Introduction
Solid pseudopapillary tumor of the pancreas (SPTP) is a low-malignancy and rarely diagnosed tumor, which was first reported by Dr. Frantz in 1959 [1]. SPTP accounts for 1–3% of pancreatic tumors and 9% of pancreatic cyst tumors [2] and is dominant in young female patients (> 90% of cases) mostly between 30 and 40 years old [3]. In recent years, the prevalence of SPTP has increased due to the popularization of computed tomography (CT) examination. Most SPTPs are observed in the distal areas of the pancreas, while only one-third of the tumors are found in the head of the pancreas [4, 5]. In most cases, surgical resection was used to treat SPTPs. Although the malignancy rates are between 3.6% and 56% [6], the assessing standard for malignancy is flexible and is highly dependent on the preoperative image and intraoperative anatomy but not pathology. However, even with metastasis, the overall 5-year survival rate is approximately 97% [7].
Aim
With the development of laparoscopic techniques, the minimally invasive approach for treating SPTP is accepted by the young population, benefiting from its smaller incision compared with open surgery while maintaining the same level of safety [8, 9]. However, few studies focus on this specific tumor. We summarized our limited knowledge of SPTP treatment and the outcome of the patients, especially on the evolving operative management from open to laparoscopic surgery.
Material and methods
In this study, 39 patients with SPTP between January 2013 and December 2020 were analyzed. SPTPs were pathologically confirmed for all the patients based on histological and immunohistochemical examinations after the surgery. The demographics, perioperative, intraoperative, and pathological variables of the SPTP patients with surgery treatment were described.
Follow-up included clinical manifestations, laboratory examinations, and abdominal enhanced CT scan. The patients were followed up in an outpatient setting by phone interview every year.
Results
General characteristics of resected SPTPs
During the last 8 years, a total of 39 cases were identified and surgically resected with SPTP in our center. The general characteristics of SPTP are listed in Table I. There were 32 (82.1%) female patients and 7 (17.9%) male patients, and the mean age was 34.0 ±12.1 years, ranging from 13 to 62 years. The prevalence of SPTP was dominant in young females, as the number of patients under 40 years was 29 (74.4%) and 89.7% were female (26/29). The mean tumor diameter was 4.81 ±2.36 cm, ranging from 1.2 to 13 cm.
Table I
Demographics and clinical features of 39 patients with SPTP
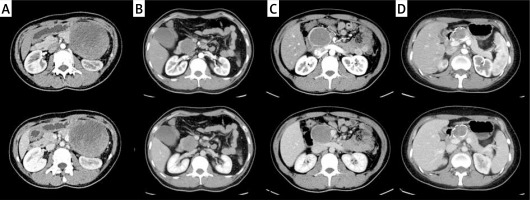
The SPTPs were incidentally found without specific symptoms in nearly half of the patients (n = 19, 48.7%), while the rest of the patients presented with abdominal pain (n = 7, 17.9%), abdominal discomfort (n = 9, 23.1%), back pain (n = 3, 7.7%), or slight jaundice (n = 1, 2.6%). It is possible for the tumor to appear in any area of the pancreas. Among the 39 cases, the majority of the tumors (19 cases) were located in the body and tail of the pancreas (48.7%), 12 were located in the neck of the pancreas (30.8%) and 8 in the head of the pancreas (20.5%). A preoperative CT scan or magnetic resonance imaging (MRI) was performed on all the patients to confirm the presence of a pancreatic mass. Of the 32 tumors examined in our hospital, 25 were solid-cystic, 3 were completely cystic and 4 were completely solid. Intracystic hemorrhage was observed in 3 cases and calcification was observed in 6 cases (Photo 1 shows the different CT manifestations of SPTP). The diagnosis of SPTP was confirmed radiologically in 23 cases. Endoscopic ultrasound-guided fine-needle aspiration biopsy (EUS-FNA) was conducted in 3 patients; among them, two were suspected to be neuroendocrine tumors and one was diagnosed as SPTP.
