The invention of supraglottic airway devices (SGADs) has initiated a new era of modern airway management and it is considered to be an important milestone towards improving patient safety during anaesthesia. The LMA-Classic (inTRAvent Medical, Maidenhead, UK) was the first SGAD introduced into clinical practice in 1983 by Archie Brain and was regarded as a first-generation SGAD [1]. First-generation SGADs were just a simple airway tube that was initially designed for securing the airway as an alternative to an endotracheal tube during emergency situations. It subsequently showed benefits for patients undergoing general anaesthesia [2]. Over the years, numerous enhancements have resulted in the creation of improved second-generation SGADs that aimed to allow a higher positive airway pressure while reducing the risk of pulmonary aspiration by adding a gastric access port for evacuation of the stomach contents [3].
The i-gel (Intersurgical Ltd, Wokingham, UK) is an evolutionary example of the second generation of SGADs (Figures 1 and 2). It takes its name from the soft gel-like material from which it is made. Its shape, softness and contours accurately mirror the perilaryngeal anatomy to create the perfect fit and a reliable perilaryngeal seal without the need for an inflatable cuff. This key feature means the insertion of the i-gel is designed to be easy, rapid and consistently reliable. There are a few other special features of the i-gel that can provide additional benefits, including a gastric channel, an integral bite block, a buccal cavity stabiliser and an epiglottic rest. The gastric channel has proximal and distal ends that can provide an early warning of regurgitation, allowing for the passage of a nasogastric tube to empty the stomach contents and facilitate venting; the integral bite block reduces the possibility of airway channel occlusion; the buccal cavity stabiliser aids in insertion and eliminates the potential for rotation; and the epiglottic rest reduces the possibility of epiglottic ‘down folding’ and airway obstruction [4, 5].
FIGURE 1
Comparison of i-gel and Baska Mask supraglottic airway devices from anterior view; top: Baska Mask, bottom: i-gel
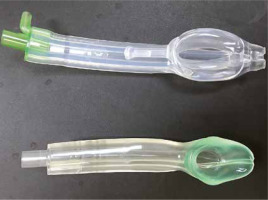
FIGURE 2
Comparison of i-gel and Baska Mask supraglottic airway devices from lateral view; top: Baska Mask, bottom: i-gel
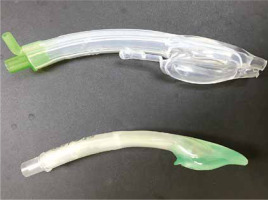
The Baska mask (Logikal Health Products, Morisset, NSW, Australia) is another innovative SGAD (Figures 1 and 2). It is marketed as a third-generation SGAD, but this claim is questioned and challenged by some authors [6]. It has an advanced self-sealing variable pressure cuff that can produce an effective seal with the larynx, which increases proportionately with the increase of airway pressure during positive pressure ventilation. The airway opening is also advanced, which provides superior seal patency and increased protection against gastric overflow. It also has superior gastric reflux drainage with a large distal aperture located at the upper oesophagus and open to the sump cavity for easy drainage of gastric fluid. A suction attachment is available and suitable for placement on either side of the drainage to keep the sump area clear and minimise the risk of aspiration. There is also a bite block to protect the airway tube from being compressed by a patient’s bite. The Baska mask also has an additional part that further differentiates it from the i-gel called an insertion tab, which is used for manually curving the mask for easy insertion. The Baska mask achieves a high seal pressure, effective ventilation and quick access to drain gastric contents [7, 8].
The similarity of these two SGADs are the non-inflatable cuff features, which are considered by some to be the main feature of the so-called third-generation SGADs in comparison to the earlier second-generation SGADs, such as the LMA-Proseal, LMA-Fastrack, etc. There have been very limited studies comparing the igel and Baska mask. Therefore, the aim of this study is to compare the effectiveness of the i-gel and the Baska mask in terms of quality of insertion, quality of ventilation and incidence of post-insertion complications. We hypo-thesised that the Baska mask as a newer SGAD with non-inflatable cuff is better in terms of quality of insertion, quality of ventilation and incidence of post-insertion complications than the i-gel.
METHODS
This was a single-blinded, randomised controlled trial, comparing the Baska mask and i-gel in patients undergoing elective surgery. The study was conducted after obtaining approval from the Human Research Ethics Committee of the Universiti Sains Malaysia (approval code: USM/JEPeM/17050246) and written consent from patients.
