Introduction
Type 1 diabetes mellitus (DM1) is a chronic autoimmune disease characterized by insulin deficiency and resultant hyperglycaemia [1]. The most common short-term complications of DM1 include hypoglycaemia, hyperglycaemia, and diabetic ketoacidosis (DKA). Long-term complications of diabetes include retinopathy, nephropathy, and neuropathy [2].
In the pathogenesis of DM1, chronic inflammation plays an important role in the development and progression of diabetes [3]. Accumulative evidence suggests that chronic activation of pro-inflammatory pathways in target cells of insulin action may contribute to obesity, insulin resistance, and related metabolic disorders, including type 2 diabetes mellitus (DM2). The identification of potential pathways connecting inflammation to diabetes has produced growing interest in targeting inflammation to help prevent and control diabetes and related conditions, as well as improving risk stratification for diabetes by using inflammatory biomarkers as potential indexes [4].
Early screening and ongoing medical care are essential to reduce long-term complications of diabetes [5]. Diabetic retinopathy (DR) is a diabetic microangiopathy characterized by microaneurysms, capillary non-perfusion, and ischaemia within the retina. Effective screening is a desirable method for the early detection and successful treatment for DR [6].
Diabetic nephropathy (DN) is one of the most frequent and serious chronic complications of DM1 and occurs as a result of microvascular lesions in the renal glomeruli [7]. Early detection of microalbuminuria and proper treatment may reverse or delay the progress of diabetic kidney disease [8].
Fibroblast growth factor 21 (FGF21) is a member of the endocrine FGF subfamily, which is expressed predominantly in the liver and stimulates glucose uptake through the induction of glucose transporter 1 (GLUT1) in adipocytes, therefore playing an important role in metabolic regulation. It is a stress-inducible hormone that plays important roles in glucose and lipid homeostasis as well as regulating energy balance, done through a hetero-dimeric receptor complex comprising FGF receptor 1 (FGFR1) and cofactor β-klotho [9]. FGF21 production in humans responds positively to consumption of glucose, and it is assumed that the serum FGF21 concentration is associated with glycaemic control. It can be used as a predictor or biomarker of diabetes because its serum level increases in obesity and diabetes [10].
Although FGF21 has lipid-lowering, anti-inflammatory, and anti-oxidant properties, its circulating levels are often elevated in different metabolic diseases, accounting for its involvement in glucose metabolism, energy homeostasis, atherosclerosis, inflammation, and oxidation [11]. Circulating FGF21 levels are elevated in obesity and patients with DM2, dyslipidaemia, and impaired glucose tolerance. It has been proposed that the elevated level of FGF21 (hyper-FGF21-nemia) in obesity-related disorders is attributed to FGF21 resistance, a phenomenon reminiscent of hyperinsulinaemia and insulin resistance. Although several previous studies have reported the elevated levels of FGF21 in type 2 diabetic patients, its role in DM1 remains unclear [12].
Aim of the study
This study assesses FGF21 levels in children with DM1, in comparison to controls, and correlates them with diabetes duration, glycated haemoglobin (HbA1c), and diabetic microvascular complications.
Material and methods
This cross-sectional study includes 50 children with DM1 recruited from the Paediatric and Adolescent Diabetes Unit, Ain-Shams University. They were compared to 50 matched controls. Ethical approval from Ain-Shams University Research Ethics Committee was obtained before the start of the study, and informed consent was taken from all patients and their legal guardians prior to enrolment. The study was performed in the period from January 2020 to December 2021.
The inclusion criterion was children with DM1 defined according to the International Society of Paediatric and Adolescent Diabetes (ISPAD) guidelines 2018 criteria [13]. The age group was between 5 and 16 years. Exclusion criteria were other types of diabetes, e.g. DM2 and MODY, and refusal from the patient’s caregiver.
All included patients were subjected to the following: full clinical history, including duration of diabetes, type and dose of insulin (IU/kg/day), and history of diabetic complications. A clinical examination was done including vital signs and anthropometric measurements with assessment of weight (kg), height (cm), and body mass index (BMI) Z-score [14]. A fundus examination was done by an expert ophthalmologist using a direct ophthalmoscope through dilated pupils for assessment of DR [15].
Laboratory investigations included mean blood glucose level (in the last month), and haemoglobin A1c (glycated haemoglobin) level using turbidimetric inhibition immunoassay (TINIA) for whole blood using a Cobasc311Auto analyser [16]. Serum level of FGF21 hormone was analysed using enzyme-linked immunosorbent assay (ELISA) kit supplied by Bioassay Technology Laboratory, Shanghai, China, with a detection range of 7 pg/ml – 1500 pg/ml and a sensitivity of 3.52 pg/ml [17].
