Summary
Early effects of balloon aortic valvuloplasty (BAV) are satisfying, with high reduction of gradient and weaning of prostin and inotropes in patients with critical aortic stenosis; however, despite improvements in both technique and available technology, freedom from reintervention after BAV remains suboptimal. There remains no consensus regarding risk factors and procedural techniques that impact the long-term outcome of this procedure. Our study shows favourable early results of BAV, with good long-term survival. Aortic regurgitation remains a significant complication of BAV. Further research is needed to improve the procedural techniques and identify risk factors to improve long-term effects.
Introduction
Balloon aortic valvuloplasty (BAV) is considered as a first choice of treatment of isolated congenital aortic stenosis (AS) in some centres [1, 2], while in other centres the surgical approach remains the mainstay (especially in combined aortic valve (AV) disease) [3]. According to large, multicentre studies BAV provides satisfying results [2], comparable to a surgical approach [3, 4].
Despite improvements in both technique and available technology of BAV [2], long-term effects remain suboptimal [4]. Many studies have tried to identify risk factors of unsuccessful early outcome, freedom from reintervention, and mortality; however, the data acquired from those studies is inconsistent [1, 2, 5–7].
Moreover, the majority of published studies concerning BAV in congenital AS included results from all age groups [1, 8, 9]. Those studies predominantly focused on young children and teenagers [5, 10, 11], while studies focusing on BAV in neonates are limited [3, 12]. Analysing BAV in congenital AS among patients of all ages greatly enlarges the study sample, but makes the study sample heterogeneous, neglecting the aetiology, complex anatomy, periprocedural difficulties, and challenges of neonatal patients.
Aim
Our study reports short- and long-term outcomes of BAV in neonates from a single tertiary centre. The aim of the study is to identify risk factors and predictors of suboptimal early and long-term results of the procedure.
Material and methods
Study group
From 1998 to 2021 a total of 144 patients with congenital AS underwent primary BAV in our Clinic. BAV is the method of choice for treatment of isolated congenital AS in our centre. Patients with coexisting significant aortic regurgitation (AR) are referred for a surgical procedure. Informed consent was obtained from all parents before BAV.
Inclusion criteria for our study group were as follows: age ≤ 28 days at the time of the procedure and no prior AV intervention. Overall, 43 patients with a median age of 9 days (range: 1–28 days; 8 females) were included in our study. Their average weight was 3.34 ±0.56 kg.
BAV indications and technique
Indications for BAV were consistent with the American Heart Association statement from 2011 [13]. Indications for the procedure were as follows: valvar AS with peak-to-peak systolic valve gradient > 50 mm Hg in cardiac catheterization or > 50 mm Hg mean gradient in echocardiography study, and regardless of valve gradient in critical AS in 17 patients. Patients with moderate or severe AR were referred for surgery. Patients with features of HLHS (i.e. reversal of flow in ascending aorta, hypoplastic mitral valve, hypoplastic LV (LV long axis ratio to heart long axis ratio < 0.8, LVEDV < 20 ml/m2)) were qualified for univentricular correction and were not included in this study. In our study we identified a critical aortic stenosis as an AS resulting in reduced preprocedural EF and dependence on Prostin or inotropic infusion [8, 13, 14].
The procedures were performed under general anaesthesia and antibiotic prophylaxis. Vascular access was obtained through the femoral artery in 37 patients, the carotid artery in 5 patients, and the umbilical vein in 1 patient (traversing the patent foramen ovale (PFO) into left atrium, left ventricle (LV) and across the AV). In the majority of patients pressure measurements were acquired from ascending aorta and LV; afterwards an aortography was performed.
However, since 2018, a new protocol focused on reducing the manoeuvres across the AV has been introduced to reduce the risk of AR. According to the new approach, LV pressure measurement, and in special situations aortography, are replaced with intraprocedural echocardiographic measurements. This approach reduces the need for exchanging the catheters and wires across the vulnerable AV in neonates.