The inclusion criteria were patients aged 18 to 60 years, of American Society of Anesthesiologists (ASA) class I and II, and who had a body mass index (BMI) of less than 35 kg m-2. Patients were excluded from the study if they were pregnant, undergoing laparoscopic or head and neck surgery, had a previous history of difficult intubation or were at risk of gastric aspiration. Patients who required unforeseen tracheal intubation, muscle paralysis or unplanned intensive care unit admission post-operatively were withdrawn from this study.
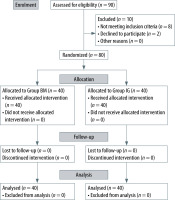
Patient eligibility screening was conducted during pre-operative assessment at least a day before surgery. A total of 80 adult patients who were scheduled for elective surgery were randomised to two groups using computer-generated randomisation software: Group BM: Baska mask (n = 40) and Group IG: i-gel (n = 40). The randomisation allocations were subsequently concealed in a sealed envelope. All selected patients were given a 3.75 to 7.5 mg premedication tablet of midazolam the night before the surgery. Upon arrival at the operation theatre (OT) reception area, the envelope containing the randomisation allocation was opened by the anaesthesia nurse to determine the type of SGAD that would be used for the respective patient. The selected SGAD was not revealed to the operator until just before the insertion (Figure 3).
Upon arrival inside of the OT, standard anaesthetic monitoring was applied to all patients, which included non-invasive blood pressure monitoring, an electrocardiogram, capnography and pulse oxi-metry. The preparation of the SGAD was done by the anaesthesia nurse. The size of the Baska mask and that of the i-gel were chosen based on the patients’ ideal body weight, as per the manufacturers’ recommendations for each device. The size recommendation for the Baska mask was size 3 for patients < 50 kg, size 4 for patients 50 to 70 kg and size 5 for patients > 70 kg, whereas the size recommendation for the i-gel was size 3 for patients 30 to 60 kg (small adult), size 4 for patients 50 to 90 kg (medium adult) and size 5 for patients > 90 kg (large adult). Preparation of the Baska mask and the i-gel with appropriate lubrication using lignocaine gel was done according to the manufacturers’ recommendations. The insertion technique for each device was also done according to the manufacturers’ guidelines.
All device insertions were performed by the anaes-thesia medical officer in charge in the specific OT. All anaesthesia medical officers were post-graduate specialty trainees in anaesthesia who had had more than 3 years of experience in anaesthesia practice and already had previous experience inserting at least 20 SGADs. The specific SGAD was prepared by the anaesthesia nurse and was handed to the ope-rator just prior to the insertion. The insertion was performed with the patient’s head in the neutral position. Successful placement was confirmed by the presence of adequate bilateral chest expansion and a satisfactory end-tidal carbon dioxide waveform. The insertion time, which was defined as the time taken from the moment the device was handed to the operator until satisfactory ventilation was achieved, was recorded by the anaesthesia nurse using a digital timer. After successful insertion, the ease of insertion was rated based on a 4-point verbal rating scale (VRS; Table 1). A maximum of three attempts was permitted for each patient and each new attempt was considered to be a re-insertion of the device.
TABLE 1
Ease of insertion scale for SGAD insertion
If the SGAD did not function effectively, the manipulations could be performed either by increasing the depth of insertion, rotating the SGAD or withdrawing the SGAD slightly. Each manipulation was recorded as a manoeuvre attempt, and if these manoeuvres were unsuccessful in achieving effective ventilation, the device was removed. If the problem was predominantly due to a large leak, a device that was one size larger was inserted. If the initial size was considered too large, a smaller device was inserted. A change in device size was recorded as an insertion attempt. If the SGAD insertion failed after three attempts, tracheal intubation was performed. Insertion failure was defined as either three unsuccessful attempts or if the entire process of insertion exceeded 120 s. This included the time when the airway device was removed from the mouth and bag-mask ventilation in between insertion attempts was needed. If patients had transient post-induction apnoea, ventilation was supported until adequate spontaneous ventilation returned. Anaesthesia was maintained with sevoflurane throughout surgery without the use of any muscle relaxants.
