Summary
The most widely investigated poly-L-lactic acid BRS, the Absorb BVS, was withdrawn from the market due to concerns that were mainly driven by an unexpected high thrombogenicity of bioresorbable vascular scaffolds (BVS) compared with drug-eluting stents. In addition, the Novolimus-eluting DESolve (Elixir Medical Corporation, Sunnyvale, CA, USA) was introduced. This is the first study to investigate the acute mechanical performance of a Novolimus-eluting BVS in patients with different clinical presentations using optical coherence tomography (OCT). We found that clinical presentation did not determine acute mechanical performance as assessed by the final OCT pullback. There was evidence of more mechanical complications in terms of fractures and a higher percentage of incomplete strut apposition in the group of patients with chronic coronary syndrome.
Introduction
Bioresorbable vascular scaffolds (BVS) are the latest innovation in the field of interventional cardiology for the treatment of patients with coronary syndrome. They were introduced into routine clinical practice to overcome limitations such as permanent caging by the device and chronic inflammation. Because they dissolve after several years, BVS offer transient vessel support. Thus, they allow and support the restoration of vasomotor function and also facilitate future surgical revascularization [1]. Whereas initial studies of outcome comparing BVS with drug-eluting stents (DES) showed promising results, randomized clinical trials and meta-analyses raised concerns regarding adverse events due to higher early and late thrombogenicity of BVS compared with DES for the treatment of de novo lesions [2, 3]. These findings were also consistently observed in so-called “real-world” registries [4]. Ultimately, the most widely investigated poly-L-lactic acid (PLLA) BRS, the Absorb BVS (Abbott Vascular, Santa Clara, CA, USA), was withdrawn from the market. The causes of scaffold thrombosis (ScT) are multifactorial. However, the notably high rates of early ScT soon pointed towards periprocedural aspects that contribute to its occurrence [4, 5]. Hence, adaptation of the implantation technique and increased experience of the operator led to lower thrombosis rates. Furthermore, the lesion type and the clinical presentation of the patient appear to play a role in BVS performance [6]. It has been shown that the risk of device thrombosis after percutaneous coronary intervention (PCI) is increased in patients with acute coronary syndrome (ACS). Given the greater strut thickness of BVS to compensate for a decreased radial strength, concerns have been raised with regard to ScT in BVS as this might increase shear stress and flow disturbance, especially in the hypercoagulable setting of ACS. Therefore, ACS has been an exclusion criterion for most of the randomized trials, although it is an appealing scenario for BVS: younger patients, fewer prior events, and softer plaque. In addition, the Novolimus-eluting DESolve (Elixir Medical Corporation, Sunnyvale, CA, USA) was introduced.
Aim
In the present study we investigated the acute mechanical device properties of the Novolimus-eluting DESolve BVS in patients who underwent implantation and compared these parameters with the patients’ clinical presentation.
Material and methods
Consecutive patients who underwent PCI with Novolimus-eluting BVS were enrolled in this retrospective study irrespective of their clinical presentation. The index procedure was carried out between January 2014 and August 2015. Patients were classified into ACS and non-ACS groups. Here ACS was defined as ST-elevation myocardial infarction (STEMI), non-ST-elevation myocardial infarction (NSTEMI), or unstable angina.
The DESolve BVS is a polymeric device with a strut thickness of 150 μm that elutes Novolimus. It has greater radial strength and a faster degradation process than the Absorb BVS, and it can be overexpanded up to 0.5 mm above the nominal diameter. Bench tests even showed a higher overexpansion capacity during post-dilatation without increasing fractures [7]. Furthermore, the DESolve BVS is able to self-correct for minor malapposition [8].
PCI was performed in a single, high-volume centre in accordance with standard clinical practice. The radial approach using a 6 French guiding catheter was favoured and used whenever feasible. At the beginning of the procedure all patients were administered 70 U/kg body weight unfractionated heparin. Prior to lesion preparation patients were administered intracoronary nitroglycerine. Lesion preparation was initiated with pre-dilatation utilizing a non-compliant balloon that corresponded 1 : 1 to vessel size. The use of a debulking device was left to the operator’s discretion. Similar to balloon sizing, BVS sizing was approximately 1 : 1 with respect to the vessel diameter. Its deployment was accomplished by slowly inflating the stent balloon (1 atm over 10 s, 2 atm over 10 s, then 2 s per atm). The final pressure was maintained for 20 s. Post-dilatation was performed with a non-compliant balloon in accordance with the maximum expansion limits of the BVS.