The aortic annulus was mainly measured based on fluoroscopy imaging; the mean AV annulus was 6.5 ±1.4 mm in UKG and 7 ±1.3 mm in angiography. Transcatheter balloon was chosen appropriately to achieve a balloon to annulus ratio of 0.9–1. The following balloons were used: Tyshak (NuMED, USA), Osypka VACS II (Osypka, Germany), Valver (Balton, Poland), and Crossrail (Guidant (now Abott), USA). The median balloon diameter was 6 mm (range: 4–8 mm), and the average balloon/annulus diameter was 0.9 ±0.07 mm, with a maximum balloon/annulus ratio of 1.08. After initial valvuloplasty, if the result was not satisfactory (residual gradient > 35 mm Hg or > 50% of initial gradient), subsequent valvuloplasty was performed with the same or a 1–2 mm larger balloon. In patients with critical stenosis better opening of the AV and improved LV function was considered as a satisfactory procedural effect. According to our new protocol, the gradient on AV as well as separation of the aortic leaflets was assessed with use of intraprocedural echocardiography, and the need for additional valvuloplasty was based on echocardiographic findings.
Transthoracic echocardiography was routinely performed before, during, and after the procedure. Aortic regurgitation was graded with the use of both echocardiography and aortography. Aortography assessment of aortic regurgitation was performed with the use of Seller’s criteria [2, 8, 15]. The echocardiographic assessment of AR was performed with the standard criteria used in similar studies [14]. The AR was deemed as significant when it met the moderate aortic regurgitation echocardiographic findings, or Sellers’ criteria grade II when postprocedural echocardiography did not provide enough data (2 patients from the beginning of the study).
Adenosine infusion was administered in 6 (14.0%) patients; rapid ventricular pacing was not used in this study group. A new approach, focused on reducing manoeuvres across AV, was applied electively in 6 patients. Moreover, in neonates in critical condition with need for immediate gradient relief, a similar approach was performed to reduce the procedure duration (in 5 patients).
Classification of early outcomes
In patients with adequate ejection fraction (EF) early postprocedural success was based on gradient reduction and the presence of aortic regurgitation. Success of the procedure was based on the classification presented in previous studies [2, 8]:
Adequate outcome (Group I): patients with isolated AS, residual gradient < 35 mm Hg (mean echocardiography or peak-to-peak gradient), and no/minor AR. Patients with combined AV disease, residual gradient of < 35 mm Hg and minor change in AR.
Inadequate outcome (Group II): patients with isolated AS, residual gradient > 35 mm Hg (mean echocardiography or peak-to-peak gradient), or more than mild AR. Patients with combined AV disease, residual gradient of > 35 mm Hg, or change in AR of at least one angiographic grade.
In patients with critical AS the gradient reduction was not taken into account because in many of those patients the gradient through the AV rises after reducing the afterload – due to improved LV contractility. In those cases, improved EF as well as weaning off Prostin or inotropic support was used as a determinant of adequate early outcome (group I).
Follow-up was gathered from the outpatient clinic. The follow-up endpoints were death and need for reintervention (BAV or surgery). The median follow-up available in 32 (74.4%) patients was 50 months (range: 1–245).
Statistical analysis
Statistical analysis was performed with the use of Statistica 12 (StatSoft), and statistical significance was defined as p < 0.05. The normality of distribution was assessed with the use of the Shapiro-Wilk test. For normal distribution, data are presented as mean value ± standard deviation, and for non-normal distribution, data are presented as median value and range. The t-test and U-Mann-Whitney test were used to compare quantitative data in 2 groups for normal and non-normal distribution, respectively. Comparison of qualitative data in groups was performed with χ2 Pearson test and χ2 Yates test in small sample size groups. The Kaplan-Meier curve of survival was used to assess and portray survival and FFR in our study.
Z-score values were derived from [16].
Results
Baseline characteristics
Patients’ baseline characteristics are presented in Table I. AV morphology was regarded as bicuspid in 22 (51.2%) patients, tricuspid in 12 (27.9%) patients, unicuspid in 2 (4.7%) patients, and undefined in 7 (16.3%) patients. AV was hypoplastic (AV annulus diameter Z-score < –2) in 9 (21%) patients. Mild preprocedural aortic regurgitation was noted in 8 (18.6%) patients. Coexisting congenital heart defects included the following: ostium secundum in 8 (18.6%) patients, aortic coarctation in 4 (9.3%) patients, and ventricular septal defect in 3 (7%) patients. Non-trivial mitral regurgitation occurred in 11 (25.6%) patients and fibroelastosis in 4 (9.3%).