Heart rate, systolic blood pressure (mm Hg), diastolic blood pressure (mm Hg), mean arterial pressure (mm Hg), and oxygen saturation were recorded at baseline before induction, and at 1 min, 3 min, 5 min, 10 min and 20 min after SGAD insertion. The oropharyngeal leak pressure (OLP) was measured after successful insertion and satisfactory ventilation using the closed-circuit mechanical ventilator in the operating room. The airway pressure was gradually increased by keeping a flow rate of 4 L min-1 with a maximum pressure limit of 40 cm H2O. The pressure at which audible noise was detected by a stethoscope just lateral to the thyroid cartilage was recorded as the OLP. The peak airway pressure (PAP) in cm H2O was documented 5 min after successful SGAD insertion. The volume of expired and inspired tidal volume (VT) was also documented at 5 min after successful insertion.
After surgery was completed, sevoflurane inhalation was stopped and the patient was prepared for extubation. The airway device was removed when the patient was awake and fulfilled all the criteria of recovery from anaesthesia. Complications, such as injury of the teeth, gums or tongue, were assessed after the removal of the SGAD. The presence of blood staining on the SGAD was documented. The patient was then monitored at the post-operative recovery bay and was interviewed to assess whether complications of throat pain/soreness, nausea or vomiting were present. If a patient experienced post-operative nausea or vomiting (PONV), anti-emetics were administered. The degree of throat soreness was assessed using a 4-point VRS before discharge from the OT recovery bay. A rating of 0 was considered no pain, 1 as mild pain, 2 as moderate pain and 3 as severe pain.
The sample size was calculated using Power and Sample Size Calculations software, version 3.0 (January 2009, 1997–2009 by William D. Dupont and Walton D. Plummer). Based on the study by Ekinci et al. on number of insertion attempts, the failure rate among controls was 0.9999. If the true failure rate for experimental subjects was 0.825 [9] we were required to study 40 experimental subjects and 40 control subjects to be able to reject the null hypothesis with a power of study of 0.8. Therefore, the total sample size was 80.
The statistical analysis was conducted using SPSS software version 24.0 (IBM Corp., USA). The analysis for categorical data was conducted using the c2 test or Fisher’s exact test. The analysis for numerical data was conducted using the independent t-test or Mann-Whitney test. Power of study was 0.8 and a P-value of .05 was considered statistically significant.
RESULTS
A total of 80 patients were recruited for this study, and patients were divided into two groups with 40 patients in each group based on SGAD type. There were no significant differences in demographic profile in terms of age, gender, weight, height, BMI, Mallampati score, thyromental distance, mouth opening measurement or duration of surgery between Group BM and Group IG (Table 2).
TABLE 2
Demographic profile
In terms of ease of insertion, there was a significant difference between the two groups (P < 0.001). Group IG showed a higher percentage in the ‘very easy’ insertion category than Group BM (62.5% and 10.0%, respectively). There was also no failure of insertion in Group IG when compared to Group BM (0.0% and 5.0%, respectively). However, the majority of Group BM (77.5%) were still within the ‘easy’ insertion category, which was still considered to be a good outcome (Table 2). Group IG also showed significantly shorter median insertion time than Group BM (13.3 s [interquartile range, IQR 7.8] and 17.0 s [IQR 9.6], respectively; P < 0.001; Table 2). In terms of number of insertion attempts, there were no significant differences between the groups (P = 0.055). However, based on percentage, Group IG showed a slightly higher percentage in the successful single attempt category than Group BM (92.5% and 77.5%, respectively; Table 2). In terms of number of corrective manoeuvres after insertion, there was a significant difference between the two groups (P = 0.003), with Group IG showing a higher percentage in the no manoeuvre at all category compared to Group BM (92.5% and 72.5%, respectively; Table 2).
In terms of ventilation quality, Group IG showed a significantly lower value in generated PAP than Group BM (11.5 cm H2O [2.2] and 12.7 [1.8] cm H2O, respectively; P = 0.010; Table 3). However, the mean PAP for Group BM was still considered within a good range. There were no significant differences in other ventilation parameters, such as inspired and expired VT or OLP (Table 3).
TABLE 3
Quality of supraglottic airway device insertion
TABLE 4
Ventilation parameters and oropharyngeal leak pressure
TABLE 5
Complications of supraglottic airway device insertion
In terms of complications, there was a significant difference in terms of throat soreness between the two groups (P = 0.042). Group IG showed a higher percentage of no throat pain than Group BM (67.5% and 42.5%, respectively). There were no significant differences in other complications, such as blood staining on the device, PONV and airway trauma.