Frequency domain optical coherence tomography (OCT) was performed using the Ilumien Optis system (St. Jude Medical, Inc., Minneapolis, MN, USA). Pullbacks were performed in an automatic manner or manually, if necessary, at 36 mm/s during contrast injection at a rate of 3 to 5 ml/s. After having placed the imaging catheter distally to the treated lesion, the pullback was recorded until either the guiding catheter was reached or the maximum pullback length was completed. In the latter case, or if the treated lesion was incompletely recorded, a second sequential pullback was combined to image the whole lesion. Data from the final pullback just before the end of the procedure were used for the analysis in this study.
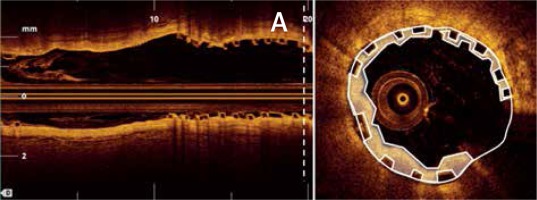
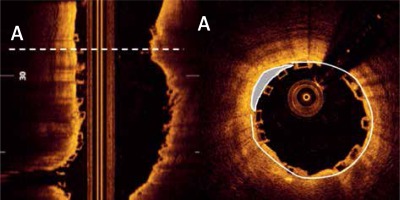
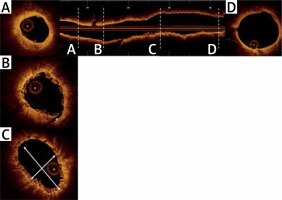
OCT measurements were performed offline using the LightLab Imaging workstation (St. Jude Medical, Inc.). Longitudinal cross-sections of the final pullback were analysed at 1-mm intervals within the stented lesion and 5 mm proximally and distally to the scaffold (Figure 1). The following quantitative parameters were determined manually: the percentage of incomplete strut apposition (ISA) at 1-mm intervals, calculated as a percentage of the total number of malapposed struts divided by the total number of struts (Figure 2); the ISA area; the tissue prolapse area (Figure 3), defined as the projection of tissue into the lumen between struts; residual area stenosis (RAS), calculated as (1-minimum lumen area (MLA)/reference vessel area (RVA)); the eccentricity index, computed as the ratio between the minimum and maximum diameters; the symmetry index, defined as the difference between maximum scaffold diameter and minimum scaffold diameter divided by the maximum scaffold diameter. If no meaningful value for proximal or distal RVA was obtained, the largest luminal cross-sectional area at the distal or proximal end of the scaffold was used. An edge dissection was defined as any disruption of the vessel luminal surface at the edges of the scaffold with a visible flap (> 300 μm). A scaffold fracture was assumed if isolated struts were observed to be unopposed within the scaffold lumen or if struts were stacked. The analysis of the OCT pullbacks was conducted by the local core lab for intravascular imaging.
Figure 1
A – Distal reference vessel area (DRVA) = 3.77 mm2, B – asterisk indicating a distal edge dissection, C – cross section with minimum eccentricity index (minimum/maximum diameter) = (2.29 mm/3.96 mm) = 0.57, D – proximal reference vessel area (PRVA) = 11.59 mm2. Reference vessel area (RVA) = (PRVA + DRVA)/2 = (11.59 mm2 + 3.37 mm2)/2 = 7.48 mm2
Quantitative coronary angiography (QCA) was carried out with the help of offline QCA software (CAAS QCA, Pie Medical Imaging BV, The Netherlands). The following parameters were assessed during post-hoc analysis: reference vessel diameter (RVD) through automatic interpolation, minimum lumen diameter (MLD), percentage area stenosis (AS), and lesion length. Analysis was independently performed by two experienced interventional cardiologists.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics (SPSS Statistics 23, IBM Deutschland GmbH, Ehningen, Germany). Continuous variables with normal distribution are expressed as means and standard deviations; categorical variables are given as number and percent. The χ2 and Fisher’s exact tests were used for comparison of categorical variables, and Student’s t-test or the Wilcoxon rank-sum test was applied for continuous variables. P-values < 0.05 were considered statistically significant.