Table I
Baseline characteristics and comparison of patients with non-critical vs. critical aortic stenosis
On admission, 10 (23.3%) patients required mechanical ventilation, and 11 (25.6%) patients were duct dependent and required PGE2 infusion. Overall, 17 (39.5%) patients met the criteria for critical AS (dependence on Prostin or inotropic infusion or significantly reduced EF). Baseline characteristics and comparison of patients with critical vs. non-critical aortic stenosis are presented in Table I.
Early effects of BAV
All patients survived the procedure, although 1 patient required external cardiac massage during the procedure due to bradycardia and hypotension.
Overall early survival (< 30 days after the procedure) was 93%: 1 patient died due to sepsis, 1 patient after surgical valvuloplasty, and 1 patient due to severe ventricular rhythm disturbances. All of those patients had critical aortic stenosis. Early survival in patients with critical AS in our study group was 82.4%.
The gradient decreased from 67.5 ±26.3 to 21.3 ±12.6 mm Hg (p < 0.001). Significant AR occurred in 13 (30.2%) patients; however, only 2 patients developed moderate/severe AR and 1 patient severe AR; 3 (7%) patients met fluoroscopy Seller’s criteria grade III and none of them grade IV.
Overall, 28 (65.1%) patients met the criteria for adequate early outcomes (group I).
Among patients with non-critical aortic stenosis 15 (57.7%) patients met criteria for group I, whereas in patients with critical AS 13 (76.5%) patients met criteria for group I.
In the non-critical stenosis group, out of 11 patients with inadequate early outcomes (group II):
in 6 (23.1%) patients the residual gradient was > 35 mm Hg,
in 3 (11.5%) patients the gradient reduction was < 50%,
in 5 (19.2%) patients significant AR was observed.
In some of those patients more than one criterion of inadequate early outcome was fulfilled.
Among the patients with critical aortic stenosis 3 died and 2 required reintervention in immediate follow-up (< 30 days after initial procedure). Early complications included pulse loss in femoral artery in 7 (16.27%) patients (in 4 of whom the pulse loss resolved after anticoagulation or thrombolytic therapy), septicaemia in 4 (9.3%) patients, and pericardial tamponade in 1 (2.3%) patient.
Among patients with bicuspid AV, 13 (59.1%) had criteria of group I, with tricuspid 9 (75%) patients, in unicuspid 1 (50%) patient, and in undefined/borderline anatomy 5 (71.43%) patients.
We were not able to find patient-related predictors of optimal outcome. Ejection fraction, LV dimension, aortic valve annulus diameter, age, weight, or peak gradient on aortic valve did not affect the results of the procedure.
Procedure-related predictors of optimal early outcome (group I)
As mentioned before, in the whole study group the balloon diameter was chosen appropriately to achieve a balloon/annulus ratio of 0.9–1. We did not observe any impact of the balloon/annulus ratio on early outcomes. The balloon/annulus ratio in group I was 0.9 (range: 0.75–1.08), and in group II it was 0.9 (range: 0.77–1).
Adenosine did not have a significant impact on overall optimal early effects: 3 (50%) patients who were given adenosine qualified for group I versus 25 (67.57%) patients in whom adenosine was not administered. However, only 1 (16.67%) patient in whom adenosine was administered developed significant aortic regurgitation versus 12 (32.43%) patients in whom adenosine was not administered.
The new approach, which focused on reducing manoeuvres across AV, did not prove to have a significant impact on the procedure outcomes, with 4 (66.67%) patients in the new protocol group versus 24 (64.86%) patients in traditional procedure fulfilling the criteria for group I. Accordingly, new-onset significant aortic regurgitation occurred in 2 (33.3%) patients who underwent the procedure according to the new protocol versus 12 (32.43%) patients in whom a traditional approach was performed.
Survival
Survival in our study group was 90.7% (derived from the Kaplan-Meier estimator), with 3 patients dying periprocedurally (< 30 days after initial procedure) and 1 patient after a Glenn procedure at the age of 9 months. All 4 patients who died in our follow-up had critical AS; additionally, 2 of them had hypoplastic AV. One-year survival in patients with critical AS was 65% vs. 100% in patients with non-critical AS, whereas 1-year survival in patients with hypoplastic AV was 59% vs. 94% in patients with non-hypoplastic AV.