Postoperative outcome
The surgery approach was selected mainly related to the location and size of the tumor. With the development of surgical techniques, the application of laparoscopic surgery has been increasing. Distal pancreatectomy was conducted in twenty-two patients (seven open with one spleen sparing, fifteen laparoscopic with seven spleen sparing), pancreaticoduodenectomy was conducted in 7 patients (four open, three laparoscopic), middle pancreatectomy was conducted in 8 patients (seven open, one laparoscopic), and tumor enucleation was conducted in 2 patients (one open, one laparoscopic). The mean operation time for both open and laparoscopic surgery was 254.1 ±107.3 min. Four patients from 2013 to 2015 were treated by open surgery due to the lack of laparoscopic pancreatic surgical programs in our institute. Since 2015, 20 out of 35 (57.1%) patients have been successfully treated with laparoscopic operations. Notably, due to a clear view after the enlargement in the distal pancreatectomy (DP) group, it was easier to preserve the spleen in the laparoscopic approach. One patient underwent pancreaticoduodenectomy (PD), in which the superior mesenteric vein (SMV) was resected and the artificial vessel was reconstructed. Another patient underwent enucleation with partial duodenectomy owing to a suspected local invasion of the tumor. All the patients got the R0 resection margin both in the open and laparoscopic surgery. The most common postoperative complication was Grade B-C pancreatic fistula (17.9%). Other complications included abdominal infection (12.8%), biliary fistula (5.1%), chyle fistula (5.1%) and bleeding (2.6%). One patient who developed intra-abdominal bleeding underwent further interventional arterial embolism. The total mortality rate of all the 39 patients for surgery was 0%.
Minimally invasive procedures were successfully conducted in 51.3% (20/39) of the patients. We compared the duration of the surgery, blood loss, length of the incision, postoperative fasting time, postoperative hospital stay and complication rate between the patients who received the minimally invasive surgery and those who did not (Table II). No significant difference was observed in regard to the duration of surgery ((263.2 ±78.7) min vs. (244.6 ±130.1) min, p > 0.05), blood loss ((175.0 ±103.1) ml vs. (236.3 ±231.5) ml, p > 0.05), or complication rate (Clavien-Dindo II-IV) (25% vs. 31.6%, p > 0.05) between the laparoscopy and open group. Significant differences were detected in postoperative fasting time ((1.8 ±1.1) days vs. (3.8 ±1.8) days, p < 0.05), postoperative hospital stay ((9.6 ±3.4) days vs. (14.9 ±10.7) days, p < 0.05) and incision length ((5.3 ±1.8) cm vs. (18.2 ±4.3) cm, p < 0.05). Additionally, we further divided the patients into four subgroups: enucleation, distal pancreatectomy, middle pancreatectomy, and pancreaticoduodenectomy (Table II). We then compared the same indexes between the minimally invasive surgery group and the non-minimally invasive surgery group separately. Comparing laparoscopic surgery with open surgery, there was nearly no difference in either the blood loss or the complication rate between the two treatment types in all subgroups (p > 0.05). In the distal pancreatectomy group, although laparoscopic surgery involved a longer operation time, it was associated with a shorter postoperative fasting time ((1.3 ±0.8) days vs. (3.1 ±1.8) days, p < 0.05) and a shorter postoperative hospital stay ((8.4 ±2.3) days vs. (10.6 ±1.8) days, p < 0.05) compared to the open surgery. The incision length was smaller in all laparoscopic subgroups (p < 0.05). In addition, there was no difference in the complication rate in every subgroup separately.