Results
A total of 79 patients were enrolled in this study. All patients were implanted with a Novolimus-eluting BVS. Thirty-four patients presented with ACS, and 45 patients were diagnosed with chronic coronary syndrome (non-ACS group). Patients in the ACS group were significantly younger (non-ACS 64.9 ±8.5 years vs. ACS 57.4 ±7.5 years, p < 0.001) and less frequently treated for arterial hypertension (100% vs. 79.4%, p < 0.001). Patients in the two groups had a similar prevalence of diabetes mellitus (13.3% vs. 25.5%, p = 0.15) and smoking (58% vs. 70%, p = 0.24) as well as a similar left ventricular ejection fraction (56.1 ±95 vs. 53.8 ±10.5%, p = 0.39) and number of diseased vessels (Table I).
Table I
Baseline characteristics
| Parameter | Non-ACS (n = 45) | ACS (n = 34) | P-value |
|---|---|---|---|
| Age [years] | 64.9 ±8.5 | 57.4 ±7.5 | < 0.001* |
| Female sex (%) | 60.0 | 67.6 | 0.46 |
| Hypertension (%) | 100 | 79.4 | < 0.001* |
| Hyperlipoproteinemia (%) | 75.6 | 52.9 | 0.04* |
| Diabetes mellitus (%) | 13.3 | 26.5 | 0.15 |
| Current smoker (%) | 57.8 | 70.6 | 0.24 |
| Family history (%) | 33.3 | 41.2 | 0.47 |
| Prior PCI (%) | 35.6 | 14.7 | 0.04* |
| Prior MI (%) | 42.2 | 8.8 | < 0.001* |
| Left ventricular ejection fraction (%) | 56.1 ±9.5 | 53.8 ±10.5 | 0.39 |
| Clinical indication (%): | < 0.001* | ||
| Stable angina | 100 | 0 | |
| STEMI | 0 | 35.3 | |
| NSTEMI | 0 | 17.1 | |
| Unstable angina | 0 | 47.1 | |
| Number of diseased vessels (%): | 0.46 | ||
| 1 | 20 | 23.5 | |
| 2 | 22.2 | 32.4 | |
| 3 | 57.8 | 44.1 | |
Lesions were typically located in the RCA in the non-ACS and the LAD in the ACS group (p = 0.01). They did not differ significantly with respect to AHA/ACC lesion classification (Table II). In most cases lesions were classified as being of type B1 (35.6 vs. 43.8% for non-ACS vs. ACS, respectively). QCA analysis showed no difference between the two groups with respect to reference vessel diameter (2.6 ±0.7 mm vs. 2.4 ±0.5 mm; p = 0.43), area stenosis (74.5% vs. 74.4%; p = 0.89) and lesion length (10.6 ±4.5 mm vs. 10.0 ±5.0 mm; p = 0.48).
Table II
Angiographic and QCA lesions’ characteristics
| Parameter | Non-ACS (n = 45) | ACS (n = 34) | P-value |
|---|---|---|---|
| Target vessel (%): | 0.01* | ||
| LAD | 22.2 | 52.9 | |
| LCX | 24.4 | 20.6 | |
| RCA | 53.3 | 26.5 | |
| AHA/ACC lesion classification (%): | 0.70 | ||
| Type A | 26.7 | 31.3 | |
| Type B1 | 35.6 | 43.8 | |
| Type B2 | 24.4 | 15.6 | |
| Type C | 13.3 | 9.4 | |
| QCA analysis: | |||
| RVD [mm] | 2.6 ±0.7 | 2.4 ±0.5 | 0.43 |
| MLD [mm] | 1.2 ±0.5 | 1.1 ±0.4 | 0.15 |
| AS (%) | 74.5 | 74.7 | 0.89 |
| Lesion length [mm] | 10.6 ±4.5 | 10.0 ±5.0 | 0.48 |
* Significant difference p < 0.05. LAD – left anterior descending artery, RCX – left circumflex artery, AHA – American Heart Association, ACC – American College of Cardiology, QCA – quantitative coronary angiography, RVD – reference vessel diameter, MLD – minimal lumen diameter, AS – area stenosis, ACS – acute coronary syndrome.