DISCUSSION
Our study was conducted to compare the two latest types of second-generation SGADs: the Baska mask and i-gel. Our results showed that the i-gel resulted in a significantly shorter median insertion time, a higher percentage in the ‘very easy’ ease of insertion category, a higher percentage in the no requirement of corrective manoeuvre category and a higher percentage in the no post-operative throat pain category than the Baska mask. However, the Baska mask showed superiority in the category of higher generated PAP, which might indicate a better cuff seal. Otherwise, there were no significant differences in number of attempts, inspired or expired VT, OLP and complications such as blood staining on the device, PONV and airway trauma.
There have been some recent studies comparing the Baska mask and i-gel and the conclusions have been mixed. A few studies have shown that the parameters of the i-gel are superior to the Baska mask, which is supported by our study. Bindal et al. conducted a comparison study between three types of SGADs – the Baska mask, i-gel and LMA-Classic – in 150 patients undergoing outpatient urologic interventions. Out of the few parameters assessed, only insertion and ventilation times were significantly different between the three groups. The Baska mask showed the longest insertion and ventilation times among the groups, with 12.04 s ± 6.25 s and 21.26 s ± 8.53 s, respectively. The shortest time was shown by the LMA-Classic group, which was 5.78 s ± 1.72 s for insertion time and 11.72 s ± 4.72 s for ventilation time. The first-attempt success rates were also highest for the LMA-Classic at 98%, followed by 92% for the i-gel and 88% for the Baska mask. Besides that, 20% of the Baska mask group required additional manoeuvres, which was more than the other groups. This study concluded that the LMA-Classic and igel showed superior results in insertion and ventilation times, first-attempt success rates and no additional manoeuvres than the Baska mask [10]. Kara et al. conducted a comparison study between the Baska mask and i-gel in 200 patients undergoing urologic surgery. The i-gel showed significantly shorter median insertion time than the Baska mask (7 s [5–12] and 14 s [6–25], respectively). However, there were no significant differences in the other parameters (the number of device placement attempts, sealed pressure and the number of post-operative complications) [11].
A few studies have highlighted the superiority of certain parameters of the Baska mask over the i-gel. Garg et al. compared these two SGADs in 100 female patients undergoing short, elective gynaecological procedures. The study concluded that the Baska mask offered a superior airway seal pressure and better ease of insertion than the i-gel. The airway seal pressure achieved was significantly higher with the Baska mask than the i-gel (35.8 cm H2O ± 10.3 cm H2O and 26.9 cm H2O ± 7.5 cm H2O, respectively). Other parameters and complications were comparable [12]. Another study by Sachidananda et al. in 50 patients undergoing minor surgical procedures also showed that the sealing pressure of the Baska mask was significantly greater than that of the i-gel (28.9 cm H2O ± 3.5 cm H2O and 25.9 cm H2O ± 2.5 cm H2O, respectively). Other parameters, such as first-time insertion success rate, insertion time and complications, were comparable between the groups [13]. Choi et al. compared 97 patients undergoing elective laparoscopic cholecystectomies and found that the Baska Mask demonstrated significantly higher OLP than the i-gel (29.6 cm H2O ± 6.8 cm H2O and 26.7 cm H2O ± 4.5 cm H2O, respectively). However, other parameters, such as insertion time, fibre-optic view of the glottis, the use of airway manipulation, heart rate, mean arterial pressure, PAP, lung compliance, and perioperative complications, were comparable [14]. A study by Kara et al. showed that the Baska mask provided a significantly higher PAP than the i-gel (15.8 cm H2O ± 0.9 cm H2O and 14.9 cm H2O ± 1.7 cm H2O, respectively) [11], which was supported by our study.
Even though our results showed that the i-gel was superior in some of the parameters, the margin of difference was actually not large. Perhaps, with more training for the right technique and early use of the insertion tab – which is a unique feature of the Baska mask – the ease of insertion will be improved. Familiarity with certain devices will improve the smoothness of the insertion. Even though the operators in our study were experienced in inserting other types of first- or second-generation SGADs, most of them were new to the i-gel and Baska mask, as they had only recently been introduced and become available in our centre. The inter-observer variability was also one of the limitations of our study. As described above, the Baska mask showed superiority in OLP in some of the studies. Even though our study showed comparable OLP, a higher generated PAP is reflective of good cuff seal. Compared to other recent studies comparing these two SGADs, our study assessed additional parameters on quality of ventilation, which were the value of expired and inspired VT. Both SGADs showed comparable value of these parameters, which gave an impression that the non-inflatable cuff SGADs were able to generate adequate VT during ventilation.