Reinterventions
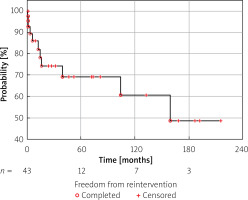
Overall, 11 (35.5%) patients required reintervention at a median of 12 months after the procedure (range: 1–215 months). Reinterventions included BAV in 5 patients, surgical aortic valvuloplasty in 2 patients, Ross operation in 2 patients, surgical AV replacement in 1 patient, and Norwood operation in 1 patient (a patient with borderline HLHS, who underwent Norwood and Glenn operation, did not survive until completion of Fontan circulation). The following graph represents the Kaplan-Meier curve of freedom from reintervention (FFR) (Figure 1).
Early outcomes of BAV in long-term follow-up
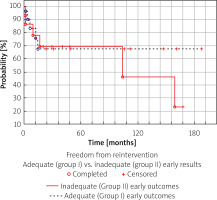
Patients in whom the procedure was classified as adequate (group I) had a significantly higher freedom from reintervention in long-term follow-up than patients in whom the procedure was classified as inadequate (group II). In group I, the 15-year FFR was 71% vs. 22% in group II (Figure 2).
Aortic regurgitation
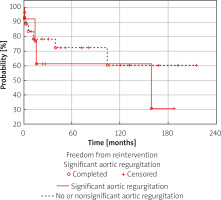
Patients with significant postprocedural AR had a lower long-term FFR, with 30% 15-year FFR vs. 60% 15-year FFR in patients without significant AR. While these data show that aortic regurgitation has a very high burden on the need for reintervention, the overall adequacy of periprocedural success has a more prominent impact on late results (Figure 3).
Aortic valve morphology
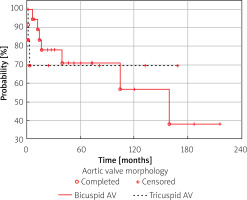
While early results for both tricuspid and bicuspid AV were similar, in long-term follow-up patients with bicuspid valve had a significantly lower freedom from reintervention at 10 years of follow-up, i.e. 57% vs. 69% in patients with tricuspid aortic valve (Figure 4).
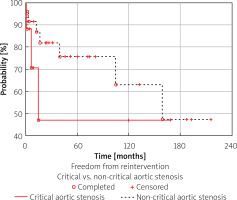
Critical vs. non-critical aortic stenosis
Patients with critical aortic stenosis require early interventions significantly more often than patients with non-critical aortic stenosis. However, in long-term follow-up the need for reintervention in both groups became closer to one another, with 47% of patients with critical neonatal aortic stenosis not requiring any form of reintervention at 10 years of follow-up compared to 63% of patients with non-critical aortic stenosis (Figure 5).
Discussion
The definition of critical AS is ununified [7, 8, 14, 17, 18]; we classified patients as critical when their survival was dependent on prostaglandin infusion, with significant depression of LV function, and those who required inotrope infusion, which was commonly used in other studies [8, 13, 14]. The reasoning behind this classification is not random; it is well known that these patients, mainly due to decreased gradient through the AV caused by depressed LV function, require drastically different approaches of qualification to the procedure and monitoring of the effects of those procedures. Applying the same standards to those patients would make a comparison of the effects of BAV inadequate. Moreover, those patients were responsible for the majority of early mortality and reinterventions (all cases of early mortality in our group occurred in patients with critical AS), which is similar to previous studies [14]. In this group of patients BAV should be treated as an acute life-saving procedure, and as such the early results are favourable, with high rates of improved LV function, and weaning off prostaglandins and inotropes.
In patients deemed as non-critical the early results of BAV are very promising, with great reduction of gradient and low early mortality (none in our study group), need for early reintervention, and significant complications.
Significant aortic regurgitation remains a common aftermath of BAV. The data on postprocedural AR are diverse and not always synonymous, ranging from 7% to 27.6% [8, 19], while the occurrence of AR in our study group was higher (30.2%). It is important to note that almost all the cases were classified as moderate, and the majority of them appeared in patients with critical stenosis.