Lesion preparation was initiated with pre-dilatation in most cases in both groups (93.3% vs. 97.1%; p = 0.46). Usage of non-compliant balloons was comparable in the two groups (81% vs. 75.8%; p = 0.58). There was no difference between the groups with respect to maximal pressure applied during pre-dilatation or the size of the balloon used (Table III). The implanted scaffold mean diameter was 3.1 ±0.4 mm vs. 3.1 ±0.4 mm (p = 0.89) and the mean length was 19.7 ±5.9 mm vs. 19.8 ±5.6 mm (p = 0.73). The deployment pressure applied did not differ between the non-ACS and ACS groups. In accordance with the concept of routine pre-dilatation, final post-dilatation was performed in most cases (80% vs. 88.2%; p = 0.33). Diameter, length, and inflation pressure were similar in the two groups (Table III).
Table III
Procedural characteristics
OCT findings are summarized in Table IV. A total of 20,008 struts and 1,593 cross-sections were analysed. Final mean and maximum scaffold diameters were similar in the non-ACS and ACS groups (mean: 3.1 ±0.4 mm vs. 3.0 ±0.4 mm; p = 0.11; maximum: 3.5 ±0.5 mm vs. 3.4 ±0.4 mm; p = 0.34). Likewise, the mean scaffold area and mean lumen area did not differ between the two groups (Table IV). The mean residual area stenosis was 14.5% in patients presenting with a chronic coronary syndrome and 19.5% in patients presenting with ACS (p = 0.39). Assessment of geometric parameters revealed a mean eccentricity index of 0.78 ±0.13 vs. 0.78 ±0.06 (p = 0.42) and a symmetry index of 0.42 ±0.10 vs. 0.42 ±0.09 (p = 0.97). OCT showed only a few dissections that occurred similarly in both groups (6.6% vs. 5.9%; p = 0.50). Fractures, however, tended to occur more frequently in the non-ACS group (22.2 vs. 5.9%), although this difference was not significant (p = 0.07). The percentage of malapposed struts was 2.7 vs. 1.9% (p = 0.15). OCT analysis revealed a slightly, although not significantly, greater area in the ACS group (2.7% vs. 1.9%; p = 0.15).
Table IV
Optical coherence tomography findings
No adverse event occurred within the ensuing post-procedural period. In particular, no scaffold thrombosis was documented.
Discussion
This is the first study to compare the acute mechanical performance of a Novolimus-eluting BVS with clinical presentation by means of OCT. The patient collective comprised two groups: those presenting with ACS and those with chronic coronary syndrome. The principal findings are that: 1) good acute mechanical performance of the device was noted irrespective of the initial clinical presentation of the patient; and 2) patients with chronic coronary syndrome tended to have more mechanical complications, e.g. scaffold fractures and ISA.
The study population fulfilled the typical criteria for the implantation of BVS. This holds true especially for patients with ACS, who were relatively young and had a short history of coronary disease. Furthermore, the lesion complexity, as assessed using the criteria of the American College of Cardiology/American Heart Association [9], was predominantly simple and none of the cases involved a bifurcation.
The study shows that Novolimus-eluting BVS can be implanted with good acute mechanical performance in both groups. This finding is based on our measurement of parameters derived from previous studies that investigated the success of stent deployment [10, 11], which predominantly made use of intravascular ultrasound (IVUS) to evaluate acute and long-term clinical outcome in patients who were treated with either bare metal stents or first-generation DES [12, 13]. The authors found that a minimum cross-sectional lumen area < 5.5 mm2 and an in-scaffold RAS > 20% increases the risk of stent thrombosis. Patients treated in our cohort showed an average RAS of 14.5% in the non-ACS group and 19% in the ACS group, values that are not significantly different (p = 0.39). The number of scaffolds that showed an RAS > 20% was slightly higher in the ACS group, although this difference was not significant (p = 0.34). There was also no difference in MLA between the groups in our patient cohort (6.2 ±2.1 vs. 5.6 ±1.5; p = 0.21). Although IVUS criteria may not automatically be applied to BVS in every case, we believe that the parameters that are generally accepted for the assessment of adequate deployment point towards a good acute result as they are above the published reference values in both groups, especially if it is taken into account that OCT has been shown to measure lower absolute areas than IVUS [14].
