were reported by Santy during the First world war, the concept and term “golden hour” was coined by Cowley almost sixty years later, in 1976 [49, 51, 52].
Injuries of the distal trachea, carina, and proximal main bronchi are extremely challenging to manage. The main goal is ensuring airway continuity.
Therefore, the anaesthesiologist and the surgeon should work together for successful treatment. ventilation and/or oxygenation can be difficult in patients with penetrating airway injury, even after tracheal intubation and mechanical ventilation support. in these cases, more invasive advanced oxygenation techniques such as extracorporeal membrane oxygenation (eCMO) can be used [53].
PRIMARY EXAMINATION
Resuscitative thoracotomy can be performed in patients with cardiopulmonary arrest. if the patient is haemodynamically unstable and has respiratory distress, the possibility of tension pneumothorax, massive haemothorax, and cardiac tamponade should also be considered, and resuscitative thoracotomy should be performed when necessary. life-threatening injuries need systematic team management with adequate evaluation and resuscitation of respiratory, cardiovascular, and neurological disorders. The steps that should be considered at primary examination include keeping the saturation between 94% and 98% through high-flow oxygen by ensuring the cervical stabilization, preparation of cross-match-compatible blood products, blood collection for complete blood count, biochemical analy sis, and blood gas analysis; ensuring wide intravenous access, providing chest and pelvic X-rays of the patient, achieving early analgesia, and focused sonography (FAST) evaluation for the trauma [54].
In cases where the cardiac arrest occurs after penetrating injury, respiratory tract stabilization with endotracheal intubation, bilateral chest tube insertion, and cardiopulmonary resuscitation with urgent thoracotomy, pericardiotomy, and cross-clamp placement to the aorta are required. if the respiratory sounds are absent unilaterally, the neck veins are distended, and the tracheal shift is visible on chest X-ray, the definite diagnosis of tension pneumothorax can be made. in such a case a chest tube should be inserted. Temporary needle thoracostomy may also be considered to gain time.
Massive haemothorax can also lead to the absence of respiratory sounds (on the affected side). Blood pressure should be optimised by rapid fluid resuscitation, and a chest tube should be placed. if initial drainage is > 1500 ml or drainage continues for 2–3 hours as > 200 ml h-1 (> 150 ml h-1 for elderly patients), the patient should be taken directly to the operating room. Administration of positive end-expiratory pressure (up to 10–15 cm H2O) without increasing air leakage may be considered in order to reduce bleeding in patients who develop massive haemothorax [9].
In cardiac tamponade, distention of the neck veins, quiet heart sounds and cyanosis can be observed. if FAST cannot be performed, subxiphoid pericardiocentesis should be performed, and if the result is positive, the patient should be transferred to the operating room immediately for thoracotomy or median sternotomy.
In a patient with penetrating airway trauma, respiratory distress symptoms such as tachypnoea, hypoxia, altered consciousness, pneumothorax, and the presence of shock signs such as tachycardia, hypotension, as well as decreased pulse pressure should be evaluated in detail. if there is time and the patient is cooperative, standard airway evaluation should be applied. Although the management is easier if the patient is cooperative, the risk of aspiration should always be considered. The mechanism of the trauma and whether it is penetrating or blunt should be determined. Concomitant injuries in the head, neck, spinal cord, thorax, or skeletal region and the location and degree of airway damage should be evaluated [55]. Awake fibreoptic bronchoscopy should be considered in patients whose general condition is stable [56]. Placing a tracheal tube beyond the vocal cords in patients with airway transection may cause various results such as enlargement of the injury, a simple airway laceration, airway obstruction, pneumomediastinum, and the formation of a false passage. Therefore, in this group of patients, maintaining spontaneous ventilation of the patient by preserving airways integrity is important, and hence intubation by conventional methods may increase the risk of tracheal rupture. Fibreoptic intubation should be considered.
Positive pressure ventilation can alter the placement of an endotracheal tube (eTT) or even cause a complete transection by itself, so it should be avoided [57].
Low tidal volume, permissive hypercapnia ventilator strategies are required until the surgery is performed [15]. if there is severe airway damage and bleeding, rigid bronchoscopy may be required, but it is contraindicated in patients with an unstable cervical spine [58].
If the patient is not cooperative, multidisciplinary approaches should be applied depending on the level of injury. video-laryngoscopy should be used in combination with rapid sequence induction in supralaryngeal injuries. in infra-laryngeal or tracheal injury, rapid sequence induction with rigid bronchoscopy or fibreoptic intubation should be performed. A double lumen tube, bronchial blocker, or wilson tube should be used in bronchial injuries. in cases where emergency tracheostomy is required, eCMO or cardiopulmonary bypass can be considered as an alternative method [59].
Single lung ventilation via selective intubation of the left or right main bronchus can be considered in cases where the tracheal rupture extends to the carina [34]. endobronchial blockers can be used to provide ventilation in the absence of a double-lumen tube.