Due to the high impact of AR on long-term results [5], which has also been proven in our study, it is of utmost importance to reduce the incidence of aortic regurgitation.
Based on previous studies [9, 14, 20], a consensus emerged that in order to minimize the incidence of AR the balloon/annulus ratio should not exceed 1; however, the size of the balloon chosen inside the safe range of 0.8–1.2 did not affect the incidence of AR occurrence.
Various studies tried to identify risk factors of AR and acute adequate effects [1–3, 6, 9, 21]; however, apart from neonatal age and critical AS [2, 8, 11] there remains no consensus regarding risk factors of AR. Likewise, our study failed to identify the risk factors of AR. In recent years new strategies have been studied to reduce the incidence of AR.
The effects of rapid ventricular pacing are conflicting, with some studies showing no significant effects [22–25], while others show very promising results [26]. In our study group rapid ventricular pacing was not performed due to increased risk of ventricular fibrillation and no convincing data regarding its results. Use of rapid ventricular pacing might be subject to change with regards better outcome and new techniques minimizing vascular damage [26]. In our institution adenosine is used in some neonates with preserved ventricular function to stabilise the balloon catheter. While the occurrence of acute significant AR is lower in patients in whom adenosine was used, a larger study is needed to assess its effect.
Based on the concept that manoeuvres (e.g. exchanging wires, sheaths, and catheters) across the valves cause damage to the vulnerable neonatal valve tissue, which is a mainstay in other neonatal procedures such as preemie PDA closure [27], in recent years we have introduced a new approach. The new approach, used since 2018 in 6 patients, focuses on echocardiographic measurement of gradient through the AV, atraumatic passing of AV with a soft guidewire (preferably coronary guidewire), and omitting the invasive gradient measurement in favour of echocardiographic assessment. While crossing the AV with a soft guidewire might prolong the procedure, we think that atraumatic valve crossing is more important.
Unfortunately, we were unable to prove the superiority of the new approach; a larger study group and longer follow-up period is needed to fully assess the usefulness of this approach.
BAV has always been considered as a palliative procedure [20], and despite the advances in interventional cardiology, it remains as such. The FFR of BAV in congenital AS remains low, at around 50% 10 years after initial procedure in large-scale, multicentre studies [4, 11, 28]. Our study shows a favourable long-term effect, with freedom from reintervention of 60% at 10-year follow-up in the neonatal-only study group. Hochstrasser et al. [5] reported excellent long-term results of BAV in the paediatric population, with an FFR at 10-year follow-up of 89%; unfortunately, their study did not provide enough information about the procedural technique.
Our study shows that long-term follow-up and management of patients after BAV is crucial due to common late reinterventions occurring as late as 13 years after neonatal procedure. This data is consistent with reports of progressive AR in patients following BAV [14]. Our study shows that patients with significant postprocedural AR and those who did not meet the criteria of adequate early outcomes have worse long-term results. While those findings are not revolutionary, especially because convincing measures of improving those outcomes have not been found, they prove the usefulness of the early outcome qualification used in previous studies [2, 8]. Moreover, the ability to better predict the long-term outcomes after BAV is a great tool for the management of patients and counselling of parents about the upcoming prognosis.
There is widespread discussion on the comparison of long-term results of BAV versus surgical aortic valvuloplasty (SAV). Large-scale multi-centre studies show similar long-term results of BAV and SAV with comparable survival and FFR in both groups [4, 28]. However, some studies claim superior results of SAV with better freedom from reintervention in long-term follow-up [3, 29]; however, when reviewing the methodology and study population, we can find a higher rate of critical stenosis in the BAV group [3, 29], and a higher proportion of tricuspid AV in the SAV groups [29]. Moreover, primary SAV results in the formation of postoperative adhesions, leading to more complicated further surgical reinterventions. Because critical aortic stenosis is a risk factor of poor late outcome [2, 8, 11], and because we have shown that bicuspid AV is connected with a higher rate of reintervention, it is our belief that without a true randomized clinical trial it is impossible to prove superiority of either technique.
Conclusions
Balloon aortic valvuloplasty is a safe and successful acute treatment in neonates with aortic stenosis. Long-term follow-up of BAV is dependent on early results of the procedure and remains suboptimal. Preventable risk factors of poor outcome and procedural techniques aiming to improve it are still not well documented.