OCT analysis did not show a significantly different post-procedural scaffold geometry. Assessment of the acute geometric parameters depicted a mean eccentricity index of 0.78 ±0.13 and 0.78 ±0.06 for the non-ACS and ACS groups, respectively. The clinical impact of asymmetric expansion has been evaluated in the MUSIC trial [15]. Here, first-generation DES with an eccentricity value of 0.7 were associated with favourable angiographic results at the 6-month follow-up. Furthermore, the impact of post-procedural eccentricity of Absorb BVS has been studied as part of the ABSORB II trial [16]. Suwannasom et al. found that BVS in general are more frequently associated with asymmetric and eccentric morphology than DES. This finding was independently associated with higher event rates. The symmetry index provides additional insight into the shape of the BVS; if the index is near zero, the BVS is symmetric throughout the entire length [17]. Both the eccentricity and symmetry index in our cohort demonstrated a good geometric shape, and the results are consistent with those reported in the literature [17, 18].
Tissue may prolapse through the scaffold struts after the implantation of BVS. Hypothetically, and according to the nature of thrombotic occlusion of a vessel (at least in patients with STEMI), one would expect more thrombotic tissue to prolapse in patients with ACS. However, we did not observe a larger prolapse area in our group of ACS patients. It has to be noted that post-dilatation balloons and pressure inflation did not differ significantly between the groups. Nevertheless, there was a slight tendency for more tissue in the ACS group that might not have reached significance due to the proportion of STEMI patients enrolled (35.3%). Diletti et al. were able to show that BVS are able to entrap more thrombus due to their structure [19]. The clinical relevance of prolapse area in the long run is unclear, but in the short term this is of importance as the magnitude of prolapse area is related to increased post-procedural myocardial injury and elevation of CK-MB (creatine kinase) [20].
It was previously assumed that malapposition can lead to stent-related effects and increase adverse event rates [21], but only recently was malapposition identified as a predictor of late and very late ScT in patients treated with BVS [22]. This was irrespective of the timing of the finding during follow-up. Malapposed struts may disrupt laminar flow and activate platelets due to high shear stress [23], thereby contributing to the multifactorial aetiology of ScT. Accordingly, previous studies were able to show that usage of IVUS and OCT to guide the implantation process of DES can reduce event rates [24, 25]. ISA rates of 2% have been reported in the literature [18]. However, in the prospective, multi-centre PRAGUE 19 study, OCT analysis revealed only 1.1% malapposed struts, a much lower proportion than observed in the Absorb Cohort B study (3.5%) [26, 27]. The rate of ISA in our cohort was 2.7% in patients who were treated with chronic coronary syndrome and 1.9% in those treated due to ACS. Although these values are not statistically different, the numerically slightly higher frequency in the non-ACS group is consistent with the finding of more fractures in the non-ACS group because it has to be assumed that the underlying plaque morphology and especially composition may have a negative influence on the expansion and apposition capabilities and may also favour strut discontinuity of BVS. This is supported by Shaw et al., who were able to demonstrate that expansion and eccentricity of Absorb BVS are significantly impacted by coronary artery plaque composition, morphology, and burden [28]. On the other hand, our group was able to show that this dependence is not always applicable to DESolve BVS [29]. The reason for this discrepancy may be a rigorous lesion preparation but may also be the ability of the DESolve BVS to self-correct for minor malapposition. Furthermore, Ormiston et al. performed a bench test that showed the safe threshold for post-dilatation with an Absorb BVS 3.0 to be 3.8 mm at 20 atm, whereas it was 5.0 mm with 20 atm for the DESolve 3.0 mm [7]. Overall, this might have contributed to the lack of significance with respect to the observation of fractures and malapposed struts between the two groups in the present cohort and may suggest an inferior mechanical performance of BVS in stable lesions despite similar indices. This observation is consistent with the findings of Baquet et al., who analysed 26 consecutive patients with OCT 6–8 weeks after BVS implantation (46% ACS) [30]. They reported a numerically higher proportion of protruding and malapposed struts in patients with chronic coronary syndrome than in patients with ACS: protruding 1.26% vs. 4.3%; malapposed 0.39% vs. 2.48%, respectively. On the other hand, they did not observe any difference in the amount of strut coverage or in the proportion of uncovered scaffolds between ACS and chronic coronary syndrome patients.