The first publication on jet ventilation in a case with tracheal injury was published in 1986; in a patient with an 8 cm tracheal separation involving the right main bronchus, the cuff of eTT (size 7) was not inflated and placed in the left main bronchus with single lung ventilation; the cuff was also not inflated in the post-operative period in order to accelerate the healing process [60]. in a case report published by Naghibi et al. [61] in 2003, two intrabronchial catheters were placed directly into the open chest by the surgeon, and a high-pressure oxygen-containing jet ventilator was attached to each catheter. As a result, the patient’s peripheral oxygen saturation could be increased from 60% to 100% with hand-triggered jet ventilation with a respiratory rate of 12–20 per minute. The flexibility and small diameter of catheters during jet ventilation allow for effective ventilation during airway reconstruction in haemodynamically stable patients. in addition, jet ventilation makes it easier for the surgeon to suture the tracheal injury [34]. extracorporeal membrane oxygenation or cardiopulmonary bypass may be considered in patients with airway trauma in whom conventional ventilation is not possible, the damaged tracheal particle forwarded to the distal side, and the airway opening could not be protected.
In a 2018 case presentation by Carretta et al. [53], eCMO support was used in a patient with an acute blunt traumatic tracheal injury. in another study published by Aprile et al. [62], it was stated that femoro-femoral cannulation is mandatory in thoracic trauma since the neck region cannot be preferred for cannulation.
Continuation of eCMO treatment may be considered during the postoperative period in order to reduce the barotrauma of the airway and lungs. Although veno-venous eCMO is an effective way to maintain adequate gas exchange, veno-arterial eCMO may be required to provide haemodynamic stability during surgery [53, 62]. Prevention of nonluminal ventilation, improvement in the exploration of the surgical field, reduction of the need for anticoagulation with increasing biocompatibility, reduction of the post-operative high-pressure ventilation requirement, facilitation of airway repair, and early extubation are among the advantages of using eCMO during tracheal surgery.
All necessary equipment and personnel for an emergency tracheostomy should be available before manipulation of the injured airway. if endotracheal intubation cannot be achieved, emergency tracheostomy is the only proper option. in the presence of tracheal injuries, the tracheostomy line should preferably be located in the damaged area in order to preserve the normal trachea, which aims to facilitate surgical repair of the trachea [63].
All in all, there may be little to no time for airway evaluation. if this is the case, the anaesthesiologist should manage the patient according to the worst-case scenario and consider the possibility of airway trauma along with possible investigations and airway interventions that are required. This includes evaluating what personnel and equipment are needed, and especially whether tracheostomy or surgical cricothyroidotomy will be performed [64].
SURGICAL TREATMENT
In a study from 2005 including 71 patients, it was reported that 60% required emergency airway due to tracheobronchial trauma, standard orotracheal intubation was required in 75% of the patients, orotracheal intubation under the guidance of flexible bronchoscopy was required in 10%, intubation from the open cervical wound was required in 10%, and tracheostomy was required in only 5% [63]. Minor tracheal wounds usually heal spontaneously within 48 hours or can be treated non-operatively by temporary orotracheal or nasotracheal intubation.
Surgical treatment methods performed depending on the regions of the trachea and bronchial tree in traumatic injuries are shown in Table 4. while airway injury is level i and ii, clinically stable patients who have spontaneous ventilation or require minimal ventilator support, have tracheal ruptures with a length of 2 cm or less, without oesophageal damage, minimal mediastinal air, and non-progressive pneumomediastinum or subcutaneous emphysema may be treated conservatively [34].
TABLE 4
Surgical treatment methods applied depending on the regions of the trachea and bronchial tree in traumatic injuries [34]
Patients with worsening subcutaneous emphysema, progressive pneumomediastinum, pneumothorax with persistent leakage, persistent pneumothorax despite the thoracic tube, or oesophageal wall prolapse towards the tracheal lumen should undergo urgent surgery.
NON-SURGICAL APPROACHES
In recent years, minimally invasive methods have become more preferred in the management of patients with a tracheal injury who are under high surgical risk due to the severity of the underlying disease and comorbidity. The first of these methods is the “watanabe spigot” silicone tube with a conical shape and protrusions on the sides to hold onto the bronchial wall designed by a Japanese pulmonologist in three different sizes (5, 6 and 7 mm) [65]. Such devices were originally introduced in clinical practice to treat persistent pneumothorax with pulmonary fistula and prolonged air leaks.
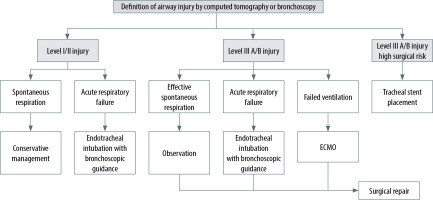
The temporary use of a self-expanding metallic stent (SeMS) is another minimally invasive treatment method with advantages in tracheal injury patients. The stent mechanically provides blockage on the defected tracheal area in addition to an effective inflammatory response with granulation tissue formation, which can potentially increase the closure of the tracheal defect. The stents can be and should be removed and extracted after adequate recovery (4–6 weeks) since infection, stent migration, metal fatigue, granulation tissue formation leading to tracheal stenosis, and mucus occlusion can be seen due to long-term stent placement [34] (Figure 6).
Consequently, clinical suspicion is important in early diagnosis in patients with thoracic trauma. Blind intubation should not be performed. Bronchoscopy is considered the gold standard in diagnosis. Primary surgical repair should be done as soon as possible to prevent infections. eCMO can be very useful in providing effective gas exchange in tracheobronchial injuries where traumatic ventilation is not possible. Multidisciplinary teamwork is vital for the safe provision of the airway.