Table II
Operative procedure and postoperative outcome for SPTP
| Variable | Duration of surgery [min] | Blood loss [ml] | Length of incision [cm] | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Laparoscopic | Open | P-value | Laparoscopic | Open | P-value | Laparoscopic | Open | P-value | |
| Enucleation | 144 | 113 | – | 200 | 50 | – | 5 | 12 | – |
| Distal pancreatectomy | 241.5 ±56.3 | 176.4 ±57.2 | < 0.05* | 170.0 ±116.6 | 200.0 ±88.6 | > 0.05 | 5.1 ±1.6 | 18.4 ±2.2 | < 0.05* |
| Middle pancreatectomy | 350 | 272.7 ±98.2 | – | 250 | 238.6 ±208.6 | – | 4 | 17.0 ±5.5 | – |
| Pancreatico-duodenectomy | 382.3 ±40.7 | 347.5 ±182.7 | > 0.05 | 166.7 ±23.6 | 342.5 ±323.1 | > 0.05 | 6.3 ±2.6 | 21.2 ±2.2 | < 0.05* |
| Total | 263.2 ±78.7 | 244.6 ±130.1 | > 0.05 | 175.0 ±103.1 | 236.3 ±213.5 | > 0.05 | 5.3 ±1.8 | 18.2 ±4.3 | < 0.05* |
| Variable | Postoperative fasting time [day] | Postoperative hospital stay [day] | Complication rate (%) | ||||||
| Laparoscopic | Open | P-value | Laparoscopic | Open | P-value | Laparoscopic | Open | P-value | |
| Enucleation | 2 | 8 | – | 6 | 12 | – | 0 (0/1) | 100 (1/1) | – |
| Distal pancreatectomy | 1.3 ±0.8 | 3.1 ±1.8 | < 0.05* | 8.4 ±2.3 | 10.6 ±1.8 | < 0.05* | 20 (3/15) | 14.3 (1/7) | > 0.05 |
| Middle pancreatectomy | 2 | 4.1 ±1.0 | – | 13 | 12.0 ±4.1 | – | 0.0 (0/1) | 28.6 (2/7) | – |
| Pancreatico-duodenectomy | 3.7 ±0.9 | 3.3 ±1.6 | > 0.05 | 15.3 ±2.1 | 28.3 ±16.7 | > 0.05 | 66.7 (2/3) | 50 (2/4) | > 0.05 |
| Total | 1.8 ±1.1 | 3.8 ±1.8 | < 0.05* | 9.6 ±3.4 | 14.9 ±10.7 | < 0.05* | 25 (5/20) | 31.6 (6/19) | > 0.05 |
Pathology assessment and immunohistochemistry
Pathology assessment mainly included the location, size, and capsule completeness of the tumor, local or vascular invasion, lymph node metastasis and immunohistochemistry (IHC) expression. All patients reached the negative surgical margins and none of them had lymph node metastasis or distal metastasis. The results of IHC studies were summarized in Table III. Ki-67, β-catenin, vimentin and CD-10 were all 100% positively stained, while α-ACT and CD-56 were nearly 100% positive in the detected samples. Chromogranin A (CgA) was 100% negative in the detected samples. Thirty-eight patients had a Ki-67 index ranging from 1% to 20%; among them six were over 4% and one was as high as 20%. The remaining immunohistochemical profiles were differently expressed.
Table III
Results of immunohistochemical profile of SPTPs in 39 patients
Follow-up
The last follow-up was conducted in March 2021. The median follow-up time was 24 months (range: 3–84 months). All patients were disease-free except one who developed single liver metastasis at 19 months after surgery and then underwent liver tumor resection in another hospital. No new recurrence or metastasis was detected during the 6-month follow-up after the resection of the hepatic metastatic lesion. A whole-life follow-up is indispensable after resections of SPTPs due to potential malignancy of the relapse and metastatic capacity.
Discussion
As a rarely diagnosed tumor, SPTP is considered low malignancy and occurs mostly in young women. The reported female/male ratio of the patients was 10 : 1 [10, 11] while in our study the ratio was 32 : 7. Female hormones seem to affect pathogenesis, but no differences in sex hormone-receptor protein expression have been found so far [12].