There are single- and multi-centre observational registries and also randomized trials that assessed BVS performance in ACS patients. The Mainz ACS registry compared 150 patients treated with BVS vs. a control group that was treated with DES during the same period and reported similarly favourable outcomes [31]. In the first propensity matching comparison of BVS in STEMI patients, the major adverse cardiovascular event (MACE) rate in the BVS group was higher than in the DES group [32]. However, the use of an early implantation technique contributed substantially to the event rates, as they were reduced with increasing experience: ScT occurred primarily in the acute phase and in patients without post-dilatation. Imori et al. confirmed the relevance of post-dilatation in ACS settings [33]. The first randomized trial including ACS patients was the EVERBIO II trial [34]. Patients (39% presenting with ACS) were randomly assigned to a BVS or an everolimus-eluting stent (Promus Element; Boston Scientific, Marlborough, Massachusetts, USA) or a biolimus-eluting stent (Biomatrix Flex, Biosensors Europe SA, Morges, Switzerland). The patient- and device-oriented endpoints did not differ after 9 months. No definite stent thrombosis was reported. In addition, the TROFI II trial focused solely on STEMI patients [35]. One case of definite sub-acute ScT occurred in the BVS group (1.1% vs. 0%; p = ns). These data are consistent with our findings, as none of the patients in our cohort showed an adverse event within the ensuing post-procedural period. In particular, no ScT was documented.
While initial studies showed comparable outcomes for BVS and DES, meta-analyses raised concerns regarding adverse events due to higher early and late thrombogenicity of BVS. Ultimately, the Absorb BVS was withdrawn from the market. Elixir Medical Corporation adapted and followed this decision with the PLLA-based DESolve BVS. However, a multicentre post-market analysis showed reasonable follow-up data with an ScT rate of 1% [36]. This may be explained by a favourable acute mechanical result that seems to be less dependent on the underlying lesion morphology, as shown previously by our study group [29]. This is the first study showing a feasible acute mechanical performance of DESolve BVS with OCT irrespective of the initial clinical presentation. New BVS platforms are being developed due to the lingering desire for BVS with transient vessel support. They are currently under clinical investigation [37, 38]. The present study supports this development by contributing reasonable mechanical outcome data for patients presenting with acute and chronic coronary syndrome. Especially patients presenting with ACS represent an important cohort and should not be excluded from large, randomized trials in the future.
There are several limitations associated with this study. Its registry nature with retrospective collection of the patients’ data has inherent limitations and the evidence provided should be seen as hypothesis generating. In addition, the DESolve BVS is no longer available as Elixir Medical Corporation followed the decision of Abbott Vascular, which took the Absorb BVS off the market due to the reported adverse clinical events. Furthermore, although the protocols used for lesion preparation, scaffold deployment, and post-dilatation were the same for all operators contributing to this study, their adaptation due to the operator’s decision may have affected the final acute mechanical result and cannot be excluded. Furthermore, the sample size of the study was small, and we did not perform an analysis of the plaque morphology, which might have helped to understand the increasing mechanical discontinuities seen in the non-ACS group. Further randomized clinical trials are required to investigate the use of BVS in the setting of acute and chronic coronary syndrome, ideally with broad application of intravascular imaging.
Conclusions
In randomized trials mid- and long-term outcomes of PCI procedures with BVS have been assessed predominantly in patients with stable coronary disease, and relatively simple lesions and patients presenting with ACS were excluded, although these patients in particular might benefit from the BVS platform. We were able to show that the clinical presentation does not have an effect on acute mechanical performance of a Novolimus-eluting BVS as assessed by OCT.