The urinary albumin creatinine ratio (UACR) was measured using the immuno-turbidimetric method (Cobas Integra 800; Roche Diagnostics, Mannheim, Germany) [18].
Statistical analysis
All data were gathered, revised, and coded, and then entered to the Statistical Package for Social Science (SPSS), version 27 for Windows, SPSS Inc., Chicago, IL, USA. Quantitative data were presented as mean, standard deviation, and range when they were normally distributed, and it was presented as median with inter-quartile range (IQR) when their distribution was found to be non-parametric. Qualitative variables were presented as number and percentage. Comparison between groups with qualitative data was performed by using χ2 test, between data with parametric distribution by using an independent t-test, and for non-parametric data it was performed by using the Mann-Whitney test. Spearman correlation coefficients were used to assess the correlation between 2 quantitative parameters in the same group. Receiver operating characteristic curve (ROC) was used to estimate the best cut-off point for the FGF21 level with its sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and area under curve (AUC). The confidence interval was set to 95%, and the accepted margin of error was set to 5%. So, the p-value was considered at the level of < 0.05.
Results
Fifty children with DM1 with mean age 10.70 ±2.74 years were compared to 50 matched controls. Their HbA1c range was 6.3 – 15%, mean ±SD (8.48 ±1.49); UACR ranging from 0.9 to 348 mg/gm creatinine, median (IQR): 6.2 (2.7–17.3); insulin doses ranging from 0.5 to 2 units/kg/day, mean ±SD (1.08 ±0.32). Ninety-eight per cent of the studied children with DM1 were on basal bolus regimen, 2% on insulin pump, and only 4% of them had abnormal changes in fundus examination, as described in Table I. A comparison of clinicodemographic data of the studied children with DM1 and controls is described in Table II.
Table I
Clinical and laboratory data of studied children with DM1 (n = 50)
Table II
Comparison of clinicodemographic data of children with DM1 and controls
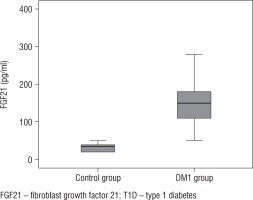
The median FGF21 level in children with DM1 was 150 pg/ml, range (50–350). The FGF21 level was significantly higher in children with DM1 than in controls (p < 0.001), as shown in Figure 1.
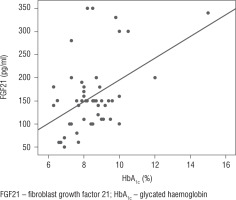
The FGF21 level was found to be significantly and positively correlated with diabetes duration, mean blood glucose level, and HbA1c (p < 0.001, p = 0.015, p = 0.018, respectively), as illustrated in Table III and Figure 2.
Table III
Correlation of FGF21 level with other studied parameters in children with DM1
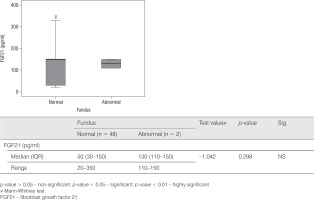
The FGF21 level was not significantly elevated in children with DM1 with diabetic nephropathy or retinopathy (p = 0.122, p = 0.298, respectively), as illustrated in Table III and Figure 3.
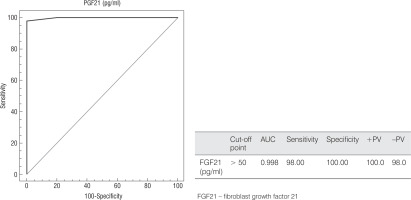
The ROC curve for the FGF21 level estimated a cut-off point of > 50 pg/ml to differentiate between children with DM1 and controls with 98% sensitivity, 100% specificity, 100% PPV, and 98% NPV, as shown in Figure 4.