SPTP usually lacks specific manifestations and nearly 50% of the patients in our study presented asymptomatically. The incidence of SPTP has increased due to more application of the CT scan. SPTP is commonly featured in calcification, cystic change, and internal hemorrhage owing to a lack of vessels [13]. CT imaging of SPTP with an HU value over 62.6 of the solid component on the delayed contrast phase and progressive enhancement appears more malignant [14]. In addition, the FDG uptake of SPT on PET-CT was associated with tumor cellularity, proliferation index, and histological malignancy [15].
Immunohistochemistry (IHC), a diagnostic gold standard for SPTP, helps to differentiate SPTP from pancreatic neuroendocrine tumors (pNETs) and other cystic tumors [16]. β-catenin is considered the most important feature of SPTP [17–19], which is 100% positively stained as well as Ki-67, vimentin and CD-10 in our study. It is reported that the Ki-67 index over 4% is associated with relapse and metastasis during the follow-up [3, 20, 21]. In our study, the patients with liver metastasis after surgery had a 20% Ki-67 rate.
The curative treatment for SPTP is surgical resection. We performed operations once the abdominal imaging suggests a possibility of SPTP, or when the pathology of EUS-guided biopsy suggests cellular atypia. The selection of surgical approach mainly depends on the location and size of the tumor. With the technological advances in recent years, the laparoscopic operation is rapidly gaining popularity in pancreatic tumor resection. Even for highly technically demanding pancreaticoduodenectomy, the procedure could be completed under laparoscopy after mastering the technical details as well as tips and tricks [22]. The laparoscopic approach benefits from the better view of the vessels attributable to the magnified images and special view from the inferior posterior side of pancreas. Patients treated with the laparoscopic approach seem to be less debilitated with less wound infection, a shorter hospital stay, an earlier recovery and an improved cosmetic outcome [10]. Minimally invasive surgery is an ideal treatment for SPTP, because SPTP is typically rarely invasive to the surrounding tissues while highly associated with the body and tail of the pancreas.
Laparoscopic distal pancreatectomy has become the gold standard approach to deal with distal pancreas lesions [23]. As low-grade malignant tumor, SPTP is a potential candidate for splenic preservation during distal pancreatectomy, which has demonstrated feasibility and safety compared with traditional distal pancreatectomy, including a similar rate of postoperative complications, less epigastric varices formation and reduced risk of postsplenectomy sepsis [24, 25]. In our report, about half of the patients had a laparoscopic resection. In our study, the minimally invasive approach seems to have a superior therapy outcome to the open operation in terms of the incision length, postoperative fasting time and length of hospital stay, while the blood loss and complication rate were similar between the two approaches. These results suggested that compared with traditional open surgery, minimally invasive surgery may obtain similar safety and be superior with a smaller incision and shorter recovery time.
Based on the published data, the overall 5-year survival rate and ten-year survival rate for SPTP are 88–97% and 89.6–96%, respectively [2, 7, 12, 26, 27]. The recurrence rate for SPTP ranges from 1.9% to 15% as reported [2, 12, 27, 28]. Some reports suggested that features such as pancreatic duct dilation, vessel infiltration, and capsule damage may be associated with recurrence [29, 30]. Metastases can be present at diagnosis or years after resection. Metastases mostly occur in the liver and peritoneum [31]. Most centers offer further surgery if the metastatic disease is resectable [11], and these patients can survive long term after the resection [32, 33]. According to the previous studies, most recurrences occurred more than 5 years after resection; thus, an over 5-year follow-up is indispensable after resections of SPTPs [4, 34].
Conclusions
SPTP is a rare disease with low malignancy. Minimally invasive surgery is proven to be a feasible and safe approach to treat this disease because of the smaller incision, shorter postoperative fasting time and shorter postoperative hospital stay. The 5-year and ten-year survival rate is quite good after surgery. Long-time surveillance is essential after resections. Further research investigating the molecular mechanism and clinical characteristics of SPTP is needed to assist the diagnosis, treatment, and prediction of SPTP.