Discussion
FGF21 is a circulating hormone with an important role in metabolic regulation. Its production in humans responds positively to glucose consumption and it is thought to be associated with glycaemic control [10]. The level of endogenous FGF21 is elevated in patients with impaired glucose tolerance, and it progressively increased as a compensation or response to the deterioration of metabolic imbalance [19]. Regarding the relation between FGF21 level and DM1, results are scarce and contradictory. The current study showed higher FGF21 serum levels in children with DM1, with significant positive correlation to diabetes duration and HbA1c. On the other hand, lower levels of FGF21 are reported in patients with DM1, in contrast to patients with DM2, when compared with healthy individuals in several studies. A study by Arking et al. assessed the hepatokine FGF21 in children and youths with DM1 and DM2 diabetes, and they found that FGF21 levels were higher in DM2 (113.1 vs. 70.6 pg/ml, p = 0.03) than DM1 [20]. Another study by Rosell et al. found insignificant correlation between FGF21 concentration and HbA1c, and that patients with DM1 had lower levels of circulating FGF21 as compared with the reference group, which was contrary to the present study, which showed significant positive correlation between FGF21 level and HbA1c in the DM1 group, who had higher FGF21 serum concentrations than the control group [21]. A study by Xiao et al. found that the level of FGF21 in DM1 is significantly lower compared with age- and sex-matched controls. They attributed the lower circulating FGF21 in patients with DM1 to a reduction in FGF21 production by the pancreas. However, the liver is the primary site for FGF21 production, while the pancreatic contribution of circulating FGF21 is largely unknown [12].
Glucose is considered as one of the major factors affecting the secretion of FGF21. A study by Yang et al. showed that the elevation of insulin levels under normal conditions inhibited the production of FGF21 [22]. On the other hand, Zibar et al. found no correlation between blood glucose and postprandial FGF21 with insulin treatment of DM1 patients. This result suggests that the reduction of circulating FGF21 in DM1 patients is due to the abnormal capacity for FGF21 production [23]. Another study by Gallego-Escuredo et al. showed that the FGF21 level is higher in obese diabetic patients, while after adjusting for BMI, the correlations of FGF21 levels with indicators of abnormal glucose homeostasis were not significant, concluding that although worsened in diabetic obese patients, obesity itself appears as the predominant determinant of the abnormalities in FGF21 levels [24].
A study by Lin et al. showed that, coinciding with the present study, the fasting blood glucose levels of the patients were significantly greater and the FGF21 concentrations were higher in the patients than those of the controls (p < 0.001), which showed significant positive correlation between blood glucose levels and FGF21 concentrations [25].
Microvascular and macrovascular complications are the leading causes of morbidity and mortality in patients with diabetes. A study by Jian et al. showed that circulating FGF21 is positively correlated with urinary albumin excretion, an indicator of microangiopathy, unlike the current study, which showed insignificant correlation between UACR and FGF21 level [26]. Another study by Esteghamati et al. showed that serum FGF21 levels were significantly elevated in patients with microalbuminuria (median [IQR]: 269.50 [188.50] pg/ml) compared to their normoalbuminuric diabetic peers (median [IQR]: 103.50 [75.75] pg/ml) and non-diabetic people (median [IQR]: 99.00 [126.75] pg/ml) [27]. Moreover, a study by Suassuna et al. stated that there was a positive correlation between serum FGF21 levels and urinary albumin excretion in T2D patients, where serum FGF21 levels progressively increased in parallel with the progression from normal to microalbuminuria and from micro to overt albuminuria, even before impairment of renal function becomes apparent [28].
FGF21 has not been extensively studied in DR, although there is evidence in other systems to suggest that it could be important in early detection of diabetic microvascular complications. A study by Rosell et al. found that DM1 patients with retinopathy had significantly lower FGF21 concentrations (10.0 ng/l) as compared with the healthy reference group (37.1 ng/l; p = 0.02) [21]. Another study conducted by Lin et al. used routine ophthalmic examination to show that serum FGF21 concentrations are higher in diabetic patients with DR than in those without DR, suggesting that serum FGF21 may play a role in the pathogenesis of DR, while the current study showed insignificant correlation between routine screening of DR by fundus examination and FGF21 level [25]. Another study by Heidari and Hasanpou found that serum FGF21 concentrations were higher in diabetic patients compared to controls and are higher in diabetic patients with DR than in those without DR [29]. Similarly, a study by Jin et al. showed that there was a significant difference in serum FGF21 level among 3 groups of diabetic patients; a first group with no DR, a second group with mild or moderate non-proliferative retinopathy (NPDR), and a third group with severe NPDR or proliferative DR (PDR); they observed a positive association between serum FGF21 level and DR (p < 0.01). A serum FGF21 level > 478.76 pg/ml suggested the occurrence of DR and a level > 554.69 pg/ml indicated sight-threatening DR (p < 0.01) [30].
Conclusions
FGF21 was significantly higher among children with DM1 than controls. However, no significant relation was found between it and diabetic retinopathy and nephropathy. This could be attributed to the small sample size of patients having diabetic retinopathy and nephropathy in the current study. Hence, large prospective studies are needed to elucidate the role of FGF21 in the development and progression of DM1 and its relation to diabetic microvascular complications.